
Giant Panda’s Vegetarian Plight: An Evolutionary Dilemma?
News to Know
Abstract
Despite its carnivore-like gut, the giant panda is a reminder of the pre-fall world in which no bear was carnivorous.
News Sources
- Science: “Pandas’ gut bacteria resemble carnivores’”
- Nature World News: “Giant Pandas Meant to Eat Meat, Not Bamboo”
Is the giant panda a poorly evolved vegetarian? Or highly specialized, well-designed herbivore, a living link to a time when all animals were vegetarians? Despite having teeth, jaws, and a short digestive tract typical of the more carnivorous members of the bear family, the giant panda depends on a bamboo diet. Though occasionally enjoying a meaty meal,1 the giant panda subsists primarily on a daily diet of 20 to 30 pounds of bamboo leaves, stems, and shoots. The giant panda is an endangered species, and its vulnerability to habitat destruction makes understanding of how it meets its nutritional needs of great importance if it is to be protected rather than allowed to succumb to the evolutionary ideal of survival of the fittest.2
Survival of the Veggie Bear

Giant pandas, like these at the Chengdu Research Base of Giant Panda Breeding, must spend most of their time eating, as they only digest about 17% of the bamboo they eat. Image courtesy of Chengdu Research Base of Giant Panda Breeding. View larger image.
Scientists have long puzzled how the giant panda survives on its bamboo diet. It digests only about 17% of its cellulose-rich but protein-poor diet, and it must spend most of its time eating in order to consume 12-15% of its body weight in bamboo daily. Most grass-eating3 mammals have long digestive tracts populated by various cellulose-digesting microbes. The giant panda is notably lacking in the usual cellulose-digesting bacteria, and its digestive tract is too short to take full advantage of any microbial helpers that might be there. Some possible cellulose-digesting bacteria—Clostridia species—were identified in the panda gut as possible helpful agents in a 2011 study, but a new study of the panda’s abundant output casts doubt on this. A team of scientists from China’s Chengdu Research Base of Giant Panda Breeding confirms in the journal mBio that the giant panda’s gut is inhabited by pretty much the same microbes that live in the gut of carnivores, and those vary seasonally.4 Even the Clostridia species that the giant pandas in their study harbor are not consistently those associated with cellulose digestion.5 Lead author Zhihe Zhang explains the panda’s problem:
Unlike other plant-eating animals that have successfully evolved, anatomically specialized digestive systems to efficiently deconstruct fibrous plant matter, the giant panda still retains a gastrointestinal tract typical of carnivores. The animals also do not have the genes for plant-digesting enzymes in their own genome. This combined scenario may have increased their risk for extinction.6
Lacking a long digestive tract, suitable enzymes, and a cellulose-digesting microbial population (microbiome), the giant panda, according to coauthor Xiaoyan Pang, represents an evolutionary failure:
This result is unexpected and quite interesting, because it implies the giant panda's gut microbiota may not have well adapted to its unique diet, and places pandas at an evolutionary dilemma.7
Specifically the study’s authors consider the panda and its gut microbes a coevolutionary failure, writing:
The giant panda appears not to have evolved a gut microbiota compatible with its newly adopted diet, which may adversely influence the coevoutionary fitness of this herbivore.”8
Evolutionists assume the symbiotic dependence of an animal and its microbiome to be evidence they evolved jointly. However an ecological relationship that promotes such co-selection and mutual dependence cannot explain the origin of either bears or microbes because, for one thing, both would have to already exist before either could evolve into being. Complex symbiotic relationships developing under the influence of natural selection offer no support for molecules-to-man evolution.

This is Tai Shan, a giant panda at the Smithsonian National Zoological Park in Washington, DC. Giant pandas eat primarily bamboo, dexterously gripping and stripping it with their famous “panda’s thumb.” Image courtesy of Wikimedia Commons.
While its short digestive tract and lack of a herbivorous microbiome magnify the giant panda’s habitat-dependent danger by making the availability of a continuous large supply of various bamboo species essential to its survival, these facts are not the result of evolutionary failure. Furthermore, the giant panda’s specialized adaptations make it well suited for its ecological niche and in no way demonstrate any sort of molecules-to-man evolution.
Bears
Bears, including giant pandas, are all members of the mammalian family Ursidae.9 While other bears are native to Eurasia and the Americas, the giant panda (Ailuropoda melanoleuca) is native to several regions of southwest China. Larger (Ailuropoda baconi) and a smaller (Ailuropoda microta) extinct species have also been found in China’s fossil record. Despite its unusual diet and the unique “panda thumb” that helps it hold onto its dinner, the giant panda is thought by evolutionists and many creationists to share a common bear ancestor with other bears.
The giant panda’s teeth, jaws, and short digestive tract are like that of other bears. The giant panda has a large radial sesamoid10 bone and an enlarged accessory wrist bone that enable it to grasp and strip the bamboo on its menu. These structures are of only an insignificant size in other bears.11 Not only anatomy but also genetic and biochemical evidence supports the giant panda’s position as a well-adapted herbivorous member of the bear “kind,” a bear variation that likely diverged from other bears in the Ursidae family. Yet variation within a created kind is consistent with biological observations and the biblical record, which indicates in the opening chapter of Genesis that God designed each kind of animal to subsist on vegetation and to reproduce “after its kind.”
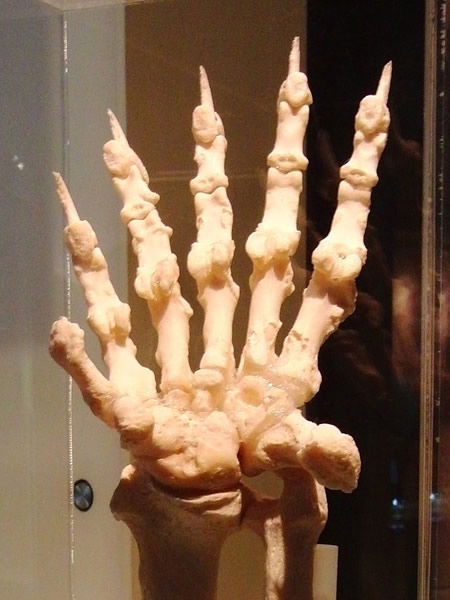
This left forepaw of a giant panda shows the enlarged wrist bone (to the left of the “fingers”) and the large radial sesamoid bone (shown here projecting on the right) that together equip the panda to efficiently manipulate the bamboo they must consume in large quantities. (Read more about it.) Image courtesy of National Museum of Nature and Science, Tokyo, Japan. Momotarou2012.
The original bears that God made, like all other animals, were vegetarians. Most bears are now omnivorous, and most eat meat or fish though they are able to survive on fruit, berries, nuts, and honey. The giant panda’s specialized preference for bamboo, a food with less available nutrition than the usual veggie-selections of other bears, makes it unusual. However, having to eat most of the time and seasonally forage in different regions is not exactly a major problem for an animal—at least so long as it habitat is not destroyed. Furthermore, most wild animals—and even many humans in this sin-cursed world—are subject to both seasonal deprivations12 and the vicissitudes of nature and man that affect the availability of their food supply. That does not mean they are poorly evolved but only that the designs and adaptations they possess are sometimes not adequate to face the challenges in a sin-cursed world.
The giant panda is not a victim of evolution-gone-wrong.
Carnivorous behavior entered the world God made in the wake of man’s sin. And in the wake of the global Flood, the animals disembarking from the Ark faced a world in which habitats and resources doubtless differed from those available before the Flood. While some animals became extinct—and still do—many adapted to the various ecological niches in the non-ideal post-Flood world. For many animals this evidently meant dependence upon a carnivorous diet to meet their nutritional needs. (Read more about this in cats, for instance, in “No Taste for Meat?.”) And yet while bears became or had become meat-eaters, the giant panda evidently remained largely vegetarian or reverted to its vegetarian past. It was able to chew bamboo with its strong jaws and large teeth. And it was able to employ a pincer grip using a uniquely designed wrist bone and large sesamoid bone flanking its five fingers to grip and strip bamboo. The giant panda’s adaptations for its specialized herbivorous diet may have fit it for its environment through the ordinary processes of natural selection and other mechanisms, or the Creator’s design—and we need not be dogmatic about which—and its specialization may ultimately spell its doom, but the giant panda is not a victim of evolution-gone-wrong.
Balanced Diet
Furthermore, while the giant panda’s nutritional adaptations remain puzzling, its behavior reveals an additional adaptation that, like its bamboo-grabbing paws, demonstrate its fitness for its ecological niche. Like all animals the giant panda needs a nutritionally balanced diet. And some animals seem to know how to balance their diet. A study published online in Functional Ecology in 2014 showed that the giant panda ranges about seasonally to locate the foods that meet its needs, even within the constraints of its bamboo menu.13 Primatologist Jessica Rothman, commenting on the study, noted, “There is strong evidence that animals try to forage as effectively as possible to meet their nutritional needs, mixing dietary items to provide a full complement of nutrients. In areas with only one edible plant, animals may try to consume different parts of the same food.”14
Researchers found that giant pandas do just that. By tracking pandas with GPS collars in the Qinling Mountains, they discovered that the animals forage according to the seasonal life cycle of the two bamboo species in the region. The species—wood bamboo and arrow bamboo—inhabit different elevations and therefore produce sprouts and leaves at different times. Younger bamboo shoots are richer in nitrogen and phosphorus, providing more protein than older shoots. The pandas migrated to higher elevations as the shoots at lower elevations matured, following the more nutritious growth. Bamboo shoots are low in calcium, however, and after mid-summer the pandas’ migratory patterns followed the growth of bamboo leaves, which are richer in calcium. Giant pandas mate in the spring, but like other bears their embryonic implantation is delayed. Implantation occurs when their calcium intake, needed for bone growth and milk production, increases. The giant panda cubs are born in late summer at a time when the mothers are feeding on the calcium rich leaves.15
We see in the giant panda not a poorly evolved bear but a vegetarian equipped to survive.
The giant panda’s nutritional needs are highly dependent on the availability of multiple species of bamboo in its habitat. While the giant panda lacks digestive efficiency, its devotion to eating over more energetic activities, its unique bamboo-gripping hands, and its nutritionally optimized foraging patterns have made the giant panda well suited to its ecological niche. We see in the giant panda not a poorly evolved bear but a vegetarian equipped to survive, however habitat dependent it is in today’s relatively inhospitable post-Flood world. Protection of the giant panda as an endangered species may rely on further investigations of how it meets its nutritional needs and keeping a close eye on preserving its habitat; however, there is no reason to assume this is an animal that evolution left behind. Rather it is a treasure trove of good designs, equipped by our Creator to adapt and survive—even if not ideally—to its unique place in the cursed creation. Moreover, the giant panda’s diet is a reminder of the vegetarian nature of all animals in the good world God created, before it was spoiled by the sinful rebellion of mankind.
Like the giant panda, people must also struggle to thrive in this sin-cursed world, though human struggles are generally of a far different nature. While people cope creatively with a world of suffering and death, the ultimate solution to our greatest plight involves trust in Jesus Christ, by whom and for whom all things were created (Colossians 1:16–17), as the Savior who sacrificed Himself for us.
Further Reading
For More Information: Get Answers
Remember, if you see a news story that might merit some attention, let us know about it! (Note: if the story originates from the Associated Press, FOX News, MSNBC, the New York Times, or another major national media outlet, we will most likely have already heard about it.) And thanks to all of our readers who have submitted great news tips to us. If you didn’t catch all the latest News to Know, why not take a look to see what you’ve missed?
(Please note that links will take you directly to the source. Answers in Genesis is not responsible for content on the websites to which we refer. For more information, please see our Privacy Policy.)
Footnotes
The giant panda occasionally eats meat, such as a rodent or dead animal.
-
Read more about the conundrum of conservation created by the evolutionary worldview in Brian Thomas, “Should We Let the Pandas Die Off?,” Institute for Creation Research, October 5, 2009, http://www.icr.org/article/should-we-let-pandas-die-off/. ICR author Brian Thomas explains,
Evolution, a system founded on continual cycles of death in which the less fit perish, provides no room for sentimentality over the loss of a weak species. In the biblical worldview, however, each creature has inherent value as a unique creation of God, a view that makes sense of mankind’s innate concern for living things. If evolution is true and if humans are really nothing more than the end products of colliding molecules, winners of a “Darwinian lottery,” then why should they care if some species don’t make it?
Bamboo is a grass, not a tree.
Z. Xue et al., “The Bamboo-Eating Giant Panda Harbors a Carnivore-Like Gut Microbiota, with Excessive Seasonal Variations,” mBio 6, no. 3 (May/June 2015): e00022-15, doi:10.1128/mBio.00022-15.
The failure to confirm the previously documented presence of cellulose-digesting bacteria in the current study might be related to the fact the subjects were captive-born residents of a panda preserve whose diets were supplemented with steamed bread. Therefore, the gut flora of these animals might have been subject to different selective pressures than those in the 2011 study. Future studies may shed more light on this matter. Nevertheless, it is clear that the giant panda faces substantial nutritional challenges.
Jenna Iacurci, “Giant Pandas Meant to Eat Meat, Not Bamboo,” Nature World News, May 20, 2015, http://www.natureworldnews.com/articles/14778/20150520/giant-pandas-meant-to-eat-meat-not-bamboo.htm.
Ibid.
Z. Xue et al., “The Bamboo-Eating Giant Panda Harbors a Carnivore-Like Gut Microbiota, with Excessive Seasonal Variations.”
Incidentally, the red panda is not a bear, but rather is something more akin to a raccoon. The sole member of the Ailuridae family, it also eats mainly bamboo, generously supplemented by an omnivorous selection of insects, eggs, birds, and mammals.
A sesamoid bone is a bone that develops within a tendon or joint capsule.
T. Hennigan, “The Case for Holobaraminic Status in Bears (Family Ursidae) and the Implications within a Creation Model of Ecology,” Creation Research Society Quarterly (Spring 2010) 46: 271–283.
Giant pandas, like many other animals, seem to have a higher mortality rate associated with the stress of winter. Fifty-two percent of the 25 dead or sick wild giant pandas observed over the course of 37 years in the Foping Reserve were noted in the months following winter. The authors conclude, “The peak in panda mortality in March and April is also consistent with an interpretation that the extended low quality of the winter diet of leaves is nutritionally stressful, highlighting the need for both mother and offspring to forage for limiting nutrients.” But of course this has nothing to do with molecules-to-man evolution. (Reported in Nie et al., “Obligate Herbivory in an Ancestrally Carnivorous Lineage.”)
Ibid.
Sandhya Sekar, “How Pandas Survive on Their Bamboo-Only Diet,” Science, July 9, 2014, http://www.sciencemag.org/news/2014/07/how-pandas-survive-their-bamboo-only-diet.
Nie et al., “Obligate Herbivory in an Ancestrally Carnivorous Lineage.”
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis


