
2.5 Mutations, Yes; Evolution, No
Darwin and Biologic Change
The answer seems to be: “Mutations, yes; evolution, no.” In the last analysis, mutations really don’t help evolutionary theory at all. There are three major problems or limits (and many minor ones) that prevent scientific extrapolation from observed mutational change to hypothetical evolutionary change.
(1) Mathematical challenges. Problem number one is the mathematical. I won’t dwell on this one, because it’s written up in many books and widely acknowledged by evolutionists themselves as a serious problem for their theory.1
Fortunately, mutations are very rare—or are they? They occur on an average of perhaps once in every ten million duplications of a DNA molecule (107, a one followed by seven zeroes). That’s fairly rare. On the other hand, it’s not that rare. Our bodies contain nearly 100 trillion cells (1014). So the odds are quite good that we have a couple of cells with a mutated form of almost any gene. A test tube can hold millions of bacteria, so, again, the odds are quite good that there will be mutant forms among them.
The mathematical problem for evolution comes when you want a series of related mutations. The odds of getting two mutations that are related to one another is the product of their separate probabilities: one in 107 x 107, or 1014. That’s a one followed by 14 zeroes, 100 trillion! Any two mutations might produce no more than a fly with a wavy edge on a bent wing. That’s a long way from producing a truly new structure, and certainly a long way from changing a fly into some new kind of organism. You need more mutations for that. So, what are the odds of getting three mutations in a row? That’s one in a billion trillion (1021). Suddenly, the ocean isn’t big enough to hold enough bacteria to make it likely for you to find a bacterium with three simultaneous or sequential related mutations.
What about trying for four related mutations? One in 1028. Suddenly, the earth isn’t big enough to hold enough organisms to make that very likely, and we’re talking about only four mutations. It would take many more than that to change a fish into a philosopher, or even a fish into a frog. It was at this level (just four related mutations) that microbiologists gave up on the idea that mutations in asexual lines could explain why some bacteria are resistant to four different antibiotics at the same time. The odds against the mutation explanation were simply too great, so they began to look for another mechanism—and they found it.
First of all, using cultures that are routinely kept for long periods of time, they found out that bacteria were resistant to antibiotics, even before commercial antibiotics were “invented.” Resistant bacteria were even found in the bodies of explorers frozen more than a century before medical antibiotic use. Genetic variability was “built right into” the bacteria. Did the nonresistant varieties get resistant by mutation? No. Resistant forms were already present. Furthermore, certain bacteria have little rings of DNA, called plasmids, that they trade around among themselves, and they passed on their resistance to antibiotics in that way. It wasn’t mutation and asexual reproduction at all, just ordinary recombination and variation within kind.
Bacteria can be made antibiotic resistant by mutation, but such forms are “evolutionary cripples.” The mutation typically damages a growth factor, so that the mutationally crippled bacteria can scarcely survive outside the lab or hospital. The antibiotic resistance carried by plasmids results from enzymes produced to break down the antibiotic. Such bacteria do not have their growth crippled by mutation. Their resistance is by design.
But why, you might well ask, would God create antibiotic resistance? It’s possible God designed antibiotic resistance in bacteria, and antibiotic production by fungi, to balance the growth of these prolific organisms in the soil. Only after the corruption of creation did some bacteria become disease causers, making antibiotic resistance “inadvertently” a medical problem.
Contrary to popular opinion, drug resistance in bacteria does not demonstrate evolution. It doesn’t even demonstrate the production of mutations that add information for new categories of protein to the bacterial genome. It does demonstrate natural selection (or a sort of artificial selection, in this case), but only selection among already existing variations within a kind. It also demonstrates that when the odds that a particular process will produce a given effect get too low, good scientists normally look for a better explanation, such as the plasmid explanation for resistance to multiple antibiotics.
At this point, evolutionists often say that “time is the hero of the plot.” That’s what I used to say to my students. “Sure, the odds are low, but there’s all that time, nearly five billion years!” Five billion years is only about 1017 seconds, and the whole universe contains fewer than 1080 atoms. So even by the wildest “guesstimates,” the universe isn’t old enough or big enough to reach odds like the 1 in 103,000,000 that Huxley, an evolutionist, estimated as the odds against the evolution of the horse. Evolutionists like Huxley do believe in miracles; they just don’t believe in the Miracle Worker.
In his chapter “Beyond the Reach of Chance,” Denton2 discusses attempts to simulate evolutionary processes on computers. He concludes with these strong words:
If complex computer programs cannot be changed by random mechanisms, then surely the same must apply to the genetic programs of living organisms. The fact that systems in every way analogous to living organisms cannot undergo evolution by pure trial and error [i.e., by mutation and selection] and that their functional distribution invariably conforms to an improbable discontinuum comes, in my opinion, very close to a formal disproof of the whole Darwinian paradigm of nature. By what strange capacity do living organisms defy the laws of chance which are apparently obeyed by all analogous complex systems? (emphasis added).
Most gratifyingly, Denton seems to look beyond the merely negative insufficiency of chance to glimpse a solution to “The Puzzle of Perfection,” as he calls it, in the “design hypothesis”:
It is the sheer universality of perfection, the fact that everywhere we look, we find an elegance and ingenuity of an absolutely transcending quality, which so mitigates against the idea of chance. . . . In practically every field of fundamental biological research ever-increasing levels of design and complexity are being revealed at an ever accelerating rate. The credibility of natural selection is weakened, therefore, not only by the perfection we have already glimpsed but by the expectation of further as yet undreampt of depths of ingenuity and complexity (p. 342).
In God’s handiwork, unlike man’s, the closer we look, the more marvelous is the perfection we see. Unfortunately, we also have evidence that the transcendent ingenuity and design Denton sees has been marred and scarred. In that sense, mathematics isn’t even the most serious challenge to using mutations as the basis for evolution.
(2) Upward or downward? Even more serious is the fact that mutations are “going the wrong way” as far as evolution is concerned. Almost every mutation we know is identified by the disease or abnormality that it causes. Creationists use mutations to explain the origin of parasites and disease, the origin of hereditary defects, and the loss of traits. In other words, time, chance, and random changes do just what we normally expect: tear things down and make matters worse. Using mutations to explain the breakdown of existing genetic order (creation-corruption) is quite the opposite of using mutations to explain the build up of genetic order (evolution). Clearly, creation-corruption is the most direct inference from the effects of mutations that scientists actually observe.
By producing defects or blocking the normal function of certain genes, mutations have introduced numerous genetic abnormalities into the human population. The hemophilia (bleeders’ disease) that afflicted the royal houses of Europe may have arisen as a mutant of a clotting-factor gene in Queen Victoria, for example; and the dreaded Tay-Sach’s Disease may have arisen in Czechoslovakia in the 1920s as a mutation in the gene for producing an enzyme crucial to brain function.
Some people like to call mutations “the means of creation,” but mutations don’t create; they corrupt! Both logically and often observationally, as in the examples above, the ordered state must come before mutations can disorder it. Mutations are real, all right, but they point to a corruption of the created order by time and chance.
As a matter of fact, human beings are now subject to over 5,000 mutational disorders. Fortunately, we don’t show as many defects as we carry. The reason they don’t show up is that we each have two sets of genes, one set of genes from our mothers and another set from our fathers. The “bad genes” we inherit from our mothers’ side are usually covered up by our fathers’ genes, and vice versa. We can see what is likely to happen when an animal is born with only one set of genes. Figure 17, based on a description in a genetics textbook, represents the rare case of a turkey that was hatched from an unfertilized egg, so it had just one set of chromosomes. The poor bird couldn’t hold its head up; instead, it bobbed up and down from a neurological disorder. The feathers were missing in patches, and it finally had to be transferred to a germ-free chamber because its resistance to disease was so low.
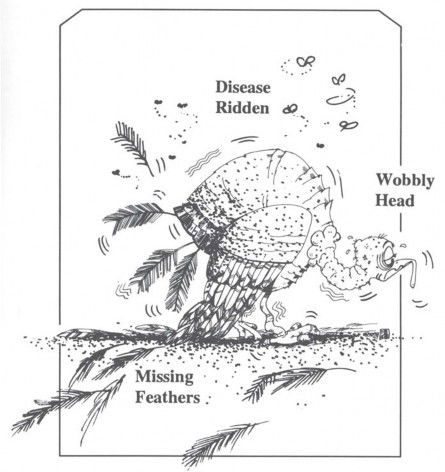
Figure 17. Mutations are mostly harmful, and, as time goes on, they impose an increasingly heavy “genetic burden” on a species. The turkey above, lacking a second set of genes to mask its hereditary defects, could scarcely survive. Creationists use mutations to help explain the origin of parasites and disease. Some evolutionists still believe that time, chance, and occasional favorable mutations provide the raw material for “upward-onward” progress, but the “post-neo-Darwinists” are looking for other means to explain evolution.
Now here’s the basis for a good horror story. Picture a mirror at the end of a dark hall. You claw your way through the spider webs to reach the mirror, and then you press a button. The mirror then splits you in two halves, so you can see what you would look like if you had only those genes you inherited from your mother’s genes or only those from your father. In the next scene, you’re writhing there in agony, your hair turning white as you fall over backward and die of fright! Unfortunately, that picture exaggerates only slightly what mutations have done to human beings and to the various kinds of plants and animals as well. If it weren’t for having two sets of genes, few of us would be able to survive.
Evolutionists recognize, of course, the problem of trying to explain “onward and upward” evolution on the basis of mutations that are harmful at least 1,000 times more often than they are helpful. No evolutionist believes that standing in front of x-ray machines would eventually improve human beings. No evolutionist argues that destruction of the earth’s ozone layer is good because it increases mutation rates and, therefore, speeds up evolution. Evolutionists know that decrease in the ozone layer will increase mutation rates, but they, like everyone else, recognize that this will lead only to increased skin cancer and to other harmful changes. Perhaps a helpful change might occur, but it would be drowned in the sea of harmful changes.
Because harmful mutations so greatly outnumber any supposed helpful ones, it’s considered unwise nowadays (and illegal in many states) to marry someone too closely related to you. Why? Because you greatly increase the odds that bad genes will show up. By the way, you also increase the odds of bringing out really excellent trait combinations. But did you ever hear anybody say, “Don’t marry your first cousin or you’ll have a genius for a child?” They don’t usually say that, because the odds of something bad happening are far, far, far, far, far greater.
That would not have been a problem, by the way, shortly after creation (no problem for Cain and his wife, for example). Until mutations had a chance to accumulate in the human population, no such risk of bad combinations existed. Mutations are often carried as “hidden genes”(recessives) that are difficult to eliminate by selection, so they tend to build up in populations. The buildup of mutations with time poses a serious problem for plants and animals, as well as for human beings, and time, evolution’s “hero,” only worsens the problem of mutational decay.
Geneticists, even evolutionary geneticists, refer to the problem as “genetic load” or “genetic burden,” terms meant to imply a burden that “weighs down” a species and lowers its genetic quality. In his article “The Mechanisms of Evolution” in the Scientific American book Evolution, Francisco Ayala3 defines a mutation as “an error” in DNA. Then he explains that inbreeding has revealed that mutations in fruit flies have produced “extremely short wings, deformed bristles, blindness, and other serious defects.” Does that sound like “the raw material for evolution?”
It’s not that beneficial mutations are theoretically impossible. Bacteria that lose the ability to digest certain sugars, for example, can regain that ability by mutation. That’s no help to evolution, however, since the bacterium only gets back to where it started, but at least the mutation is helpful.
A classic example used for decades to illustrate a beneficial mutation is sickle-cell anemia. Sickle-cell anemia is a disease of red blood cells. Why would anyone call that a beneficial mutation? Well, in certain parts of Africa, the death rate from malaria is quite high. Malaria is caused by a tiny, one-celled organism that gets inside the red blood cells and eats up the hemoglobin. Now, that particular germ doesn’t like sickle-cell hemoglobin. Carriers of one sickle-cell gene produce about half normal and half sickle-cell hemoglobin, and the malaria germ leaves them alone, too. So, carriers don’t get malaria. But the cost is high: 25 percent of the children of carriers can die of sickle-cell anemia, and another 25 percent are subject to malaria. If you want to call that a good mutation, you’re welcome to it! It seems doubtful to me that real improvement of human beings would result from accumulating that kind of “beneficial” mutation, and certainly hemoglobin’s ability to carry oxygen was not improved.
The gene for sickle-cell anemia has built up to high levels in certain African populations, not because it is “beneficial” in some abstract sense, but simply because the death rate from anemia in those areas is less than the death rate from malaria. Natural selection is a “blind” process that automatically accumulates genes for short-term survival, even if it reduces the long-term survival of the species. For that reason, evolutionists recognize that natural selection can occasionally lead to “mischievous results” detrimental to genetic quality. That’s the effect I think we’re seeing with sickle-cell anemia (Figure 18).
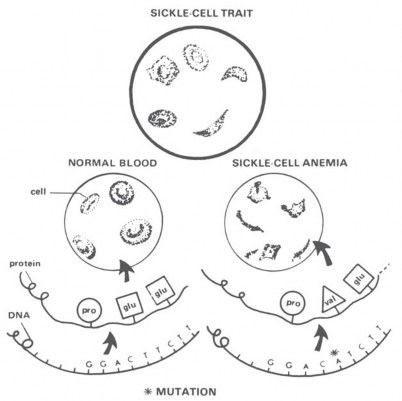
Figure 18. “Sickle-cell anemia” is often given as an example of a favorable mutation, because people carrying sickle-cell hemoglobin in their red blood cells(Ss) are resistant to malaria. But the price for this protection is high: 25 percent of the children of carriers may die of the anemia (ss), and another 25 percent (SS) are subject to malaria. The gene will automatically be selected where the death rate from malaria is high, but evolutionists themselves admit that short-term advantages—all that natural selection can ever favor—can produce “mischievous results” detrimental to long-term survival. What do you think? Is sickle-cell anemia a “mischievous result,” or a good example of evolutionary progress? (Drawing from Parker, Reynolds, and Reynolds, Heredity, 2nd Edition [Chicago, IL: Educational Methods, Inc., 1977]).
Furthermore, when the frequency of the sickle-cell gene reaches 18 percent, natural selection for it “stops.” That’s the point at which the death rates from sickle-cell anemia and malaria balance, demonstrating conclusively that sickle-cell anemia is not a suitable model for the continuous genetic expansion that evolutionists seek.
Suppose I told you I had found a way to make cars run uphill without using gasoline. Then, as you watched in eager anticipation, I showed you how applying the brakes would make the car run downhill more slowly. Would you believe I had discovered a means for getting cars to run uphill without fuel? Similarly, natural selection can and does slow the rate of genetic decay produced by accumulating mutations (as it does with sickle-cell hemoglobin), but that hardly proves that mutation-selection produces upward and onward progress!
A better example of favorable mutation might be found in a change from teosinte into corn, but the mutation was favorable to people, not to corn, which has been described as a “biological monstrosity” that could not survive on its own without man’s special care. There are many other examples of mutations “beneficial” to people: seedless grapes, short-legged sheep, hairless dogs, but these would all be harmful to the organism in its own environment and, hence, harmful in evolutionary perspective.
While taking a graduate course in evolution on his way to a master-of-science degree in biology, one of my graduates asked his professor a simple question during a lecture on mutations as the raw material for evolution: “Would you please give us some examples of beneficial mutations?” After an uncomfortably long pause, the professor finally replied, “I can’t think of any right now, but there must be hundreds of them.” He did not come back to the next class with a list—but, to his credit, he didn’t try to use sickle-cell anemia to illustrate helpful mutations.
Once again, let me say that it’s not that good mutations are theoretically impossible; rather, the price is too high. To explain evolution by the gradual selection of beneficial mutations, one must also put up with the millions of harmful mutations that would have to occur along the way. Even though he has been one of the “old guard” defenders of classic neo-Darwinian evolution, Ayala faces the problem squarely in his article in the Scientific American book Evolution. He is talking about variation within species (not kind, but species, the smallest possible unit). He says that variation within species is much greater than Darwin postulated. He speaks of such variation as “enormous” and “staggering.” Yet when he gets to the actual figures, the variation is less than I, as a creationist, would have expected. (Ayala did say his figures underestimated the real variation.)
For creationists, all this variation poses no problem at all. If living things were created to multiply and fill the earth, then great variation within kind is simply good design. There would be no price to pay for created variability, since it would result from creation, not from time, chance, struggle, and death (natural selection). (Mutations have introduced further variability since creation was corrupted, but it’s the kind of variability a bull introduces into a china shop!)
What problem did Ayala, as an evolutionist, see with all this staggering variability? Just this: For each beneficial mutation a species accumulated, the price would be a thousand or more harmful mutations. When genetic burden gets too great, offspring are so likely to have serious hereditary defects that the ability of the species to survive is threatened. Take the Florida panther, for example, which is considered an endangered species. What endangers it? Highway traffic? No. Hunting? No. Habitat destruction? No. It’s endangered by too much “evolution,” i.e., the accumulation of mutations that have riddled the reproductive and circulatory systems with so much “evolutionary progress” (read that “genetic burden”) that the small, inbred population of panthers was unable to produce a cub that could survive and reproduce.
I told my students that Florida officials should import panthers from out West to cross with the Florida panther to dilute the effects of these harmful mutations. To my surprise, that’s what happened! The Florida panther is now making a comeback—except that as it becomes a healthier panther, it is less a “Florida” panther (which was never a true species anyway). Unfortunately, there are other cases where “endangered” sickly subspecies with multiple mutational defects are “protected” from good health by preventing their interbreeding with others of their kind.
Time only makes this evolutionary problem worse. Thanks to our accumulated genetic burden, serious hereditary defects are present in perhaps 5 percent of all human births, and that percentage greatly increases among the children of closely related parents. All of us have some genetic shortcomings, and it’s really only by common consent that most of us agree to call each other “normal.”
Natural selection cannot save us from this awful situation either. Selection can and does eliminate or reduce the worst mutations—but only when these mutants come to visible (phenotypic) expression. Most mutations “hide” as recessives, “invisible” to selection, and continue to build up in secret at multiple loci, somewhat like a “genetic cancer” slowly but steadily eating away at genetic quality.
If early evolutionists had known what we know now about mutations, it’s most unlikely that mutations would ever have been proposed as the pathway to evolutionary progress.
(3) Mutations point back to creation. Mathematics and genetic load are huge problems for evolution, but the biggest reason mutations cannot lead to evolution is an extremely simple one. It’s so simple, I’m almost afraid to say it. But really, mutations presuppose creation. After all, mutations are only changes in genes that already exist.
Most mutations are caused by radiation or replication errors. What do you have to have before you can have a mutation? Obviously, the gene has to be there first, before the radiation can hit it or before it can make a copying mistake. In one sense, it’s as simple as that: the gene has to be there before it can mutate. All you get as a result of mutation is just a varied form of an already-existing gene, i.e., variation within kind (Figure 19).

Figure 19. The most logical inference from our scientific observations of mutation, selection, and genetic recombination would seem to be variation within created kinds. There’s no “genetic burden” to bear if variety is produced by creation instead of time, chance, and mutation. But could there be enough variation in each created kind to produce all the diversity we see today? Creationists now have some promising answers to that question. (Drawing from Bliss, Origins: Two Models, 2nd edition [Green Forest, AR: Master Books, 1978]).
Uncritical acceptance of evolution has so stunted scientific thinking that people give mutations god-like qualities. They act as if a cosmic ray striking a cell can cause a “mutation” that somehow assembles over 1,500 DNA bases into a brand new gene, regulators and all, that suddenly begins producing a brand new protein responsible for a brand new trait, raising the lucky mutated organism to the next higher rung on the evolutionary ladder! NOTHING remotely like that has ever, or could ever, happen!
Mutations are NOT genetic “script writers”; they are merely “typographic errors” in a genetic script that has already been written. Typically, a mutation changes only one letter in a genetic sentence averaging 1,500 letters long.
To make evolution happen—or even to make evolution a theory fit for scientific discussion—evolutionists desperately need some kind of “genetic script writer” to increase the quantity and quality of genetic INFORMATION. Mutations have no ability to compose genetic sentences, no ability to produce genetic information, and, hence, no ability to make evolution happen at all.
That simple, absolutely foundational fact completely stumped Richard Dawkins, the world’s leading spokesman for evolution as of this writing. Ina video production featuring several evolutionist and creationist leaders and skeptics,4 Dawkins argued eloquently that millions of years of mutation and natural selection would serve as a “blind watchmaker,”5 producing all appearance of design among living things without any help from some supernatural Designer. Then in a quiet, non-threatening voice, not knowing what the answer would be, the narrator asked Dawkins to give an example of a mutation that adds information.
The usually effusive Dawkins gestured, opened his mouth, but stopped before he spoke. With his eyes shifting back and forth as if searching for some answer, he started to speak several times, but always checked himself. Finally, after a long embarrassing silence, the program resumed with Dawkins speaking on a different subject—leaving unanswered the ultimate question, the origin of genetic information.
Yet, molecules-to-man evolution is all about phenomenal expansion of genetic information. It would take thousands of information-adding mutations to change “simple cells” into invertebrates, vertebrates, and mankind. If there were any scientific merit at all to mutation-selection as a mechanism for evolution, Dawkins’ reply should have been enthusiastic and overwhelming, “My three favorite examples of mutations adding information are. . . . Excellent examples among plants are . . . among insects are . . . among bacteria are . . . .” His answer, instead, was silence, and with no mechanism to add genetic information, the “evolutionary tree” can’t grow.
The problem with evolution is not some shortcoming in Dawkins, however. The problem is with the fundamental nature of information itself. The information in a book, for example, cannot be reduced to, nor derived from, the properties of the ink and paper used to write it. Similarly, the information in the genetic code cannot be reduced to, nor derived from, the properties of matter nor the mistakes of mutations; its message and meaning originated instead in the mind of its Maker.
As cogently presented by two of the world’s leading information theorists,6 information comes only from pre-existing information.7 Information systems have the “exherent,” created kind of design, which can be logically inferred from our scientific observations as explained earlier (Figure 1). Although mutations may corrupt it and selection may sort variations into different environments, it was not a “blind watchmaker” that composed the genetic script for each kind of organism, but a Creator with a plan and purpose and eyes wide open.
If the evidence and logic is so convincingly clear, why is evolution still so popular? Laying aside personal biases and spiritual concerns, there is a serious semantic problem that could cause honest confusion. After all, evolution is about the continual production of new and different genes, and mutations are continually producing new and different genes—or are they?
Genes of the same kind, like those for straight and curly hair or those for yellow and green seeds, are called alleles. There are over 300 alleles of the hemoglobin gene. That’s a lot of variation, but all those alleles produce hemoglobin, a protein for carrying oxygen in red blood cells (none better than the normal allele). By concept and definition, alleles are just variants of a given gene, producing variation in a given trait. Mutations produce only alleles, which means they can produce only variation within kind (creation), not change from one kind to others (evolution).
Genes of the same kind can be defined objectively as segments of DNA that occupy corresponding positions (loci; sing. locus) on homologous chromosomes. Homologous chromosomes are pairs that look alike, but come from two different parents, so their genetic content is similar but not identical. They pair up and then separate in the kind of cell division (meiosis) required for sexual reproduction. Genes that pair up in meiotic cell division, therefore, can be identified as genes of the same kind. Genes of the same kind are also turned on and off by the same gene regulators. Notice, it is not subjective human opinion that is telling us which genes are the same kind; it is objective, observable cellular processes.
Mutations, random changes in the genetic code, do produce “new genes” not present at creation, but the so-called “new genes” are still found at the same locus, still pair the same way in meiosis, and are still turned on and off by the same regulators, so they are really only genes of the same kind as the original, and represent only variation within kind (usually harmful variation in the case of mutations).
Notice the terms “new genes” or “different genes” can have two radically different meanings. As geneticists normally do, we have been calling genes of the same kind alleles. The genes for tongue rolling and non-rolling are “different genes” in one sense, but only variations of the same kind of gene—affecting the same trait, found in corresponding positions (loci) on homologous chromosomes, pairing up in meiosis, and turned on and off by the same regulators. They are NOT different genes in the sense that genes for tongue rolling, and genes for making sickle cell hemoglobin are! Similarly, the sickle cell gene is a “new gene” in the sense that it was not present at creation, but it is only a new (and harmful!) version of a pre-existing gene, one that occupies the same chromosomal position, pairs the same way, and is turned on and off by the same regulators as the gene for making normal hemoglobin. In fact, the gene for sickle cell hemoglobin differs in base sequence at only one position out of several hundred in the normal gene for making hemoglobin, again just variation within kind or allelic variation.
We need a new and different term to describe genes that are truly new and different—genes with information affecting a different category of trait—not just information on varieties of shirts, for example, but information on motorcycles! To refer to genes that do NOT occupy corresponding loci on homologs, that do NOT pair in meiosis, and that DO contain information on distinctive categories of traits, we will use the word genon. Genes for tongue rolling and non-rolling are different alleles, for example, but genes for tongue rolling and genes for making hemoglobin are different genons, with genes for normal and sickle cell hemoglobin as alleles of the hemoglobin genon.
The complete set of DNA specifying a kind is called its genome. The human genome includes at least 30,000 different genons, each of which could have been created in four different allelic varieties (two in each parent). Genetic defects and diseases occurring since the corruption of creation have introduced many new alleles, but no new genons. All the genes in one generation available to be passed on to the next are called the gene pool. Members of the same kind may also be defined as organisms that share the same gene pool.
The number of genes for different kinds of traits (genons) can be called the depth of the gene pool. Using earlier examples, we could say the human gene pool is 30,000 genons deep, the E. coli bacterial gene pool about 5,000, while the gene pool of a small virus may be only a dozen genons deep. The width of the gene pool refers to the amount of its “horizontal” allelic variation. Among dogs, for example, the width of a greyhound’s gene pool is very narrow; crossing pure bred greyhounds just gives you more greyhounds, all very similar in speed, color, intelligence, hair length, nose length, etc. Crossing two “mongrels,” however, can give you big dogs and small dogs, dark and light and splotchy colored dogs, dogs with long and short hair, yappy and quiet dogs, mean and affectionate dogs, etc., etc.! The width of the mongrel’s gene pool (its allelic variability) is quite large compared to the greyhound’s, but the depth of the gene pool (the number of genons) is the same for both dogs.
“Kind” is defined in terms of depth of the gene pool, the total number of different genons in a genome and a list of the traits they influence. Variation within kind is defined in terms of the width of the gene pool, the number of alleles at each gene site (locus or genon).
Creation: Facts of Life
Dr. Parker, a leading creation scientist and former AiG speaker, presents the classic arguments for evolution used in public schools, universities, and the media, and refutes them in an entertaining and easy-to-read style. A must for students and teachers alike! This is a great book to give to a non-Christian as a witnessing tool.
Read OnlineFootnotes
- Paul S. Moorehead and Martin M. Kaplan, Mathematical Challenges to the Neo-Darwinian Interpretation of Evolution, Wistar Symposium No. 5 (Philadelphia, PA: Wistar Institute Press, 1967).
- Denton, Evolution: A Theory in Crisis.
- Francisco Ayala, “The Mechanisms of Evolution,” Scientific American (and Scientific American book Evolution) (September 1978).
- Gillian Brown, From a Frog to a Prince, video, Keziah Productions.
- Dawkins, The Blind Watchmaker.
- Werner Gitt, In the Beginning Was Information (Green Forest, AR: Master Books, 2006).
- Lee Spetner, Not By Chance (New York: Judaica Press, 1997).

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis

