Cambrian Predator Wore Its Brain out in Front
Abstract
Anomalocaridids from the Cambrian explosion have puzzled paleontologists for years. And it is not just what they are but what they were evolving into that evolutionists have been concerned with. Discovery of the first anomalocaridid brain, which resembles the neuroanatomical structure of the modern velvet worm, sheds light on the evolutionary history of arthropods and of more complex animals, according the evolutionists. In reality, this animal—preserved in exquisite detail by sudden burial during the global Flood—reveals nothing about evolution but rather illustrates God’s design for even the “simplest” creatures.
Among the countless creatures preserved as fossils in the Cambrian “explosion” are anomalocaridids, a name meaning “abnormal shrimp.” (Not sure how to pronounce that big word? Listen to it at this website.)
What is an anomalocaridid? Enquiring minds want to know!

Lyrarapax unguispinus is an anomalocaridid from Lower Cambrian rock in Chengjiang in southwest China. This is the first anomalocaridid with preserved evidence that it had a brain. The mouth is the dark round structure, and the simple brain material is draped around it. Eyes—marked by the darker retinal pigment to each side—are connected to the brain. In front of this brain and the eyes are grasping frontal appendages (seen very well on the right in this view). These grasping appendages out in front of the eyes and head are a hallmark of anomalocaridids. Image by Peiyun Cong et al., via Nature.1
Scientists have been unable to agree on how to classify these extinct arthropod-like animals, but the discovery of an anomalocaridid dubbed Lyrarapax unguispinus with a well-preserved brain may have solved the mystery. Its brain was remarkably similar to the modern velvet worm’s and evolutionary researchers say they now know where anomalocaridids fit in evolutionary history: they believe these fierce, primitive predators of very little brain drove other animals to evolve better, smarter brains to escape from them.
Though Lyrarapax was only about five inches long, some anomalocaridids reached several feet. Found mostly in Cambrian (but also Ordovician and Devonian) rock layers from all parts of the globe, anomalocaridids include some of the largest animals in the Cambrian explosion. Conventionally assigned dates back to 520 million years ago, these legless invertebrates had large compound eyes, two jointed grasping frontal appendages, a non-mineralized exoskeleton of chitin, and a carapace over where the head should be.
If It Only Had a Brain!
Despite their name, anomalocaridids weren’t shrimp. Their single pair of grasping appendages, reaching out in front of where the brain should have been, is an oddity that has added to the mystery of what they were. Writing in Nature, Nicholas Strausfeld and colleagues explain, “Despite being among the most celebrated taxa from Cambrian biotas,anomalocaridids (order Radiodonta) have provoked intense debate.”2 Strausfeld, director of the University of Arizona's Center for Insect Science, says, “Our discovery helps to clarify this debate. It turns out the top predator of the Cambrian had a brain that was much less complex than that of some of its possible prey and that looked surprisingly similar to a modern group of rather modest worm-like animals.”3
Where Do You Keep Your Brain?
The anomalocaridid kept its simple bit of brain in front of its mouth, with clusters of nerve cells (ganglia) extending into the base of each frontal appendage. The eyes were behind the brain. Paleontologists say they now know where to put the anomalocaridid on the evolutionary tree of life because its brain so resembles the brain of the modern “onychophoran”—aka velvet worm—which also keeps its simple brain, with extensions into the antennae, tucked in front of its mouth and eyes.

This is a velvet worm (Aysheaia pedunculata) from the Burgess Shale. Modern velvet worms are terrestrial but look much like this one preserved in Cambrian rock. Image from Smithsonian National Museum of Natural History.4

This is a velvet worm, also known as an onychophoran. This one lives in Ecuador. Its anatomy hasn’t appreciably changed since the onychophoran in the other photo was buried in Cambrian sediment during the global Flood about 4,350 years ago. Each stubby leg has a little claw at the end. Velvet worm antennae are often referred to as “frontal appendages.” The brain of the modern velvet worm significantly resembles the newly discovered brain of another Cambrian invertebrate, the anomalocaridid. Image by B Kimmel, via Wikimedia Commons.
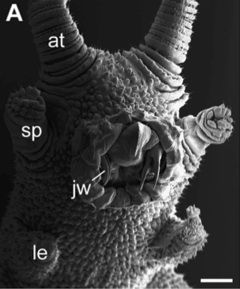
This magnified view of the underside of a velvet worm’s head shows the antennae (at), slime-squirting papillae (sp), front-most legs (le), and claw-like jaws (jw) inside its round mouth. The brain is located just at the base of the antennae, in front of the mouth. The extinct anomalocaridid had a similar arrangement, though without slime-squirters and with grasping appendages instead of antennae. Scale bar = 500 μm. Image by G. Mayer et al., via BMC Evolutionary Biology.5
The velvet worm is a tropical, terrestrial worm-like animal with unjointed pairs of legs, no exoskeleton, a pair of antenna (aka “frontal appendages”), and the ability to squirt slime to entangle its prey. Velvet worms resemble arthropods internally but externally look more like subtly segmented worms with antennae and fat legs. Modern velvet worms still look like the velvet worm Aysheaia in the Cambrian fossil record.
The velvet worm’s simple brain, extending into each sensory antenna, is located in front of its round mouth. Its head consists of claw-like jaws inside the mouth, slime-squirting papillae to the sides, and simple eyes (in most species) at the base of the antennae, which are non-grasping. With very different kinds of eyes, frontal appendages, and overall body plans, the anomalocaridid is certainly not a velvet worm, but Strausfeld says the velvet worm is the only living animal with a brain, frontal appendages, mouth, and eyes arranged like those in anomalocaridids:
These top predators in the Cambrian are defined by just their single pair of appendages, wicked-looking graspers, extending out from the front of their head. These are totally different from the antennae of insects and crustaceans. Such frontally disposed appendages are not found in any other living animals with the exception of velvet worms.6
Xiaoya Ma of the Natural History Museum in London, coauthor of the study, summed up the importance of this brain in determining not only what anomalocaridids are but perhaps more importantly their place in evolutionary history:
This is another contribution towards the new field of research we call neuropaleontology. These grasping appendages are a characteristic feature of this most celebrated Cambrian animal group, whose affinity with living animals has troubled evolutionary scientists for almost a century. The discovery of preserved brain in Lyrarapax resolves specific anatomical correspondences with the brains of onychophorans.7
Cambrian Brain Complexity
Finding complex brain material deep in Cambrian rock layers was initially a bit of a surprise to evolutionary paleontologists, who believe that complex brains shouldn’t have really evolved 520 million years ago. Back in 2012, Strausfeld, commenting on the comparatively more advanced brain of another “520-million-year-old” Cambrian invertebrate, Fuxianhuia protensa, said, “No one expected such an advanced brain would have evolved so early in the history of multicellular animals. . . . The rest of the animal is incredibly simple, so it's a big surprise to see a brain that is so advanced, as it were, in such a simple animal.”8
Now paleontologists know that not one or two but three basic brain plans coexisted in the Cambrian fossil record. “With this paper and our previous reports in Nature, we have identified the three principal brain arrangements that define the three groups of arthropods that exist today,” Strausfeld says. “They appear to have already coexisted 520 million years ago.”9
Lyrarapax unguispinus had a brain far simpler than that of Fuxianhuia, and therefore in evolutionary minds was less evolved. Both also had appendages on the head, near the brain. In fact Fuxianhuia had enough “legs on its head” to be dubbed a Cambrian “Cthulhu”! 10
But while the brain of Fuxianhuia is located behind the eyes, as seen in crustaceans, Lyrarapax has its brain out in front of its mouth and eyes. Thus there coexisted in Cambrian invertebrates more than one simple brain plan and more than one location for that simple brain. To evolutionary paleontologists, this is a big deal.
Sorting Out Arthropods
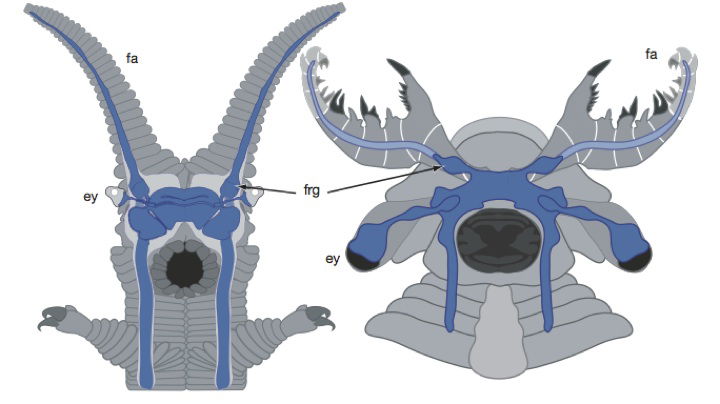
These drawings compare the neural anatomy (shown in blue) of the modern onychophoran (velvet worm) and the extinct anomalocaridid Lyrarapax unguispinus. The onychophoran (left) has antennae (fa=frontal appendages) in front of its eyes (ey) and optic nerves. The anomalocaridid (right) had grasping frontal appendages (fa) in front of its eyes and optic nerves. Behind this in each is a simple brain, draped over the mouth (in black). (frg=frontal appendage ganglia, the nerve supply for the frontal appendages.) Image by Peiyun Cong et al., via Nature.11
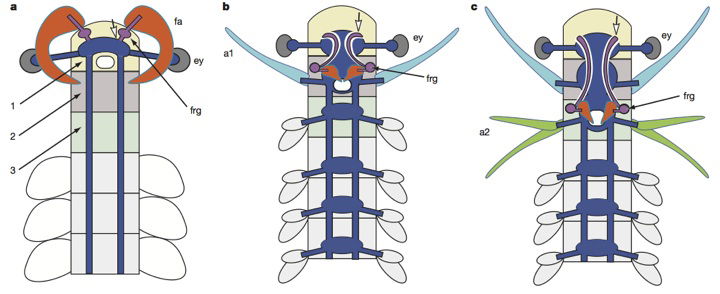
This is a hypothetical evolutionary sequence put forth in the study published in Nature, proposed to explain brain evolution and the evolutionary history of arthropods. According to the story, a “primitive” anomalocaridid (left) evolved a primitive brain (blue, with purple ganglia) attached to frontal appendages (fa) in front of its eyes (ey). Then the brain somehow migrated over evolutionary time to a position behind the eyes—to the third cephalic segment of the crustacean body (right, with purple ganglia and axon tracts that lead to the protocerebrum (open arrow). In this scenario, the frontal appendages (orange) transform into the crustacean’s upper mouth parts (orange). The creature in the center is a hypothetical transitional form invented for the purpose. Image by Peiyun Cong et al., via Nature.12
At issue is the much-debated evolutionary history of arthropods, the phylum that includes insects, spiders, and crustaceans. Evolutionists believe the last common ancestor of arthropods and “sister groups” like velvet worms would have had a much simpler body plan, a series of virtually identical body segments. This hypothetical ancestor would then have evolved specializations that transformed some segments into other structures. And in arthropods and other segmented creatures, appendages are associated with segments. Therefore, the discovery that a Cambrian arthropod even simpler than Fuxianhuia had grasping frontal appendages attached to its little bit of brain fuels the evolutionary story of how brains evolved.
Gregory Edgecombe of London’s Natural History Museum explains the evolutionary importance of “leg”-brain connection:
Being able to directly associate appendages with parts of the brain in Cambrian animals is a huge advantage. For many years now paleontologists have struggled with the question of how different kinds of appendages in Cambrian fossils line up with each other and with what we see in living arthropods. Now for the first time, we didn't have to rely just on the external form of the appendages and their sequence in the head to try and sort out segmental identities, but we can draw on the same tool kit we use for extant arthropods—the brain.13
The researchers do not simply report their findings in their paper in Nature but also sketch line drawings of a “hypothetical” ancestral creature to bridge the perceived gap between anomalocaridids and modern crustaceans. They show how brain tissue—that presumably evolved sticking out in front of an animal, hooked to its frontal appendages—migrated back along the body to the more common arthropod position while the front graspers transformed themselves into the upper mouthparts. (Don’t worry if all this seems a bit hard to follow, as it is an imaginary evolutionary scenario anyway.)
Predatory Motivation to Evolve
Not content with having dressed up their fascinating fossil with this bit of fiction, the researchers have another tale to tell, a tale of how this simple-minded predator, the anomalocaridid, terrorized the Cambrian world enough to drive other animals to evolve bigger better brains so they could get away. Strausfeld says, “With the evolution of dedicated and highly efficient predators, the pressure was on other animals to be able to detect and recognize potential danger and rapidly coordinate escape movements. These requirements may have driven the evolution of more complex brain circuitry.”14
Solution to Cambrian Complexity
The “Cambrian explosion”—the sudden appearance of innumerable complex life-forms deep in the fossil record without evidence of the evolutionary antecedents leading up to them—is a perpetual mystery for evolutionary scientists. The presence of complex brain tissue among these creatures supposedly more than half a billion years ago is equally unexplained by evolutionary thinking. This particular fossil, even with its “simple” brain, illustrates that complexity. The researchers do not attempt to explain how this creature originated but rather try to weave a path to explain how it reveals the evolutionary origin of the brain and connects the dots in the evolutionary history of arthropods.
Lining up their brains to tell an evolutionary tale is an imaginative exercise at best.
But just as struggling to line up the appendage anatomy of Cambrian critters to sort out their history has been fruitless, so lining up their brains to tell an evolutionary tale is an imaginative exercise at best. Classification into brain plans, an exercise that reveals some of the diverse designs God used in various creatures, even those we consider relatively simple, is all well and good. When evolutionists then try to connect the dots and insist that these various plans explain the origins of the brain in more complex animals and man, they are filling in the transitions from one kind of creature to the next without a biological basis.
Since we see modern animals varying and reproducing only within their created kinds, it is reasonable to instead see that our Creator God equipped all kinds of animals in the beginning with the anatomical equipment they needed to live in their various niches. Anomalocaridids, like onychophorans then and now, were apparently equipped to integrate sensory data about their environment using a very simple brain. There is no reason to assume this arrangement had to evolve in the first place or was an evolutionary way station en route to greater complexity. This remarkably well-preserved fossil is a testimony to the rapid burial in the early days of the global Flood described in Genesis chapters 6–9. Its anatomy is a testimony to the greatness of the Designer, God, who made all kinds of creatures in the beginning, about 6,000 years ago.
Further Reading
. . . about the Cambrian explosion and its creatures:
. . . about brains:
Footnotes
- Peiyun Cong et al., “Brain Structure Resolves the Segmental Affinity of Anomalocaridid Appendages,” Nature (July 16, 2014): 538–542, doi:10.1038:nature13486, figure 1.
- Ibid.
- University of Arizona, “Brain of World’s First Known Predators Discovered,” ScienceDaily, July 16, 2014, https://www.sciencedaily.com/releases/2014/07/140716131628.htm.
- “Burgess Shale,” Smithsonian National Museum of Natural History, http://paleobiology.si.edu/burgess/aysheaia.html.
- G. Mayer et al., “A Revisions of Brain Composition in Onychophora (Velvet Worms) Suggest That the Tritocerebrum Evolved in Arthropods,” BMC Evolutionary Biology 10, no. 255 (2010): figure 1, 10:255, doi:10.1186/1471-2148-10-255.
- University of Arizona, “Brain of World’s First Known Predators Discovered.”
- Ibid.
- Quoted in Elizabeth Mitchell, “Cambrian Invertebrate with Neural Complexity Similar to Modern Insects,” Answers in Genesis, October 20, 2012, https://answersingenesis.org/extinct-animals/cambrian-invertebrate-neural-complexity-modern-insects.
- University of Arizona, “Brain of World’s First Known Predators Discovered,” ScienceDaily, July 16, 2014, https://www.sciencedaily.com/releases/2014/07/140716131628.htm.
-
In the search for the evolutionary origin of the velvet worm’s antennae—aka frontal appendages—and for the evolutionary origin of arthropods, evolutionists have resorted to clues in the embryological development of these animals. Hox genes control the expression of limb development genes in many creatures. The Hox gene that controls the development of the velvet worm’s antennae also controls the development of a sensory tentacle near the mouth of a segmented marine worm and of a mouthpart of some insects. This is not the same Hox gene that controls the development of antennae in most arthropods. (This research and the debate surrounding it is discussed in numerous articles including Bo Joakim Eriksson et al., “Head Patterning and Hox Gene Expression in an Onychophoran and Its Implications for the Arthropod Head Problem,” Development Genes and Evolution 220 (June 22, 2010): 117, doi:10.1007/s00427-010-0329-1; Ralf Janssen et al., “Onychophoran Hox Genes and the Evolution of Arthropod Hox Gene Expression,” Frontiers in Zoology 11, no. 22 (March 5, 2014): doi:10.1186/1742-9994-11-22; and Georg Mayer et al., “A Revision of Brain Composition in Onychophora (Velvet Worms) Suggest that the Tritocerebrum Evolved in Arthropods,” BMC Evolutionary Biology 10, no. 255 (August 21, 2010): doi:10.1186/1471-2148-10-255 as well as in Cong et al., “Brain Structure Resolves the Segmental Affinity of Anomalocaridid Appendages.”)
Because this differs from the usual developmental pattern for antennae, evolutionists believe the velvet worm’s non-grasping, sensory frontal appendages with close connection to its anteriorly placed simple brain may help explain the evolutionary history of arthropods. However, as we often point out, embryological development within a single species follows the genetic instructions of that creature and neither recapitulates its evolutionary history nor reveals the evolutionary pathway supposedly taken by other kinds of animals. See “.”
- Cong et al. “Brain Structure . . . .”
- Ibid.
- University of Arizona, “Brain of World’s First Known Predators Discovered.”
- Ibid.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis