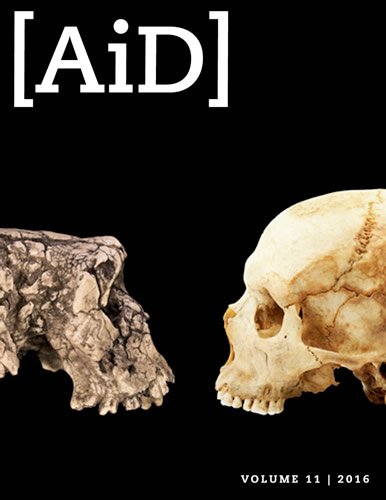
Is Your Allergy-Prone Immune System the Result of Neanderthal Influence?
News to Know
Abstract
Prone to allergies? Perhaps you can blame Neanderthals in your past.
Neanderthals are getting the rap for our allergies. Or rather the early modern humans who mixed with them are.
Recent data suggests that modern Eurasians inherited 1%–6% of their genomes from extinct people groups like Neanderthals and Denisovans. Are those genes a blessing or a curse? Two separate teams of scientists, led by Janet Kelso of the Max Planck Institute for Evolutionary Anthropology and Lluis Quintana-Murci of the Pasteur Institute, have found that three of the genes that control our immune system’s first line of defense bear a significant footprint from our archaic human cousins.
For Whom the Bell Doesn’t Toll
A strong and efficient immune system would have been vital for survival in a world without the benefits of modern medicine. People who were ill-equipped to fight infection were more likely to die off. And while our “adaptive” immune system must be educated through exposure, our “innate” immune system is inborn, its suitability to meet infectious threats governed by a person’s genes.
In centuries past, people with the most robust immune systems would tend to survive and pass on their good immunity-related genes to their offspring. Mutations that diminish immunity would naturally be selected against in a microbial-rich, dangerously infectious environment. Reasoning that those genes that vary least are likely among the most important ones in our immune system, scientists led by Quintana-Murci searched for human immunity-related genes that have not changed much over time. Kelso’s team, in an effort to determine whether the archaic human contribution to modern genomes has conferred lasting benefits or undesirable consequences, focused its analysis on those immunity genes that statistically have the largest Neanderthal footprint.
Three specific “TLR” genes got the attention of both groups—TLR1, TLR6, and TLR10. These genes code for three of the ten “Toll-like receptors” in our innate immune system. Clustered together on our fourth chromosome, they are among the modern human genes that exhibit the greatest Neanderthal contribution.1 These three TLRs are pre-programmed to recognize bacterial, fungal, and parasitic components and to trigger a rapid response to them.
The researchers suspect that for many years Neanderthal and Denisovan populations in Europe and Asia were selectively winnowed down to those best able to cope with the pathogens in the region. Then as early modern humans moved into the area and mingled with the inhabitants, they produced children who inherited immunity genes already optimally attuned to the challenges of the local environment. This is a reasonable scenario for how the modern human genome received and retained an unusually large dose of genes that provide innate immunity, the immunity we are born with. Of course variations, or in this case natural selection against variants in the archaic TLR genes, have nothing to do with molecules-to-man evolution. Whether Neanderthal or Denisovan or early modern human, all these people were simply humans. They had human DNA and human anatomy with the same sort of minor variations we see among the world’s modern people groups.
Innate and Adaptive Immunity
Our bodies are protected by two basic sorts of immunity. We inherit our innate immunity. It requires no prior exposure to possible pathogens. The innate immune system is designed to mount a response to potentially dangerous invaders within hours. A newborn, with no prior exposure to pathogens, would otherwise have little defense against life-threatening infections. And people of any age, encountering infectious agents their bodies have never met, could be in big trouble if it were not for the innate immune system.
Our ten Toll-like receptors (TLRs) are our first line of immunological defense. Present at birth, they are already programmed by the genes that encode them to recognize patterns of microbial presence. Some TLRs sit astride cell membranes and others reside inside cells, each alert to the presence of viruses, bacteria, fungi, and other microbial invaders that slip past our skin and mucous membranes. Once they detect an invader, TLRs trigger a number of cellular responses to destroy the microbial interloper.
“When the body detects that there is some foreign substance in the body,” Kelso explains, “these are the guys that react immediately,”2 triggering release of various chemical messengers and mobilizing diverse cells in the immune system.
The adaptive immune system is trained as it matures. Part of that training comes from exposure to microbes and other irritants. While Toll-like receptors are the first-responders in our innate immune system, they also help educate the cells in the adaptive immune system. To do this, TLRs prompt “dendritic cells” to capture some of the invading material and present it to the cells in the adaptive immune system. It is the adaptive immune system that develops antibodies to common infections, increasing our immunity to them after our first exposure. The adaptive immune system requires time to do this. Ultimately, though, it provides our bodies with a long-lasting, targeted, ready defense against specific pathogens.
Did Neanderthals Have the Sniffles?
The same wonderful immune system that protects us from pathogens sometimes overreacts to things that are not dangerous, resulting in annoying or even dangerous allergic reactions. Kelso explains, “What you have is . . . an increased reactivity to potential pathogens, but you also have, as a kind of consequence of that, an increased reactivity to things that are not pathogenic, things like pollen and pet hair.”3 Kelso’s group found that archaic versions of TLR1, TLR6, and TLR10 genes are more prevalent in today’s allergy-prone people. “I suppose that some of us can blame Neanderthals for our susceptibility to common allergies, like hay fever,” says Kelso.4
We don’t really know whether Neanderthals suffered from allergies or just passed along the genetic “raw material” for developing them.
Better odds for survival may have come at a price, if not for Neanderthals, at least for us. Scientists are still trying to work out why allergy-prone people develop their allergies or what can be done to prevent it. There are a number of possibilities still under investigation, and because the innate immune system helps teach the adaptive immune system what it should respond to, both are likely involved. Therefore, we don’t really know whether Neanderthals suffered from allergies or just passed along the genetic “raw material” for developing them.
Present Protection From the Past
Anthropological evidence suggests that Neanderthals were already living in Europe and Western Asia before early modern humans moved there. “They were likely well adapted to the local climate, foods, and pathogens. By interbreeding with these archaic humans, we modern humans gained these advantageous adaptations,” Kelso suggests. “What has emerged from our study as well as from other work on introgression,” she says, “is that interbreeding with archaic humans does indeed have functional implications for modern humans, and that the most obvious consequences have been in shaping our adaptation to our environment—improving how we resist pathogens and metabolize novel foods.”5
Regardless of their allergic nature, some of the genes (or rather the particular alleles of those genes) that have come down from archaic people to modern populations were protective. One archaic allele, for instance, is consistently associated with a reduced susceptibility to Helicobacter pylori, a bacterial infection associated with peptic ulcer disease.6 Because the innate immune system is our first defense against dangerous pathogens, it seems reasonable that in the ancient world people with the best innate immune systems were most likely to survive and pass on their genes to future generations.
Another case of a genetic variation traceable to an archaic human population, the gene that still enables native Tibetans to adapt to extremely high altitudes, demonstrates that genes likely introduced into the modern human gene pool by now-extinct groups of humans can have a beneficial effect.
Neanderthals, Our Neighbors From the Past
Though Neanderthals have long been viewed as brutish primitive creatures somewhat lower on the evolutionary scale than modern humans, the more we discover about them, the more we find out that they were—as Bible-believing creationists have long maintained— simply a group of human beings not all that different from us. They even organized their homes and heated water for personal use!
There is a substantial Neanderthal footprint in our genetic past. That is no surprise, for from the biblical account of history we understand that Neanderthals and early modern humans had to all be descendants of Adam and Eve and of Noah’s family. They were not sundry evolutionary products—a position neither biblical history nor experimental biology supports. They were fully human, the descendants of people who dispersed from the Tower of Babel some time after the global Flood of Noah’s day. That our innate immunity is heavily represented in our archaic DNA is a reminder that those people who faced that post-Flood world were equipped by God’s design to deal with the varying conditions they faced.
Highly Conserved or Heavenly Foresight?
We can be grateful that God designed an immune system that is equipped to protect us from microbial dangers in this sin-cursed world from the moment we enter it, and that we are likewise prepared to adapt and mount a strongly targeted response to dangers as they present themselves. Our immune system is even able to keep in check those bacteria that live symbiotically inside us and are necessary for the proper functioning of our bodies.
God designed an immune system that is equipped to protect us from microbial dangers.
Toll-like receptors of various kinds are also found in many animals. There are similar receptors in invertebrates. In fact, the first Toll-type of receptor was discovered protecting fruit flies from fungal infection. Some of the molecular building blocks from which TLRs are formed are found in plants where they are also involved in plant immunity to infection. Evolutionists are therefore fond of saying that these important components of innate immunity are “highly conserved,” especially throughout the animal kingdom. However, it should be no surprise to Bible-believers to find that Toll and Toll-like receptors are a common design found throughout the world God made.
Furthermore, the TLRs do not act alone. Even in the fruit fly the Toll receptor triggers cellular production of antifungal peptides. TLRs coordinate with each other, with diverse cell types, and with many other molecular and cellular components of the immune system to defend the host. They orchestrate an inflammatory response by initiating a network of domino effects designed to combat invading microbes. And they prompt dendritic cells to engulf dangerous microbes and show them to maturing cells in the adaptive system. The immune systems of which TLRs are a crucial component are irreducibly complex. TLRs alone would be fairly useless, even if they and the genetic information coding for them could have come into being through the random natural processes of evolution. Those taking an objective look at evolutionary claims should likewise wonder how these receptor molecules, with such enormous capabilities, also acquired the inborn heritable ability to recognize and respond to numerous components common to microbial threats, threats as diverse as viruses, bacteria, fungi, and protozoans—threats to which the host and the host’s ancestors may have never been exposed.
While man’s sin brought a curse upon this world and all the kinds of life in it, we should not be surprised to see that God had equipped the living things He made to cope with the changing and challenging conditions they would face over time. God designed this world to be inhabited (Isaiah 45:18), filled with the life He created in the beginning, in spite of man’s sin and the sentence of death to which it led. And in the fullness of time (Galatians 4:4–5), He also sent Jesus Christ into this world to offer eternal life to rebellious man, again offering life in the face of judgment and danger and death.
Pre-existing Conditions?

Though Neanderthal people living in Europe and Asia in the centuries after dispersion from the Tower of Babel eventually died out, their legacy lives on in many modern people of Eurasian ancestry. Just as we may inherit a greater risk of developing heart disease, diabetes, or cancer from our immediate family, so many of us may have inherited a greater risk of developing a variety of conditions from these more remote archaic ancestors. Scientists at Vanderbilt University examined the electronic medical records of about 28,000 people of European ancestry to find out what conditions might occur more commonly in association with a Neanderthal genetic contribution. A greater risk for depression, sun-related precancerous skin diseases, and coronary atherosclerosis are among the conditions that occur more frequently in those people with the associated archaic alleles in their genomes.
Why would people with these problems be able to survive in their harsh world and pass those conditions on to us? Remember there is not a particular gene known to consistently produce any of these problems. Their causes are complex and not fully known, but certain genetic markers are associated with a greater risk of developing them. The scientists reporting in Science suggest that one factor tying them together is the fact that Neanderthals lived in northern latitudes with less sun exposure than people from the tropics. Thus, sun-associated skin disease wouldn’t have been as much of a problem for them.
Seasonal depression—mood swings related to the length of days—is common today. The authors note, “Depression risk in modern human populations is influenced by sunlight exposure, which differs between high and low latitudes, and we found enrichment of circadian clock genes near the Neandertal alleles that contribute most to this association.”7 Were Neanderthals immune to seasonal affective disorder or more affected by it? We don’t know, but their legacy seems to be a greater vulnerability to it.
Additionally, the researchers found that archaic alleles were associated with hypercoagulable blood and a slight variation in carbohydrate metabolism. They suggest because Neanderthals likely consumed a somewhat different diet from modern people, a variation in carbohydrate metabolism, harmless or even helpful to them, is now a problem for many of us. And if Neanderthals lived a high-risk, active, outdoor lifestyle, blood that clotted a little more easily might have helped them live another day. Today, however, in populations that commonly push the envelope of “threescore years and ten” (Psalm 90:10), such an “advantage” would increase the risk of strokes and heart attacks.
Thus natural selection would not necessarily weed out alleles that might be a problem today. In the same way, a powerful and robust immune system would likely keep Neanderthals alive longer, while today most of us would perhaps prefer a little less immunity to things our bodies may never encounter and a little less hay fever.
News Sources
- NPR: Itchy Eyes? Sneezing? Maybe Blame That Allergy On Neanderthals
- The Washington Post: Got Allergies? Blame the Neanderthals
- ScienceDaily: Neanderthal Genes Gave Modern Humans an Immunity Boost, Allergies
- STAT: Thank Neanderthals for Your Strong Immune System—and Your Allergies
- Science: “Neandertal Genes Linked to Modern Diseases”
Further Reading
- Neanderthals vs. Humans: Are They Different?
- Modern Humans with Neanderthal History
- Does the Creation Model Make Predictions? Absolutely!
- Does Hugh Ross Believe in Soulless Ancient Humans?
For More Information: Get Answers
Remember, if you see a news story that might merit some attention, let us know about it! (Note: if the story originates from the Associated Press, FOX News, MSNBC, the New York Times, or another major national media outlet, we will most likely have already heard about it.) And thanks to all of our readers who have submitted great news tips to us. If you didn’t catch all the latest News to Know, why not take a look to see what you’ve missed?
(Please note that links will take you directly to the source. Answers in Genesis is not responsible for content on the websites to which we refer. For more information, please see our Privacy Policy.)
Footnotes
- These TLRs are in the top 1% of modern human genes showing Neanderthal influence.
- Rob Stein, “Itchy Eyes? Sneezing, Maybe Blame That Allergy on Neanderthals,” NPR, January 7, 2016, http://www.npr.org/sections/health-shots/2016/01/07/462132043/itchy-eyes-sneezing-maybe-blame-that-allergy-on-neanderthals.
- Sarah Kaplan, “Got Allergies? Blame the Neanderthals,” Washington Post, January 8, 2016, https://www.washingtonpost.com/news/morning-mix/wp/2016/01/08/got-allergies-blame-the-neanderthals/.
- Rob Stein, “Itchy Eyes? Sneezing, Maybe Blame That Allergy on Neanderthals.”
- Cell Press, “Neanderthal Genes Gave Modern Humans an Immunity Boost, Allergies,” ScienceDaily, January 7, 2016, http://www.sciencedaily.com/releases/2016/01/160107140408.htm.
- This was discovered by Kelso’s group, as described in Michael Dannemann, “Introgression of Neandertal- and Denisovan-like Haplotypes Contributes to Adaptive Variation in Human Toll-like Receptors,” The American Journal of Human Genetics 98 (January 7, 2016): 22, doi: 10.1016/j.ajhg.2015.11.015.
- Corinne Simonti et al., “The Phenotypic Legacy of Admixture Between Modern Humans and Neandertals,” Science (February 12, 2016): 351, 737–741, doi: 10.1126/science.aad2149.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis