Aquatic Animals
Deep Sea Creatures Produce Light
Light is essential for most animals’ survival—finding food, avoiding danger, and locating a mate. Deep-sea animals possess lights for all these purposes; and in fact, some creatures are equipped with several different lighting systems. These lights are beautiful reminders that God has conquered the darkness through Christ.
Coral Reef Growth Rates Faster Than Believed?
The Indian Ocean tsunami that killed hundreds of thousands of people several years ago also destroyed many of the coral reefs of Indonesia. These reefs are bouncing back with surprising speed, showing how quickly life can recover from a catastrophe.
Was a Plesiosaur Carcass Found?
If we did find a plesiosaur today, would that really be enough to convince an evolutionist? As much as some would like to believe that a modern-day plesiosaur has been found, there really is no good evidence at this time. This argument is one more that we believe Christians should not use.
The Portuguese Man-of-war
Each man-of-war is a collection of four types of animals. These animals are called zooids and they make up a colony. Each zooid has a different role in the life of the colony. But how did these distinct, yet connected animals ever learn to function together?
Aquatic Animals Topics
-
Fish
The diversity and streamlined design of fish and sharks testifies to the God of creation, not to the mindless process of evolution.
Articles About Aquatic Animals
-
March 12, 2026 from Ken Ham Blog
A new study suggests comb jellies are more complex than previously thought.
-
Oct. 1, 2025 from Answers Magazine
When invasive species clash with native ecosystems, biologists face tangled questions about biblical dominion.
-
July 24, 2025 from Ken Ham Blog
Based on a study of nanostructures in the skin of blue sharks, scientists believe that they can change color as they dive, better matching the water around them.
-
May 26, 2024 from Answers Magazine
The narwhal’s unusual tusk clearly didn’t grow by accident—but what is it there for?
-
Feb. 15, 2024 from Ken Ham Blog
A new study of blue whale DNA found that around 3.5% of a blue whale’s genome is actually the result of interbreeding with fin whales.
-
Nov. 16, 2023 from Ken Ham Blog
According to a new study, true crabs have evolved to leave their marine environment a stunning 7 to 17 times!
-
Feb. 5, 2023 from Answers Magazine
Next time you see a shell, consider how much care went into making this wonderful home for a tiny animal. If God cared so much for an animal’s temporary home, how much does he care for you?
-
Dec. 12, 2022 from Ken Ham Blog
I recently saw a headline that caught my attention titled, “Crabs aren’t the only thing evolution keeps making. An Expert Explains.” Now what is such a statement supposed to mean?
-
Dec. 11, 2022 from Answers Magazine
This miniature underwater Christmas tree shows how wonderfully God blends the beautiful with the practical.
-
Dec. 8, 2022 from Ken Ham Blog
Octopuses don’t live long, so their intelligence isn’t from learning—God just created their kind to be very smart. But do we share our smarts with them?
-
Sept. 4, 2022 from Answers Magazine
What makes orcas so successful at filling the oceans? If you think it’s their size and strength, listen up.
-
Aug. 8, 2022 from Ken Ham Blog
A news item highlighting a research paper announced the discovery of Idotea balthica that can pollinate red seaweed—that’s right, underwater pollinators!
-
May 14, 2022 from Answers Magazine
In 1955, an amateur fossil hunter found a bizarre fossil in Illinois. To this day, the exact nature of this monster remains a mystery.
-
June 20, 2021 from Answers Magazine
The Creator’s provision for the horseshoe crab is provision for us.
-
March 12, 2021 from Answers in Depth
In recent years evolutionists have increasingly promoted the evolution of whales as one of the most convincing examples of macroevolution.
-
Sept. 22, 2020 from Glass House
Evolutionists tout the whale series as one of the best evidences for evolution. However, there are lots of problems with the story of whale evolution.
-
July 19, 2020 from Answers Magazine
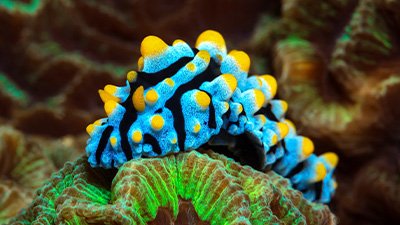
The words “slug” and “festive display of color” don’t usually come to mind at the same time. Fun facts about nudibranchs or sea slugs.
-
July 3, 2020 from Answers in Depth
Whale evolution is presented in most biology textbooks as absolute fact, often with inaccurate depictions of the supposed transitional forms.
-
March 22, 2020 from Answers Magazine
Before modern cameras and technology, the inky blackness of the deep barred its mysteries from human eyes. But now these wonders are coming to light.
-
March 1, 2020 from Answers Magazine
Even this seemingly simple animal bears the mark of divine design.
-
Nov. 1, 2019 from Answers Magazine
Horseshoe crabs are called “living fossils” because the ones you see in aquariums today look the same as their ancestors deep in the fossil record.
-
Nov. 1, 2019 from Answers Magazine
Though a mystery to evolutionists, the incredible variety of cichlid fish showcases the Creator’s brilliant design for His creatures to diversify.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis