Serratia marcescens: The Miracle Bacillus
Abstract
The objectives of this article are to explain the mysterious appearance of crimson-colored bacteria on food and communion bread/wafers, over the centuries, as well as to describe the biological basis
Keywords: Serratia marcescens, miracle bacillus, history of bacteriology, Miracle at Bolsena, Blood of Christ, prodigiosin, Robert P. Williams
Blood, Bread, and Bacteria
Imagine finding red spots on your communion bread or wafers. What might you conclude? For centuries, the church concluded that these red spots were the blood of Christ. However, we now know that these red spots were not blood, but colonies of Serratia marcescens bacteria.

Figure 1. Serratia marcescens on communion cracker. Alan L. Gillen image.
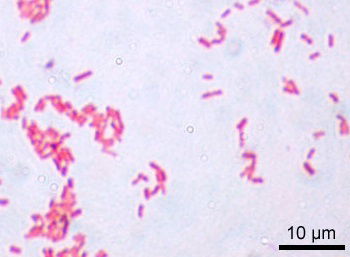
Figure 2. Gram negative cells stain (Wiki commons image). S. marcescens are Gram-negative bacteria, thus red or pink colored) 1000X. The red color is due to a counterstain called safranin.
By Y_tambe (www.gnu.org/copyleft/fdl.html), via Wikimedia Commons.
Serratia marcescens has a long history in the church, as well as in microbiology. Numerous historical incidents recount that the blood red pigment produced by S. marcescens growing in bread was interpreted as a sign of blood. S. marcescens also grows well in damp basements, on food stored in damp places, and is a frequent contaminant in the laboratory. S. marcescens has a fondness for growth on polenta (corn meal mush), bread and communion wafers, where the pigmented, aged colonies have been mistaken for drops of blood (figure 1).
The objectives of this article are to explain the mysterious appearance of crimson-colored bacteria on food and communion bread/wafers over the centuries, as well as to describe the biological basis for the blood-red pigment of S. marcescens. S. marcescens is a wonderful example of design commonly seen in the microbial world. It is most noted for its production of a bright red pigment called prodigiosin. Prodigiosin comes from the Latin word, prodigiosus, which means wonderful, amazing, miraculous, unnatural (supernatural); divine, or dealing with wonders. This is why we refer to it as the “miracle” bacillus.
S. marcescens is a Gram-negative, rod-shaped (bacillus), facultative anaerobic bacterium in the family Enterobacteraciae (figure 2). S. marcescens is often found in the soil, water, on plants and in animals, and is especially common in damp conditions. For example, the pink discoloration and slime (biofilm) on shower corners, toilet basins, and tile grout is S. marcescens bacteria that derive their nutrition from dead skin cells, skin oils, and soap and shampoo residue. After its establishment, Serratia can be difficult to eradicate completely, although possible with 3% hydrogen peroxide or a 5% bleach solution, especially if these solutions are left on the affected surface for several minutes. Rinsing and drying surfaces after use to remove Serratia’s food source and make an ordinarily hospitable environment less inviting can also prevent establishment of Serratia colonies.
The “Blood of Christ” and the History of a Red Mystery
The history of seeing red on food originates in 6th century BC, when Pythagoras reported on the blood substance that sometimes appeared on bread. Then, in 332 BC, soldiers of the Macedonian army of Alexander the Great found that their bread sometimes appeared to have blood on it. The Macedonian soldiers interpreted these bizarre phenomena as evidence that blood would soon flow in the city of Tyre and that Alexander would win. After Alexander’s troops interpreted that victory would be given, they claimed the Middle East and conquered territory all the way to India. In the 1800s, Prussian microscopist Christian Gottfried Ehrenberg (1795–1876) uncovered almost 100 historical references to the miraculous appearance of “blood” on food.
The historian and microbiologist, E. R. L. Gaughran (1969) uncovered more than 35 historical reports of blood flowing from Eucharistic bread. The first such incident was recorded in 1169 in Denmark. From the time of the Middle Ages through the Renaissance period, Serratia regularly grew on communion wafers. This led many to think this was miraculously the blood of Christ. For example, in the dark, damp churches of medieval times, sacramental wafers used in Holy Communion often become contaminated with S. marcescens, which was mistaken for blood and thought to be a miracle.

Figure 3. Miracle at Bolsena. Public domain image, from Wiki commons (click to enlarge).
The most dramatic occurrence was in 1263, four hundred years before Anton van Leeuwenhoek would observe bacteria under a microscope. A blood-like substance appeared in dramatic fashion on the communion bread and on the garments of those breaking bread. This event inspired the artist Raphael to paint his awe-inspiring masterpiece, the Mass of Bolsena, on the walls of the Vatican (Figure 3). In the painting, the German priest Peter of Prague is shown breaking the communion loaf at the Saint Christina Church in Italy. When the priest broke the communion wafer, he thought that it had blood on it and that the bread had truly become Jesus’ flesh! This convinced the priest of miracles and the doctrine of transubstantiation.1
To honor the miracle of Bolsena, Pope Urban instituted the feast of Corpus Christi (Body of Christ) in 1264. The Pope declared it an official miracle, and set aside the communion elements, plates, and garments to place them in a museum. They are still in the Vatican museum today. Neither Peter the Priest nor Pope Urban knew that S. marcescens was the probable cause of this crimson-colored substance.
Bread, both leavened and unleavened, plays a crucial biblical symbol for nutrition, life’s substance and the body of Christ (Mark 14:22). Christ also used wine to symbolize his blood (Luke 22:20). Signs and symbols are important in Christianity to help God’s people remember Him and His Creation. So, many saw the appearance of red growth on bread as still another symbol and miraculous power of the Creator of life.
The early development of microbiology often came with attempts to discredit this alleged miracle of Jesus’ blood appearing on the bread and wafers in Catholic mass. Bartholomeo Bizio, a pharmacist from Padua, Italy, discovered and named S. marcescens as a microbial cause for the “miraculous” bloody discoloration in a cornmeal mush called polenta. He looked at the red spots under a microscope and saw what he described as a fungus. (He may have confused it with red yeast, a single-celled fungus.) In 1817, Bizio moistened some bread and polenta and left them in a warm, damp atmosphere. Twenty-four hours later, both the bread and polenta were covered in red growth. In 1819, Bizio named Serratia in honor of an Italian physicist named Serrati, who invented the steamboat.
The species name, marcescens, was derived from the Latin word that meant fading away, or by extension, decaying or putrefaction (Williams 1980). Bizio chose marcescens from the Latin word for decaying because the microbe caused a putrefaction and deterioration of the polenta bread that it grew on. In 1823, he wrote that the reddening, or “bleeding” of the moist, warm polenta was due to microbial fermentation. Initially, he described S. marcescens as a type of fungus. By 1823, he named the so-called fungus as Serratia marcescens. Later in the 1850s, scientists later reclassified S. marcescens as a bacterium.

Figure 4. Christian Gottfried Ehrenberg, Public domain image, from Wiki commons.
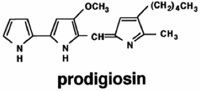
Figure 5. Chemical structures of Prodigiosin. (Gillen 2007)

Figure 6. Dr. Robert P. Williams. Alan L. Gillen image.
Perhaps, the lack of a precision in identifying S. marcescens as a bacterium was due to the poor quality of early microscopes. It was not until 1826 that the invention of the achromatic lens (taking away distracting colors at high magnifications) enabled a clear look at bacteria. J. J. Lister, a Christian optician, made many microscope improvements as to adding graduated draw tubes that made it easier to setup and focus the microscope, additional lenses to act as sub-stage condenser, and a rotating and clamping stage to manipulate the slide. The main significance to Lister-limb microscope was to provide a superb quality to the viewing of microscopic images and invent a mechanism to support a large or heavy microscope (Gillen & Oliver 2009).
Christian Ehrenberg also showed an interest in the red spots found on “bloody bread,” and in 1848 he inoculated those spots on potatoes, bread, and Swiss cheese kept moist with damp paper. In so doing, he may have been the first person to cultivate bacteria. Ehrenberg is also likely to have been the first to use the term bacteria (meaning little rods). In 1836 he had described and named a number of bacteria, including Bacterium and Spirillum. In the mid- to late 1800s, Serratia was renamed Bacillus prodigiosus (i.e., the rod-shaped bacteria with a wonderful red pigment) and Monas prodigiosus (i.e., a small, flagellated bacteria with a wonderful red pigment) before Bizio’s original name was restored in the 1920s.
Prodigiosin Production and Its Complexity
Serratia’s most salient characteristic is its bright red pigment called prodigiosin. Over the years, it has certainly got the attention of churchgoers and scientists alike. It is one of the few bacteria that produce bright pigments, most commonly red, white, or pink with colonies that are 1.5–2.0 mm with a convex elevation. Its color variation was noted as early as 1888. The first person to describe a comprehensive pathway for the biosynthesis of prodigiosin was Dr. Robert P. Williams (1920–1993), a Christian microbiologist (Figure 6).

Figure 7. UV Mutation of Serratia marcescens. Alan L. Gillen image.
Dr. Williams was particularly interested in what controlled the expression of the red phenotype in S. marcescens. Pigment production in Serratia is influenced by several variables, including temperature, nutrient media, and exposure to ultraviolet (UV) light (Figure 7). Some strains of S. marcescens are capable of producing prodigiosin, which ranges in color from dark red to pale pink, depending on the temperature, substrate, and age of the colonies. Most strains of S. marcescens are red under 27º C and white above 28º C. (Pigment and flagella production stops at ~ 28º C). Its most common colors are pink and bright flame red (Figure 8). S. marcescens turns “bloody” red and mucoid only under certain conditions: when the colonies age (umbonate elevation) and when the food, or substrate, has the proper amino acids. These amino acids are often derived from peptone and glycerol where the colonies have aged for more than a week (Figure 9). The medium should be low glucose and low phosphate for maximum production of prodigiosin (see Williams and Quadri 1980).
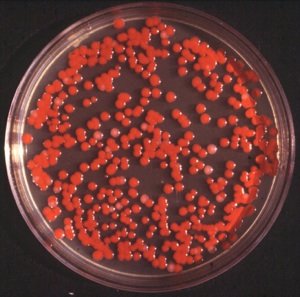 Figure 8. Serratia marcescens typical colony. By de:Benutzer:Brudersohn (www.gnu.org/copyleft/fdl.html), via Wikimedia Commons.
Figure 8. Serratia marcescens typical colony. By de:Benutzer:Brudersohn (www.gnu.org/copyleft/fdl.html), via Wikimedia Commons.
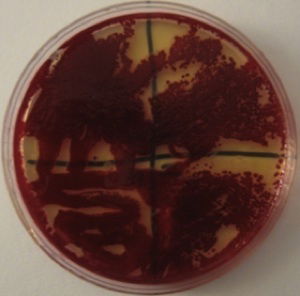
Figure 9. Serratia marcescens blood-red colonies. Alan L. Gillen image.
Figure 10. Blood-Red and White Mutants of Serratia marcescens. Alan L. Gillen image.
Prodigiosin is a time-delayed, secondary metabolite constructed from several amino acids that may accumulate in the cell as a result of primary metabolism (Williams 1973). (Antibiotics produced by bacteria are also secondary metabolites, like prodigiosin. They are not essential for survival, but very helpful in establishing bacteria’s domain in soil.) Four amino acids are needed: proline, methionine, histidine, and alanine. In the lab, they are often derived from peptone and glycerol. Proline is incorporated intact in the prodigiosin molecule; histidine is used indirectly; methionine contributes a methyl group; alanine is entirely incorporated except for a carboxyl group. Prodigiosin, a linear tripyrrole,2 is synthesized in a bifurcated pathway, in which mono- and bipyrrole precursors are synthesized separately and then couple to form the red pigment (Figure 5). There at least 10 different enzymes involved in its synthesis (Williams 1973).
The synthesis of prodigiosin is an irreducibly complex process. An irreducibly complex system is one that requires several interlacing parts to be present at the same time, where the removal of one or more parts causes the whole system to malfunction. Destroy one part and the whole system falls apart. In evolution, a new trait would have to be completely developed, no halfway measures would do. Given this requirement, new features are so complex that Darwinian gradualism is very improbable because an incompletely developed trait would offer no selective advantage.
Pigment Variations of Serratia
Serratia bacteria under warm and humid conditions form distinctive red colonies as they reach maturity. They dissolve into a fluid, viscous state with mucilaginous appearance and an uncanny resemblance to blood. Indeed, from early times, there are many records of the appearance of blood on bread, beans, and certain starch foods such as polenta and potatoes. Now, we would describe this form as a biofilm and the bread as the bacteria’s organosubstrate (i.e. biomatrix).
Although S. marcescens primarily produces bright red pigment called prodigiosin, not many know that some Serratia mutants can be white, purple, pink, orange, and even blue. It produces a wide diversity of color morphs, depending on the partial or complete synthesis of prodigiosin (Williams 1956, 1973). Serratia can “lose” its color both to prolonged sunlight (via UV mutagenesis), or if the temperature rises to our body temperature (37º C), fading away due to decreased prodigiosin production.
In order for it to turn a deep, blood-red color, the bacteria “culture” needs to age (be older than 8 days) and the substrate needs to have a form of glycerol (oil) and to be low in phosphate and sugar. It has long been known that preparing the culture with low phosphate and low glucose solutions can enhance the intensity of the pigment in broth or agar, allowing Serratia to produce enough prodigiosin to have a blood-red, crimson, or maroon appearance, not just bright red or pink.
Prodigiosin Pigment Offers Protection and Enhances Survival
The functions of the pigment have long been pondered, but only recently determined. Many articles say that there is no known function for prodigiosin. In the past, ideas range from flagellar production to the enhancement of the aerosolization of S. marcescens and removal of toxic accumulation of metabolites such as amino acids. It probably functions in all of these, adding to Serratia’s ability to not only survive but thrive in most moist places. It appears that prodigiosin offers protection for Serratia in the natural environment. The red pigment offers protection against excessive UV in sunlight, serves as an antibiotic, and has cytotoxic qualities. It appears that it is worth the energy investment to synthesize prodigiosin when it serves protection against UV light and when it has to compete with fungi in the soil, using it as an antibiotic against neighboring molds and bacteria.
Loss of Pigment May Lead to Germ Genesis
S. marcescens is easily grown in the laboratory, and may serve as a model for genomic decay leading to pathogenicity. The metabolic pathways involved in prodigiosin production are numerous and complex, so mutations affecting any of these pathways could result in loss of pigment production (Lim 2003). Most (70–93%) Serratia infections are caused by naturally occurring white mutants, which also often bear antigenic flagella and appear to be more host dependent (Ball et al. 1977; Ding and William 1983). Williams and Quadri (1980) determined that UV irradiation could transform prodigiosin-producing organisms into non-producing white mutants.
Over the past three years at Liberty University, we have sought to develop and elucidate a research model using Serratia marcescens (see Appendix, Table 1). In our three years of experiments to maximize random mutations correlated with loss of prodigiosin, radiation time influenced mortality and the number of white mutants that grew (Figure 10). When exposed to ultraviolet light for 15 seconds, bacterial mortality was 40–75% (no white mutants); 30 seconds, 75–90% (some white mutants); 120 and 180 seconds, 95–99% (numerous white mutants). (The percentages refer to mortality rate.) The degeneracy of prodigiosin production in our experiments has some similarity (i.e., some UV white mutants have flagella and have lost some metabolic capabilities) to that in clinical strains (933 and WF), suggesting a possible link to pathogenicity. The initiation rate (the time it takes to become a pathogen) and degree of pathogenicity in the UV white mutants is unknown, but those that develop flagella are more likely to become pathogenic via some undetermined mechanism(s). There appears to be an inverse relationship of prodigiosin production with motility (i.e. flagella), pathogenicity, and host dependency. The increased number of flagella seems to attract bacteriophages, which in turn, via horizontal transfer, bring about added virulent traits. Serratia, once a common saprophyte designed to break down organic matter in forests, has become a pathogen capable of breaking down human tissue. A loss of information may make it more vulnerable to becoming a pathogen.
Serratia is an Opportunistic Pathogen that Strikes the Weakened Body
Biologists once thought that S. marcescens was non-pathogenic and, because of the pigment it produces, it was used widely to trace bacterial transmission. Since 1906, physicians used S. marcescens to study how epidemics get started because this bacterium generally was considered to be harmless. In the 1950s, the U.S. Army conducted a study called “Operation Sea-Spray” to study wind currents that might carry biological weapons. They filled balloons with S. marcescens and burst them over San Francisco just to see where the “harmless” red bacteria would land. However, shortly thereafter, doctors noted a drastic increase in pneumonia and urinary tract infections.
Only since the 1960s did microbiologists recognize S. marcescens as an opportunistic human pathogen. In the hospital, Serratia tends to colonize the respiratory and urinary tracts of adults, rather than the gastrointestinal tract. Serratia causes about 2% of nosocomial infections of the bloodstream, lower respiratory tract, urinary tract, surgical wounds, and skin and soft tissues of adult patients. Outbreaks of S. marcescens meningitis, wound infections, and arthritis have occurred in pediatric wards. In most cases Serratia infections have occurred in people who have compromised immune systems or those who are aged.
Although most S. marcescens strains are still harmless saprophytes in the soil, a few opportunistic pathogens have been reported in 2011. In six hospitals, the Alabama Department of Public Health has investigated the outbreak of cases of S. marcescens bacteremia due to contaminated catheters used for a liquid nutrition formula from the Meds IV pharmacy. A total of 19 cases and 9 deaths have been reported during Spring 2011. Approximately 35% of those receiving the contaminated liquid nutrition product became ill from S. marcescens. Meds IV has voluntarily discontinued production and recalled all of its IV compounded products on March 24, 2011. Perhaps these strains, like 90% of other clinical strains, were the non-pigmented S. marcescens (Ball et al. 1977; Clayton and Graevenitz 1966; Graevenitz 1980).
The Creator’s Signature, “Red-Lettered” Bacteria
How do we know that this pigment is designed? Design is simply the purposeful arrangement of parts (or chemicals). Design is evident in the number of separate, interacting parts (chemicals) necessary to assemble prodigiosin. The greater the specificity of interacting parts and numbers of conditions to produce it, the greater the confidence that it was not made by accident. In other words, the intricacy and complexity point to creative design.
So maybe S. marcescens was not the miracle that the Pope expected, but this tiny organism does remind us of the wondrous invisible life that is all around. The pigment from Serratia may not be the blood of Christ, but it does in fact have a brilliant, crimson color that attracts attention. Its natural production of variable bright colors testifies of the Creator’s artistic abilities, and the ability of the bacterium to produce the pigment and adapt under varying environmental conditions suggests the Sustainer’s foreknowledge of this microbe’s need to survive.
The Creator formulated not only the plan for S. marcescens but also created the original non-pathogenic Serratia. He is not only the chief Architect of the red pigment, but is also the Manufacturer of the prodigiosin components. He keeps everything going because He is the Maintainer (Colossians 1:17). The predictable color of the prodigiosin at lower temperatures exists because of the order of the precise plan was produced by an intelligent cause. Prodigiosin is no accident of evolution. Most creation biologists would go a step further and say that it is clear, physical evidence of fingerprints from the Master’s hand.
Is Serratia marcescens, the “Miracle” Bacillus?
So, should S. marcescens still be called the “miracle” bacillus? Yes and no; it depends on how one defines a miracle. It is not a proof of the Catholic doctrine of transubstantiation. It is neither the blood of Christ, nor the blood of any person or animal. But, it does demonstrate God’s care for His tiniest creation, and perhaps its bright red color can be used to remind us of Christ’s sacrifice for us. It has certainly got the attention of its observers over the centuries.
A miracle is an event attributed to divine intervention (Morris 2004). Dr. Henry Morris breaks down miracles into two different types: Grade A and Grade B. Grade “A” miracles are those that involve a new creation, like those recorded in Genesis 1 and 2. Grade “B” miracles are events that fit within natural law; but their timing or frequency is unusual; they might be called miracles of providence, or answers to prayer.
The scientific establishment today insists that naturalism must govern all phenomena and that real miracles—especially the supernatural creation of all things in the beginning—are impossible. So they seek to explain away all alleged miracles in terms of natural processes. We are not using these assumptions in our study of Serratia. However, Jesus Himself cautioned against being credulous about miracles: “Except ye see signs and wonders, ye will not believe” (John 4:48, KJV). Jesus also said to people: “An evil and adulterous generation seeks after a sign” (Matthew 12:39, KJV). Therefore, we should be cautious about reports of so-called miracles. God’s “laws of nature” are good laws and are essential for the orderly operation of His creation. Genuine Grade “A” miracles would be rare outside Bible times (Morris 2004).
Consider miracles of providence. These occur when God acts within His ordained natural processes to bestow His benevolence on His creation. They may involve the precise timing of a natural occurrence, or an unusual rate of a process (Morris 2004). A providential event may be extremely unlikely from a statistical point of view, but within the realm of possibility. Usually we consider miracles to be something desirable, a gift of God’s love.
We believe in the miracles of the Bible; but historically the red pigment on food, especially communion bread/wafers, etc. was most likely S. marcescens, not blood. Given the magnitude of Serratia on food in communion instances, they may have been a miracle of providence to startlingly remind His people of the precious blood of Christ. The communion elements in the “ordinary” sense are symbols to help us remember the life and death of Christ.
We should not be surprised that bright pigments in nature remind us that beauty is a part of God’s plan. In Matthew 6:28–29, Jesus reminds us that God arrays lilies with color, beauty, providing soil and sun. He consistently cares and provides for His creation. If we extend this principle to bacteria and the pigment of S. marcescens, God provides prodigiosin for protection, survival, and extension of its domain in soil.
Although probably not the miracle that the Pope and Priest of the Middle Ages thought it was, Serratia is still a symbol of the Creator’s care. Like the metaphor, or symbol, of the bread for the body of Christ and wine for the blood of Christ, so too is the crimson-colored S. marcescens. The “miracle” is that an Awesome Artist would care enough to sustain and protect even His tiniest creations. He has left His signature on it—one of red-letter importance. It testifies to the very good and wondrous invisible life all around us. Indeed, it is something marvelous!
Dedication
This article is dedicated to Dr. Robert P. Williams. Dr. Williams was the President of American Society for Microbiology (ASM), from 1983–1985. He was a humble, brilliant researcher on S. marcescens and many other bacteria, and spent over 40 years studying prodigiosin from 1949–1992. He published 128 peer-reviewed papers, wrote 15 chapters for books, and authored his own microbiology text with Kenneth Burdon (Burdon and Williams 1968). He was a very thoughtful mentor who treated every graduate student with individual care and provision. Perhaps, the most amazing thing about his life is that he accomplished all of this in a wheelchair, having been crippled by polio in 1953. He did it all with a smile and with Christian character. Back
References
Ball, A. P., D. McGhie, and A. M. Geddles. 1977. Serratia marcescens in a general hospital. Q. J. Med. N. Ser. 46:63–71.
Burdon, K. L., and R. P. Williams. 1968. Microbiology, 6th ed. New York: Macmillan Co.
Clayton, E. and A. V. Graevenitz. 1966. Nonpigmented Serratia marcescens. JAMA 197, no. 13:1059–1064.
Ding, M. J. and R. P. William. 1983. Biosynthesis of prodigiosin by white strains of Serratia marcescens isolated from patients. Journal of Clinical Microbiology 17, no. 3:476–480.
Gillen, A. L. 2007. The Genesis of Germs: Disease and the Coming Plagues in a Fallen World. Green Forest, Arkansas: Master Books.
Gillen, A. L. 2008. Using Serratia marcescens as a model to elucidate aspects of germ genesis in bacteria. Creation Research Society Quarterly 1:240.
Gillen, A. L. and J. D. Oliver. 2009. Creation and the Germ Theory. Answers in Depth 4, no. 1.
Gaughran, E. R. L. 1969. From superstition to science: the history of a bacterium. Transactions of the New York Academy of Sciences 31:3–24.
Graevenitz, A. 1980. The Genus Serratia. Cleveland, Ohio: CRC Press.
Lim, D. V. 2003. Microbiology, 3rd ed. Dubuque, Iowa: Kendall Hunt.
McIntyre, M. 2011. CDC and ADPH investigate outbreak at Alabama hospitals; products recalled [News Release]. Retrieved from www.fda.gov/Safety/Recalls/ucm249068.htm.
Merlino, C. D. 1924. Bartolomeo Bizio’s letter to the most eminent priest, Angelo Bellani, concerning the phenomenon of the red colored polenta. Journal of Bacteriology 9:527–543.
Morris, H. M. 2004. Miracles: Do They Still Happen? Why We Believe in Them. Green Forest, Arkansas: Master Books.
Williams, R. P. 1956. Symposium on Bacterial Pigments. Bacteriol. Rev. 20:282–284.
Williams, R. P. 1973. Biosynthesis of prodigiosin, a secondary metabolite of Serratia marcescens. Appl. Microbiol. 25:396–402.
Williams, R. P. and S. M. Quadri. 1980. The pigments of Serratia, Von Graevenitz, A. and S. J. Rubin, eds. in The Genus Serratia, pp. 31–75. Boca Raton, Florida: CRC Press.
Yu, V. L. 1979. Serratia marcescens: Historical perspective and clinical review. New England Journal of Medicine 300:887–893.
Appendix: Table 1. List of Common Strains
| Name and Description | ATCC® Number | Resistance |
|---|---|---|
| 1. Nima (D-3) (Wild-type, Texas Medical Center, Blood Red) | 29632™ | Resistant |
| 2. BS 303 A (Wild-type, Pond Water, Magenta) | 13880™ | Sensitive |
| 3. BS 303 B (Wild-type, Pond Water, Magenta) | 13880™ | Resistant |
| 4. 9-3-3 (Clinical Mutant TMC, white) | 29634™ | Resistant |
| 5. WF (Clinical Mutant, white-pink) ATCC® Number: | 29635™ | Resistant |
| 6. UV Mutant (Derived from Nima) | N. A. | Resistant |
| Notes: TMC = Texas Medical Center; ATCC® = American Type Collection | ||
Footnotes
- Transubstantiation. During the Mass, at the consecration in the Lord’s Supper (Communion), the elements of the Eucharist, bread and wine, are thought to be transformed into the actual body and blood of Jesus and no longer bread and wine, but only retain their appearance of bread and wine.
- Tripyrrole is a molecule that has three pyrrole compounds linked together. It is an aromatic organic compound, a five-membered ring with the formula C4H4NH. It is a colorless volatile liquid that darkens readily upon exposure to air. Another pyrrole, tetrapyrrole, is the biosynthetic precursor to many natural products such as hemoglobin.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis


