Alexander Fleming & God’s Signature in Microbiology
An apologetic argument for declarative design in microbiology from the discoverer of penicillin, Alexander Fleming
Abstract
In logic and reasoning, a signature indicates the presence of an author; likewise, the characteristics of staphylococci indicate the presence of a Creator. Staphylococci and its “kind” are common bacteria, particularly in colonized people.1 Staphylococcus aureus has a complex molecular mechanism of assembling its golden pigment, staphyloxanthin. The biosynthesis of staphyloxanthin is a stellar example of irreducible complexity. Similar to staphylococci, the life and works of Alexander Fleming show the fingerprints of Providence. The so-called “serendipitous” achievements of Fleming have contributed to modern medicine, convincing Fleming and others that God was at work in his life. Fleming recognized that his life’s discoveries and the “weaving” of events were more than chance; it was the invisible hand of God on his life and works. The molecular complexities of staphylococci mechanisms and the achievements of Fleming indicate the signature of a divine Designer who has placed his signature on his art piece, staphylococci.
Keywords: Alexander Fleming, bacteria, cell, DNA, signature, Providence, Staphylococcus aureus, Staphylococcus epidermidis, Staphyloxanthin, Irreducible Complexity, Penicillin, lysozyme, intelligent design
Signature Style
People recognize the work of others by the autograph of the author or maker. Every person has his or her own particular writing style. A well-known signature is that of John Hancock on the US Declaration of Independence. Another famous person who had both a distinctive autograph and a style of writing was Alexander Fleming (Figures 1 and 2), who wrote names of bacteria on the agar of petri dishes. Anyone who is familiar with the history of medical microbiology is familiar with Fleming, the bacteriologist who discovered penicillin. Alexander Fleming also made “Germ Art,” pictures that he created using pigmented bacteria in petri plates, and he also “wrote” bacteria names with the same pigmented bacteria (Maurois, 1959). Thus, he left his signature on his art plates. In this article, we describe a familiar bacteria kind, staphylococci that we study weekly at Liberty University. We believe it has some signatures for recognition and it gives us evidence of an intelligent and artistic designer, who is the Creator and Author of microbes and men. There is a silent, declarative signature in staphylococci and its history (Psalm 19: 1; Romans 1:20).
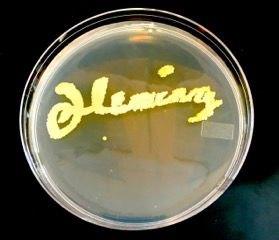
Figure 1. Signature of Fleming with pigmented bacteria on Trypticase soy agar. Image courtesy of Alan Gillen.
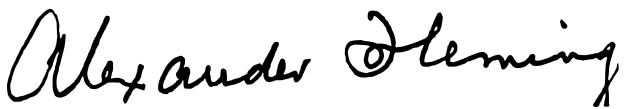
Figure 2. Signature of Fleming with pen. Reproduced from Wikimedia Commons.
Signature in the Cell
Dr. Stephen C. Meyer discusses in his book Signature in the Cell (2010) that DNA provides evidence for intelligent design, emphasizing information theory: that DNA contains the information necessary to build and run living organisms. While Meyer emphasizes eukaryotic cells, we submit the same is true for prokaryotic cells, like Staphylococcus. DNA provides evidence for specified complexity, a language, sequence, arrangement, and order of cell division. In the case of staph, the DNA codes for a sphere (coccus) in a grape-like arrangement.
Signature on the Slide
The “signature” on a microscope slide is grapelike, Gram-positive (purple) cocci (plural for coccus). The word coccus comes from a Greek word meaning "berry," (or kókkos, "granule") and all the cocci have a spherical form like that of a tiny berry. Many are not perfectly round, but are flattened on one side or are more or less elongated. Staphylococcus aureus range from 0.8–1.0 µ in diameter. Staphylococcus (Greek, staphylē, “a bunch of grapes") can divide along multiple axes, and thus forms clusters. Their descendants always form clusters from 8 to 64 spheres due to enzymes cutting the planes precisely to form “grapes.”
The Staphylococcus “kind” is a rather hardy Gram-positive bacteria type and is especially resistant to drying. These bacteria grow in both dry and moist places of the skin. They can be found on the surface, deep, or intermediate on the skin, and usually occur in large grape-like clusters (32–64 cells); however, these bacteria can be observed individually or in two-, four-, eight-, or 16-cell or larger chains at a time (Figures 3 and 4).
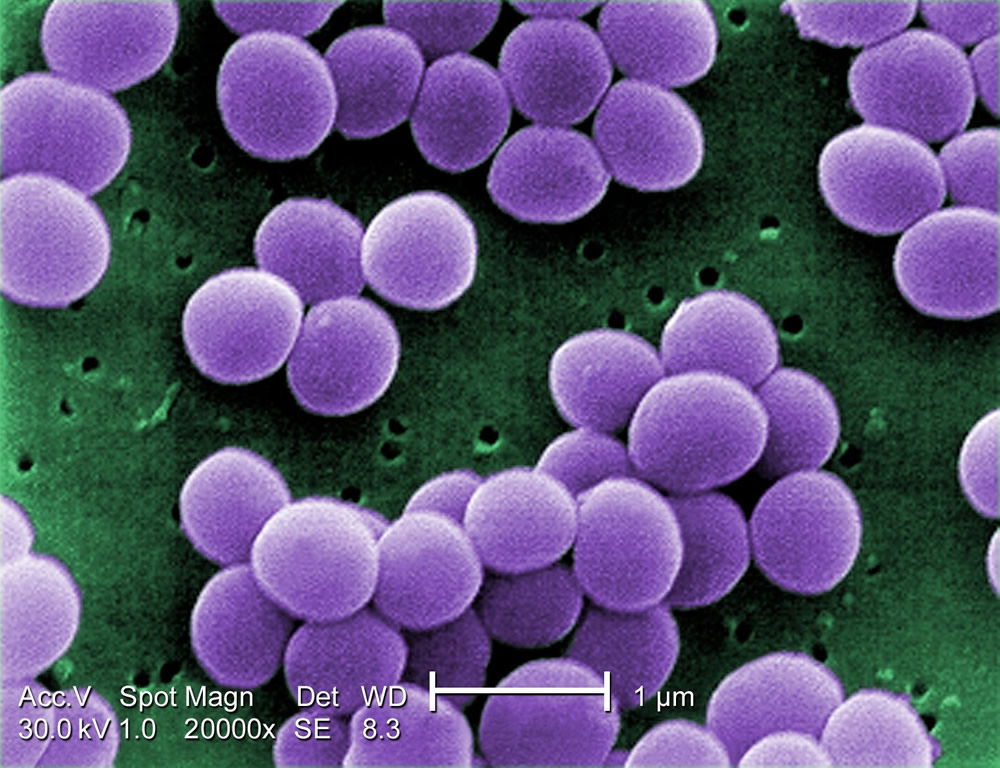
Figure 3. MRSA photo showing the purple color of Staphylococcus aureus. Reproduced from Wikimedia Commons.
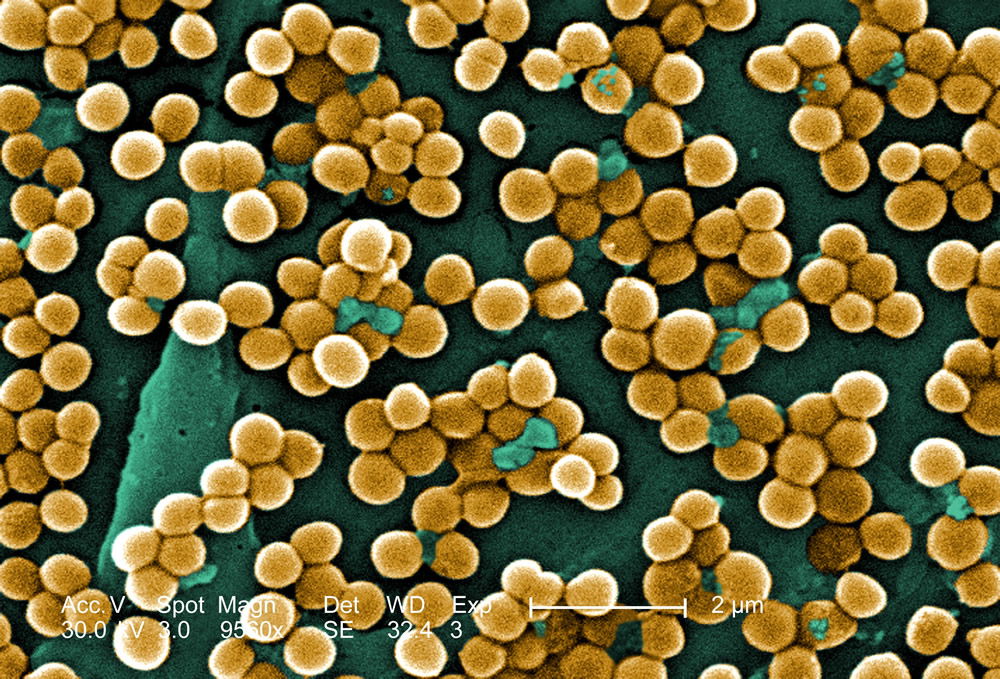
Figure 4. MRSA photo showing the golden color of Staphylococcus aureus. Reproduced from CDC. See Gillen, 2009.
Abundant, Diverse, and Adaptive in Design
At this time, over 40 species of Staphylococcus have been discovered. Many of these are found in and on the human body. While Staphylococcus is resilient and can grow at a variety of temperatures and chemical conditions, it functions very well under the conditions and temperature of the human body (Gillen, 2009, 2015). S. aureus and S. epidermidis are found on the outer layers of the skin, in hair follicles, and on the nasal membranes. S. aureus are abundant in the nose, armpits (axilla), and moist areas of the body. S. epidermidis are abundant on the surface of dry skin, and sometimes are called the “skin flake” bacteria. Other types of staphylococci are also found on the conjunctiva of the eyes, the mucus membranes that line the upper respiratory tract, and the linings of the gastrointestinal and urogenital tracts. These types of staphylococci are normally beneficial to humans, but can, at times, be harmful (Caryl, 2012).
One way that Staphylococcus is beneficial to the human body is that it produces bacteriocins. Bacteriocins are chemicals produced by one kind of bacteria that kill or stem the growth of a closely related type of bacteria. The varieties of Staphylococcus that are not harmful to humans when they exist solely on the skin produce bacteriocins that inhibit the existence of harmful bacteria on our skin.2 This clearly shows the hand of our loving Creator in preparing us to live in a fallen, cursed world. The Designer chose to make bacteria that keep us safe from other harmful types, and He designed it to flourish at body conditions. This symbiotic system points to some form of intelligence behind it.3
Some positive values of Staphylococcus include the ability to interact and engage the immune system, the ability to produce bacteriocins (i.e., natural antibiotics) (Tortora, Funke, and Case, 2015), and the ability to prevent autoimmune diseases, like multiple sclerosis. In addition, it may prevent a parasitic disease known as leishmaniasis. Cutaneous leishmaniasis, sometimes called “white leprosy,” is prevented by Staphylococcus microbiota interfacing with the immune cells in the skin. These staphylococci stimulate and “train” the immune system to effectively defend against Leishmania parasites (Gillen, 2014).
The “Golden” Rule: A Staphyloxanthin Signature Colors the Colonies on Agar
The golden appearance provides the basis for the bacterium's species name: aureus means "golden." Staphyloxanthin is a pigment name that is produced by most strains of S. aureus. Staphyloxanthin is an orange-red triterpenoid carotenoid whose biosynthesis and structure have been described. Its production is an irreducibly complex process, like prodigiosin in Serratia marcescens. Similar to prodigiosin, staphyloxanthin seems to play a role in the fitness (protection from sunlight) of Staphylococcus aureus and its ability to cope with oxidative stress.
S. aureus’ most salient characteristic is its bright golden pigment. It is one of the few bacteria that produce bright pigments. Its structure is intricate, and its synthesis is ordered, sequenced, and complex.
The biosynthesis of three distinct, separate pigments and 17 intermediary products (Marshall and Wilmouth, 1981) forming them as triterpenoid carotenoid staph golden pigment is colorfully crafted (Figure 5). It requires complicated genetics of DNA and regulator genes, and is an information-rich system. We would submit that chemical evolution, chance, and chemical necessity have not been proven capable of producing information-rich systems, and that this intelligent, artistic, and declarative system is one of design; therefore, the best explanation for the emergence of the golden pigment.
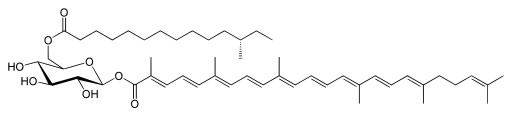
Figure 5. The structure of staphyloxanthin, the gold pigment of Staphylococcus aureus.
The synthesis of staphyloxanthin is an irreducibly complex process. An irreducibly complex system is one that requires several interlacing parts to be present at the same time, where the removal of one or more parts causes the whole system to malfunction. Destroy one part and the whole system falls apart. In evolution, a new trait would have to be completely developed—no halfway measures would do. Given this requirement, new features are so complex that Darwinian gradualism is very improbable because an incompletely developed trait would offer no selective advantage.
Signature in the Tears: Lysozymes, Precursor to Penicillin
Alexander Fleming is known for many things, but his first major discovery was on lysozyme and titled “On a Remarkable Bacteriolytic Element Found in Tissues and Secretions” in 1921 (Maurois, 1959). The lysozyme was first noticed during some investigations while he was suffering from a common cold. Nasal secretion was cultivated daily on blood-agar plates, from which Fleming grew Micrococcus luteus colonies. He described how drops of diluted nasal mucus placed on cultures of this organism inhibited their growth and cleared (i.e., lysed) its thick suspensions. This was his first so-called serendipitous “accident” and observation. The enzyme lysozyme is a chemical inhibitor of nonspecific nature. Lysozyme is found in human tears (Figure 6) and saliva. It disrupts the cell walls of Gram-positive bacteria by digesting peptidoglycan. Fleming’s most significant observation about lysozymes was some bacteriophage-type effects also on Staphylococcus from his nose on blood-agar plates. In other experiments, both staph and Micrococcus cultures in test tubes had a drop of the diluted mucus added, and within five minutes, they became clear. He successfully repeated these experiment many times.
Figure 6. Lysozyme comes from tears. Reproduced from Wikimedia Commons.
These preliminary experiments clearly demonstrated the powerful inhibitory and lytic action of nasal mucus upon coccus bacteria. He successfully repeated this many times. Later, lysozyme was found in human tears, saliva, and other fluids.
Signature of Staph Erased: The Birth of Penicillin
By 1928, Fleming became an expert on lysozyme, Staphylococcus, and war wounds. In the summer of 1928, he left a culture plate smeared with Staphylococcus bacteria on his lab bench while he went on a two-week holiday. When he returned, he noticed a clear halo surrounding the yellow-green growth of a mold that had contaminated the plate. Unknown to him, a spore of a rare variant called Penicillium notatum had drifted in from a mycology lab one floor below. Providence would have it that Fleming had decided not to store his culture in a warm incubator, and that London was then hit by a cold spell, giving the mold a chance to grow. Later, as the temperature rose, the Staphylococcus aureus bacteria grew like a lawn on blood agar, covering the entire plate—except for the area surrounding the moldy contaminant. Seeing that clear zone halo was Fleming's "discovery" moment, an instant of great insight, reasoning, and potential for penicillin (Figure 7).

Figure 7. Alexander Fleming and penicillin. Reproduced from Wikimedia Commons.
Many attribute Fleming’s stack of petri dishes left out over his two-week vacation as a sign of his untidiness. But Alexander Fleming was an expert on Staphylococcus aureus. He knew that if the cultures were started at normal incubation temperature (~37° C) and then left out at room temperature for a few days, the color intensity of the yellow-gold pigment would intensify. The different strains had slightly different shades of yellow/gold/orange and these color variants had clinical significance. The virulence of various strains correlates with pigment intensity and colony morphology. So his leaving the cultures out while he was gone on vacation may have been by design. He knew that some “signature” pathogenic S. aureus mature colonies were the smooth, opaque, little golden dome ones that required “extra” room-temperature incubation (Macfarlane, 1984, 118–119).
He proceeded to isolate the fungus that turned out to be a rare strain of Penicillium to produce the drug. He prepared the cultures of the mold, a filtrate, and found them capable of inhibiting the growth of other bacteria and published these results in 1929. Fleming said,
While working with Staphylococcus variants a number of culture-plates were set aside on the laboratory bench and examined from time to time. In the examinations of these plates were necessarily exposed to the air and they became contaminated with various microorganisms. It was notice that around a large colony of contaminating mould the Staphylococcus colonies became transparent and were obviously undergoing lysis. (Fleming, 1946)
It should be pointed out that the origin of the mold and “just right” conditions was improbable in human terms. Although mold contamination is common in the environment and in bacteriology labs, the contamination of just the right Penicillium species that produces effective penicillin is indeed rare. The chance that it contaminated the S. aureus plate at just the right time and had someone to observe it is less than one in a million (Hare, 1970).
Fleming saw his discoveries as Providence and signs of goodness from an unseen hand. It was His signature making the difference. In respect to his discovery of penicillin, Sir Alexander Fleming said, “Discoveries of this magnitude are rare . . . God took care to hide that country till he judged his people ready; then, he chose me for his whisper and I found it and its yours” (Ludovici, 1952, 196).

Figure 8. R. J. Beck Microscope (c. 1905) very similar to the one Alexander Fleming used in his lab at St. Mary’s when he discovered penicillin. Courtesy of Alan Gillen from his personal microscope collection.
Fleming saw his discoveries of both lysozymes and penicillin as the result of “The Winds of Heaven” (Ludovici, 1952, 130–131). Ludovici (1952) describes that he was “ready to ‘stretch forth his hand and try to grasp the hem when he heard the garment of God rustling through events.’” In 1945, after WWII and the Nobel Prize, the president of a penicillin pharmaceutical company (i.e., Pfizer) expressed gratitude “to the one chosen by Providence to discover and reveal the properties of the most potent weapon known to man against disease” at that time in history. On this occasion in New York, he quoted from the Gospel of St. John (John 5) that an angel stirred up the waters at Bethsaida to heal the sick man. In like manner, he said, “It was an angel who first moved the spirit of Sir Alexander Fleming when he saw, for the first time, the effect produced on a bacterial culture by a wandering mould, for the pool thus troubled has cured not one sick man but myriads of sick men” (Maurois, 1959, 201–202).
Providence, Penicillin, and the Pioneering Work of Fleming
The work of Alexander Fleming and the birth of penicillin are commonly known. The discovery is most often cited as discovered by chance or reported as serendipity. However, the authors report that this was not a chance event. We, along with Fleming, believe all these things were acts of Providence, the invisible hand of God, and our Creator directed the discovery of antibiotics. In his early life, Fleming had no particular longing for medicine or microbiology. He nearly became a surgeon, but he wound up going to St. Mary’s Hospital, where Almroth Wright influenced him so much that he became a bacteriologist and developed an interest in antiseptics, wound sepsis, and the body’s immune system. His discovery of lysozyme, his observation of the growth of a “random deposited mold,” and the fact that this random Penicillium produced an antibiotic harmless to man but lytic to bacteria all seemed to be “chance” events at first to some. But, from these apparently meaningless happenings and the order of their occurrence, there emerges unmistakable evidence of a design in which we can see the finger of God if we only look. Like many other explorers, Fleming saw just a glimpse of the possibilities and could not have imagined its greatness. But by the time of WWII, there became universal recognition of its greatness, and he accepted the honors with his characteristic humility.
His “authorized” biographer Andre Maurois (1959) said,
Looking back on his career, we find woven into the web of life a number of apparently irrelevant chance events without one of which probably not have reached its climax. There are so many of these events, and they were all so purposive that we feel driven to deny their being due to mere chance…His choice of profession, his selection of medical school, his deviation into bacteriology, his meeting with Almroth Wright, the nature of work he did with him, the chance drop of a tear, the chance fall of a mould, all these events were not surely due to mere chance. We can almost see the fingerprint of God pointed to the direction his career should take at every turn. (Maurois, 1959, 275)
Two verses that seem to sum up Fleming’s life are Proverbs 16:9 and Psalm 37:24:
The mind of man plans his way, but the Lord directs his steps.
The steps of a good man are ordered by the Lord.
God’s Signature in Science and Scripture
There is a pattern in Scripture and science beyond chance. Also, what God creates He also sustains (Hebrews 1:2). The authenticity of an autograph is recognizable to a handwriting expert. A very detailed look at the various characteristics involved determine the legitimacy of a signed item. The signature of Staphylococcus is a great example that declares the authorship of God in cells, colonies, and history. The autograph of the Invisible Hand is apparent to those who observe. Human signatures are distinct and identify authors by their pattern, style, and detail, but suspected Divine signatures must also align with His attributes given to us in His Word. We see His fingerprints and invisible hand from the Word, in creation, and in history. Creation declares the glory of God, His craftsmanship, and artistry (Psalm 19).
References
- Caryl, J. 2012. “Your Microbiome and You (Part II): Skin.” Mental Indigestion. http://mentalindigestion.net/2010/01/15/your-microbiome-and-you-part-ii-skin.
- Fleming, Sir Alexander. 1946. “Penicillin: Its Practical Application.” London: Butterworth & Co.
- Gillen, A. L. 2009. “The Genesis of Methicillin-Resistant Staphylococcus aureus.” Answers in Genesis. https://answersingenesis.org/natural-selection/antibiotic-resistance/the-genesis-of-methicillin-resistant-staphylococcus-aureus/.
- ———. 2014. “The Wonderfully Made Design of the Skin and Its Microbiome.” Answers in Genesis. https://answersingenesis.org/human-body/wonderfully-made-design-skin-and-its-microbiome/.
- ———. 2015. The Genesis of Germs: Disease and the Coming Plagues in a Fallen World. Green Forest, Arkansas: Master Books.
- Gillen, A. L., and R. Walters. 2015. “Super Staph in the Community: Is It Evolving?” Answers in Genesis. https://answersingenesis.org/biology/disease/staph-is-it-evolving/.
- Hare, R. 1970. The Birth of Penicillin and the Disarming of Microbes. London: Allen & Unwin.
- Ludovici, L. J. 1952. Fleming: Discoverer of Penicillin. London: Andrew Dakers Limited.
- Marshall, J. H., and G. J. Wilmouth. 1981. “Pigments of Staphylococcus aureus, a Series of Triterpenoid Carotenoids.” Journal of Bacteriology, 147 (3): 900–913.
- Maurois, A. 1959. Life of Sir Alexander Fleming: Discoverer of Penicillin. London: E.P. Dutton & Co., Inc.
- Macfarlane, G. 1984. Alexander Fleming: The Man and the Myth. Cambridge, MA: Harvard University Press.
- Meyer, S. C. 2010. Signature in the Cell: DNA and the Evidence for Intelligent Design. New York: HarperCollins.
- Tortora, G. J., B. R. Funke, and C. L. Case. 2015. Microbiology: An Introduction. 12th ed. San Francisco, CA: Pearson Benjamin/Cummings Pub. Co.
Footnotes
- Man is “covered” inside and outside his body with millions of microbiotic organisms (i.e., normal flora) to maintain normal bodily functions and sustain life in changing environments. The human skin contains about 3 million bacteria per cm3 (many more than the number of our own larger epidermal cells). Many Staphylococcus and Micrococcus species live on human skin and derive nutrients from the environment while producing acid to prevent the overgrowth of pathogenic organisms. S. epidermidis alone makes up 90% of the normal flora on the skin. Alan Gillen, “The Wonderfully Made Design of the Skin and Its Microbiome,” Answers in Depth 9 (July 16, 2014).
-
God built into all living things the need to sustain, repair, and reproduce. In many cases, microbes are essential for these three processes to take place in plants, animals, and humans. This would ensure continuity and stability for the times to come.
From a biblical worldview, infectious diseases and pathogenesis are a secondary state in nature caused by man’s rebellion against the Creator and the subsequent Curse (Genesis 3). It is not the way the Creator intended for man and nature. From Alan Gillen, “Microbes and the Days of Creation,” Answers Research Journal 1 (January 16, 2008): 7–10.
- Gillen, “The Wonderfully Made Design of the Skin . . .”

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis