The Genesis of the “Brain-Eating” Amoeba
At a Glance
Naegleria fowleri (Fig. 12) is a free-living microscopic ameba and causes a rare and devastating infection of the brain called primary amebic meningoencephalitis (PAM). (It is also known as Naegleriasis.) This ameba is commonly found in warm freshwater (e.g. lakes, rivers, and hot springs) and soil. Naegleria fowleri usually infects people when contaminated water enters the body through the nose. Once the ameba enters the nose, it travels to the brain where it causes PAM, which is usually fatal. Infection typically occurs when people go swimming or diving in warm freshwater places, like lakes and rivers. In very rare instances, Naegleria infections may also occur when contaminated water from other sources (such as inadequately chlorinated swimming pool water or heated and contaminated tap water) enters the nose. You cannot get infected from swallowing water contaminated with Naegleria.
The genetic differences between nonpathogenic and pathogenic amoebae are minor, and coding for stealth/entry from the nose to the brain via the olfactory nerve seems to be the only difference. Pathogenic N. fowleri appears to have emerged from a prolonged heat spell and an adaptive mutation in recent times. Rapid multiplication, diversification, and adaptation to prolonged changes in new environments appear to be themes in the Creator’s plan for life.
Abstract
The Creator made all creatures, including amoebas, with variation and plasticity in their genome for global differences of climate, terrain, and environment. God made all creatures to multiply and fill the earth. Diversification and speciation from the original kind would be normal even in a perfect world. Naegleria fowleri is a global amoeba pathogen that is the causative agent of primary amoebic meningoencephalitis (PAM), thus termed “brain-eating.” Many people are evidently exposed, based upon antibody levels, to this amoeba while swimming in warm waters. Only a small percentage succumb to the germ, but once invasion happens, only about 1% survive. Because it is such a devastating disease, most people do not survive the infection. This common pathogen is a free-living ameboflagellate present in very warm water during the summer time.
There is strong indication that the pathogenic N. fowleri differentiated from the nonpathogenic Naegleria lovaniensis on the American continent, then migrated to Europe and the rest of the world. It appears there has been an overall loss of genetic information. Nonpathogenic N. gruberia (40.9 MB1) may have been closer to the original free-living amoeba “kind.” Over time, more than 10 MB of DNA may have been lost in the transition; then another 1 MB from Naegleria lovaniensis (30.8 MB) to N. fowleri (29.6 MB). Most of these N. fowleri types are found in thermally “polluted” water. This could also be an indication that prolonged heat spells create excellent conditions for rapid adaptation. It has been postulated that at the hot surfaces in thermally polluted water (46℃, or 115° F), competition would favor the pathogenic species; but it could also be that these hot surfaces give an excellent opportunity to N. fowleri to change into different types quickly. The genetic differences between a nonpathogen and pathogen are small: adhesion to host tissue and coding for stealth/entry from nose to the brain via the olfactory nerve seems to be only differences between the two closely related species. Pathogenic N. fowleri appears to have emerged from a prolonged heat spell and an adaptive mutation in recent times.
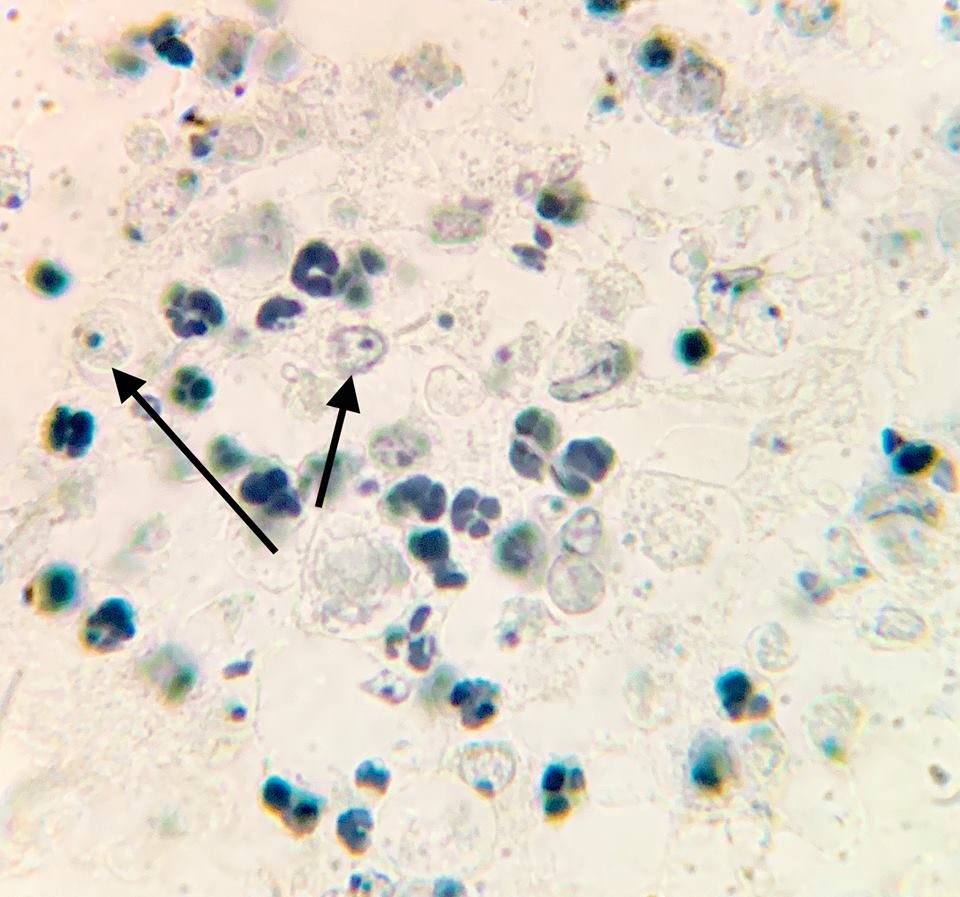
Figure 1. Naegleria fowleri at 1500 X. Histological, Hematoxylin section of mouse cerebellum infected with Naegleria fowleri, photographed at oil immersion (1500 X) with a Zeiss compound microscope lens. The image shows extensive necrosis and inflammation. The very dark cells with lobed nuclei are neutrophils, and the cells with prominent endosomes are trophozoites of N. fowleri (arrow). Note the absence of cysts and biflagellated forms. The amoeba adheres to the olfactory epithelium and invades the brain via the olfactory nerve. Image credit: Alan Gillen.
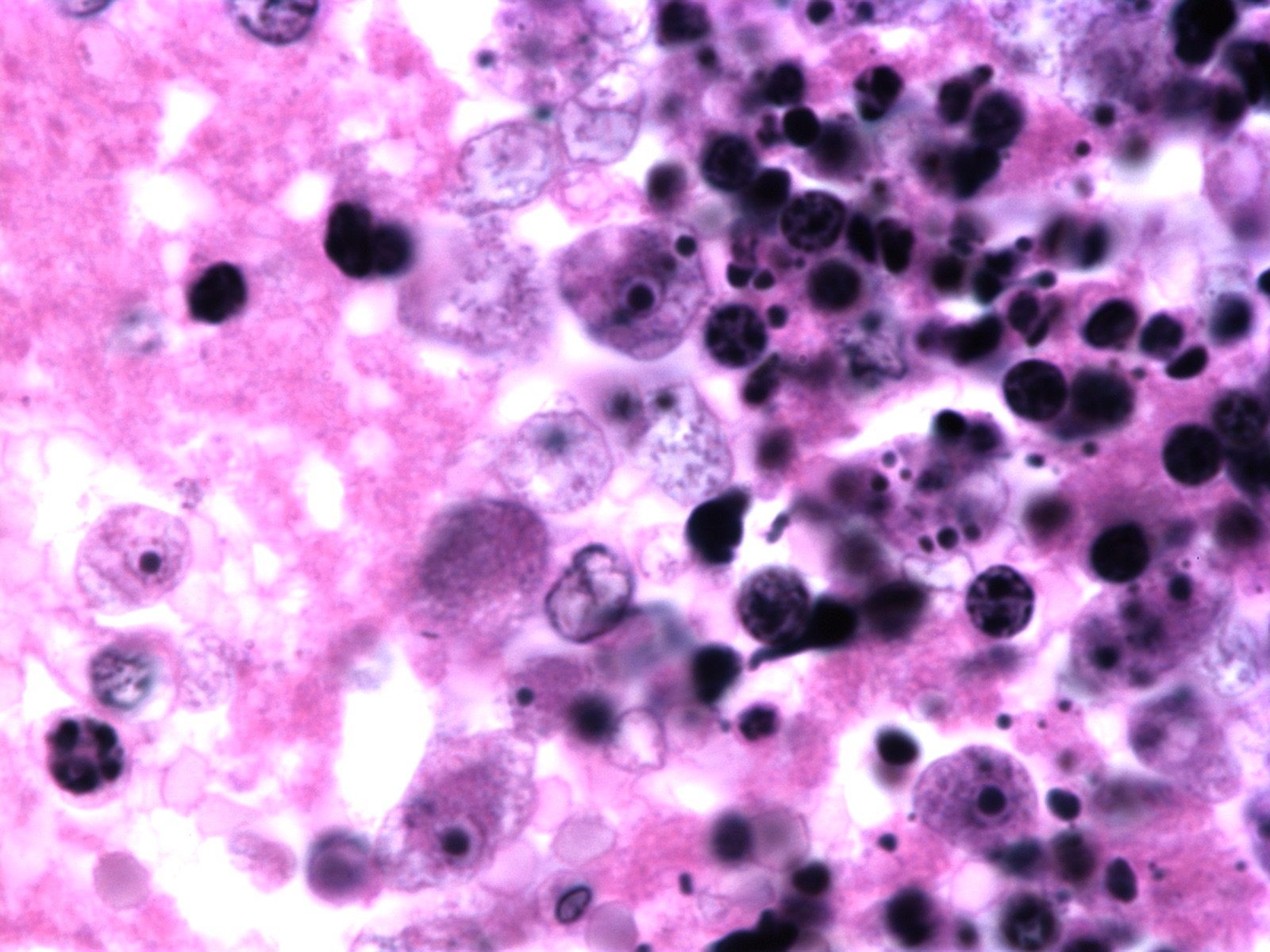
Figure 2. Naegleria fowleri H & E stain. Hematoxylin and eosin stain image from a well-stained Ward's 92W4127 slide shot at 1500x on a Nikon Eclipse E600. It shows four clear trophozoites with large endosomes surrounded by a clear nucleus (one invading healthy tissue on the left), several more out of focus with black phagocytosed material and extensive necrosis (as shown by darkly stained nuclei), and one neutrophil (bottom left). Image credit: John Sullivan.
Rapid multiplication, diversification, and adaptation to prolonged changes to environments appear to be themes in the Creator’s plan for life after man’s fall into sin. Changes in these amoeboflagellates have occurred, but they are still amoebas.
Introduction
A few years ago, in the state of Virginia, a nine-year-old boy named Christian Alexander Strickland was stricken with a rare illness after swimming and being dunked in a lake. Christian suddenly had a headache, fever, and nausea. The week before he died from meningitis (PAM), he was his usual active self. He was a very active young boy. Like many his age, he played basketball and baseball and went swimming and diving. The trouble started when contaminated water was forced into his sinus cavity. The family was told that he had been dunked while “horse-playing” with other kids at the lake. He went to a fishing camp in Henrico County (near Richmond, VA). Christian died just days after falling ill.
Initially, the diagnosis was common meningitis and encephalitis. The hospital ran lab tests on his blood and cerebrospinal fluid (CSF), but no signs of bacteria, fungi, or viruses were seen in the blood or CSF. However, the CSF contained pus and slow-moving amoeboid cells, and the indirect immunofluorescent test identified Naegleria fowleri (Fig. 3 and Fig. 12). The antibodies to the amoebae were positive. A state health official confirmed that a school-age child in the central region of Virginia had died from this rare meningitis. For over a month, state epidemiologists at the Virginia Department of Health were in the news every evening reporting the dangers of warm to hot waters in local lakes.
In the news media, we read reports about brain-eating and diarrhea-causing amoebas that kill people. Surface waters often contain microbial life, pathogens and parasites, such as freshwater amoebas Naegleria fowleri, Cryptosporidium, and Giardia. They have become widespread due to increased flooding locally and globally. Water and foodborne illnesses continue to rise across the country, and the CDC lists them as their number two priority in 2019 for monitoring, funding, and preventing. Diarrheal diseases affect over two billion people annually across the globe. What is the origin of these pathogens and parasites? Their proximate (immediate) and ultimate genesis is unknown. The purpose of this article is to provide a possible explanation for the puzzling origins of Naegleria and other amoebas. In previous articles, I have already addressed the origin of E. coli, Giardia and other pathogens and parasites.
When and how did parasites arise? These questions are a mystery for both creation and evolutionary biologists.
Where have these water parasites come from? What is their origin? Their proximate origin is of great importance to epidemiologists and public health officials. Their “first” origins are of interest to scientists as well: both creation and evolutionary biologists. Their origin and genesis are a mystery. In this article, we are attempting to provide insight on the origin of brain-eating Naegleria fowleri and similar amoebas. These are important water-borne (protozoan) parasites, but they have different explanations for their origin. They seem to share a common pre-adaption to changing conditions in their environment. The adaptive mutation that allows progression from a nonpathogen to a pathogen (strains that break the blood-brain barrier) may be an explanation for the existence of the brain-eating amoeba. A normally free-living amoeba emerges into a stealth parasite due to gene loss and a minor DNA base change.
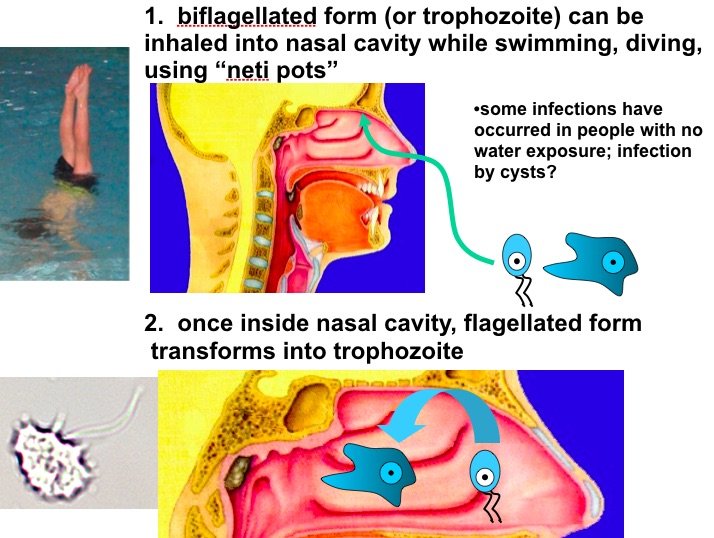
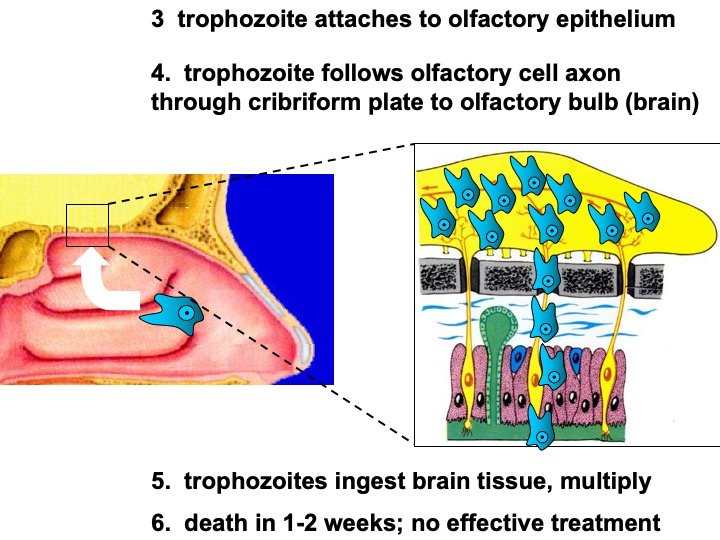
Figure 3. Amoebas invading the brain (modified lecture PowerPoint slides). Image credit: Alan Gillen.
Symptoms and Treatment
- Sudden, severe headache
- Stiff Neck
- Fever
- Nausea and vomiting
- Confusion
- Loss of Balance
- A change in the sense of smell or taste
- Sensitivity to Light
- No ideal treatment (amphotericin B, miltefosine works sometimes)
When and how did parasites arise? These questions are a mystery for both creation and evolutionary biologists. While there are no definitive answers, it is generally agreed among secular biologists that parasites “changed” among very diverse groups of free-living progenitors. One of the earlier organisms probably formed an initial causal relationship with another organism. One member of the pair, perhaps due to preadaptation, developed a gradual dependency on the other. The term preadaptation in the context of parasites denotes the potential in a free-living organism for adaptation to a parasitic lifestyle. This predisposition may never become operational if the organism remains free-living; however, if for some reason the parasite becomes associated with a potential host, this latent ability becomes important for survival should hostile environmental conditions develop. Preadaptations can be structural, physiological, and developmental (Bogitsh, Carter, Oeltmann 2019). Creationists might refer to these as mediated designs or anticipatory systems—genetic and physiological systems made to adapt and anticipate future environmental changes. These systems are a type of preadapted design.
Very Good2 Amoebas
Amoebas in today’s world already have an affinity for human host tissue without causing harm (commensalism). For example, Entamoeba gingivalis lives in the pocket of gum tissue around the teeth, and Entamoeba coli lives commensally with intestinal tissue. Both species have a preadaptation to living in hosts, and minor changes could make them pathogenic. In addition, free-living (non-pathogenic) amoebas in pond water can eat eukaryotic Paramecium, Volvox, Stentor, tardigrades, and more. This phagocytic eating behavior fits within a pre-fall world; this is a preadaptation to eating larger cells. Minor changes to these “good designed” amoebas could induce predatory behavior of large animals and humans and make them parasitic.
Figure 4. Naegleria fowleri in the brain. Image credit: CDC .
The typical pond amoeba is the best-known protozoan (Fig. 6 and Fig. 7), having a remarkably flexible body that can change shape at will. Its single-cell body is a tiny mass of living matter within a thin, flexible, semipermeable membrane. There are no visible internal structures except a nucleus. The cytoplasm, however, has numerous vacuoles and various other inclusions. Free-living, harmless amoebae, such as Amoeba proteus, reach large proportions, some having diameters of about 400 µm or more. These organisms may be found in infusions made with dead leaves or grass or in the material scooped from the bottom of shallow, quiet ponds.
Amoebae are characterized by their curious method of locomotion. They continually send out, first from one and then from another part of their surface, blunt projections called pseudopodia (“false feet”). Some of the semifluid cytoplasm of the amoeba flows into each new pseudopod, while the older pseudopodia go back into the general mass. Thus, the animal moves about slowly and irregularly, continually changing shape and never progressing long in any one direction (amoeba is derived from a Greek word meaning “change”). Amoebae capture bits of solid food by surrounding them with a pseudopod and enclosing the food particle within a vacuole. Digestive enzymes are then secreted into the vacuole. If the particle is not acceptable food, it is expelled from the cell (Fig. 6 and Fig. 7). Free-living amoebas feed on microbes such as algae, bacteria, and protists. When a typical free-living amoeba encounters a suitable food, the cytoplasm flows around the prey and engulfs it. The cytoplasm secretes powerful enzymes into the food vacuole and digests it in a food vacuole (Gillen 2019).
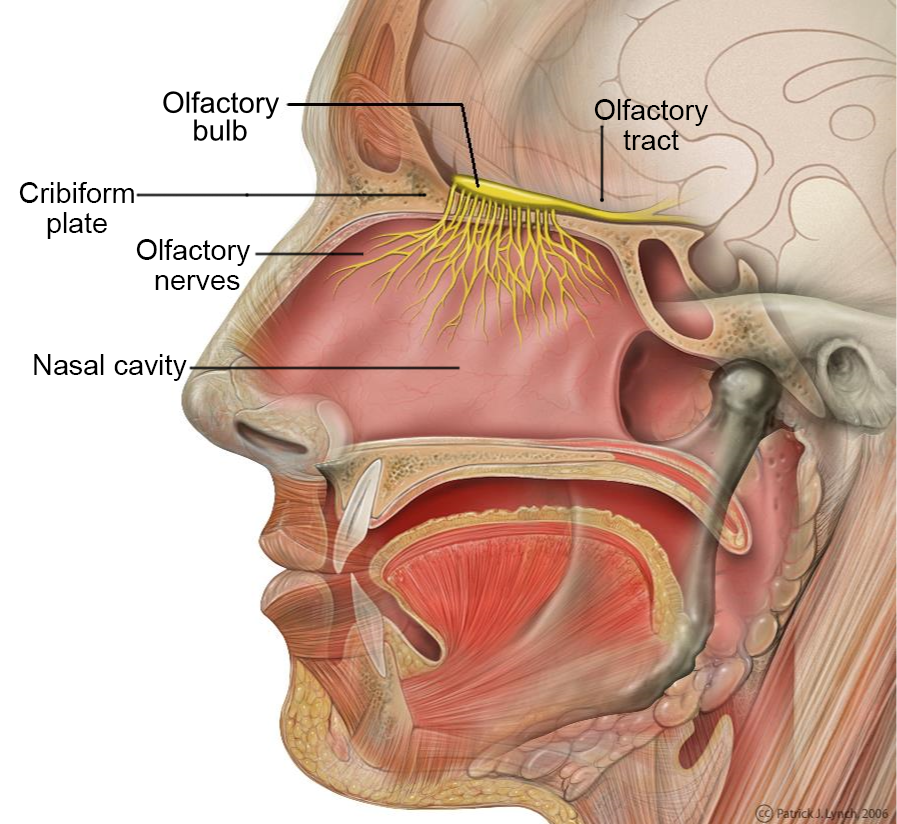
Figure 5. Head anatomy with the olfactory nerve and the olfactory bulb highlighted. The amoebas primarily invade the brain along this nerve pathway. Image credit: Patrick J. Lynch, medical illustrator. (labeled by was_a_bee) [CC BY 2.5], via Wikimedia Commons.
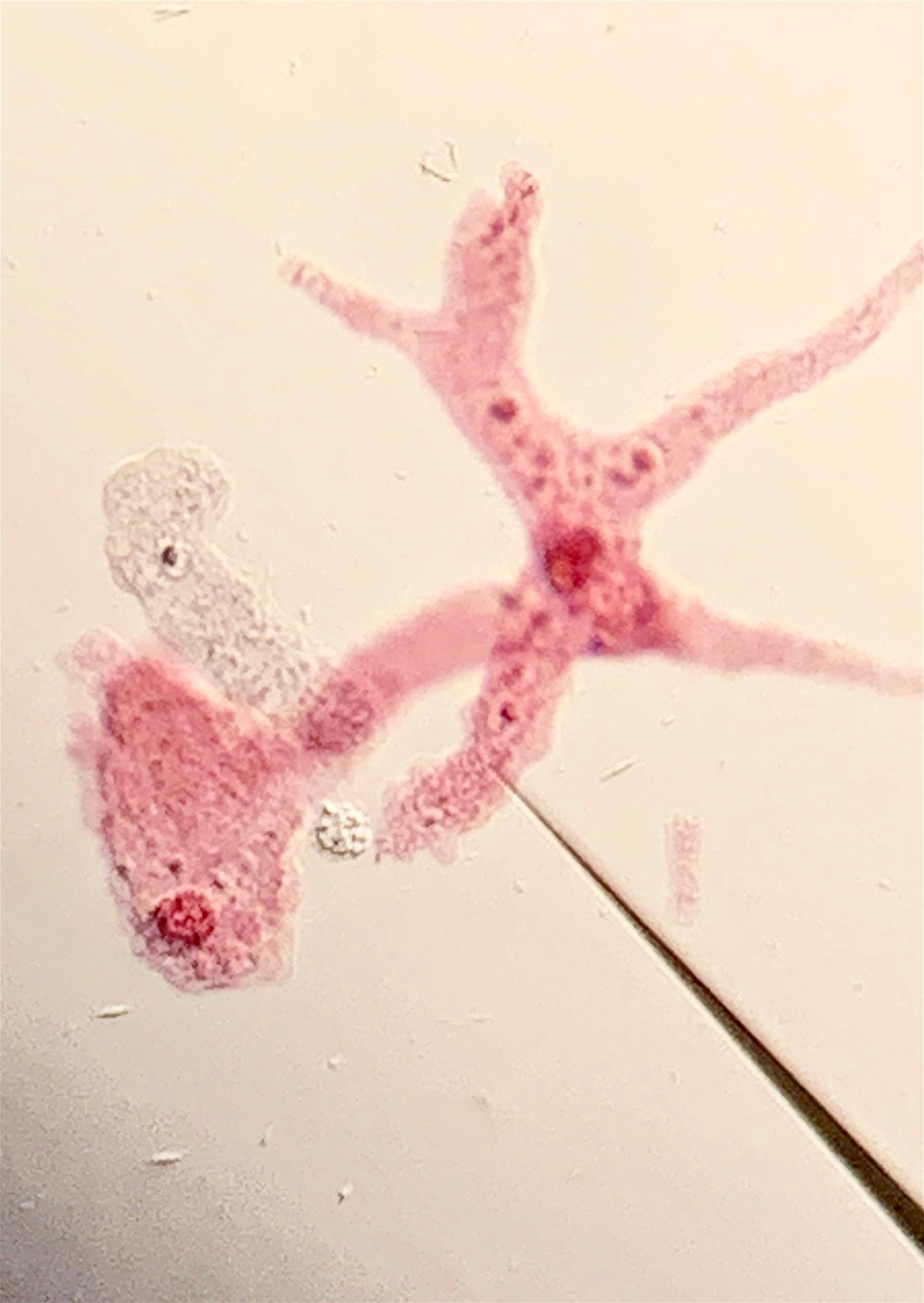
Figure 6. Amoeba proteus is a common free-living amoeba of freshwater ponds and lakes. It is the most common amoeba studied in general biology classes. Image credit: Alan Gillen.
Naegleria spp. are protists belonging to the family Vahlkampfiidae in the class Heterolobosea. More than 40 species have been characterized, but only Naegleria fowleri is known to be a human pathogen. N. gruberi, N. fowleri, and N. lovaniensis most likely “devolved” from a common ancestor based on sequence data. One species has emerged from another through gene loss or minor mutations. Secular biologists call this evolution, but it is devolution and adaptation. Even secular biologists suggest that it is plausible that N. fowleri and N. lovaniensis descended from a recent common ancestor (Fig. 8 and Fig. 9). Naegleria spp. are free-living amoebas with multiple stages. As amoeboflagellates, they are able to transform under changing environmental conditions from the amoeboid form into fast moving flagellates or resting cysts. This distinguishes them from common pond Amoeba spp. that only have the amoeboid (pseudopod) stage, most likely a different “kind.” As free-living eukaryotes with different life stages, including facultative pathogenic species, Naegleria shows a high plasticity regarding morphology and pathogenicity.
A similar freshwater, facultative, free-living amoeba genus with pathogenic species, often in the same habitat but perhaps different niche, is Acanthamoeba. Acanthamoeba species (esp. A. culbertsoni) are among the most common amoebae of freshwater ponds and lakes. Acanthamoeba are an important part of the soil ecosystem. They are responsible for regulating bacterial populations. When bacteria are consumed, nutrients are released back into the soil. In a natural soil cycle where Acanthamoeba eat bacteria, the overall effect is increased nutrient availability for plants. The cysts are viable for years in dry soil. They differ from Naegleria fowleri (37º C, or 98.6º F) in that there is no flagellated form, they are primarily a soil dweller (the major genus in soil, 27º C or 80. 6º F), and cysts form in tissues of infected individuals.
Parasitic Amoebas
At one time, free-living and terrestrial amoebas were merely considered to be elements in the microenvironment that cycle energy and prey on other small organisms (bacteria and algae) that had little impact on human health or economic importance. In the late 1950s, it became necessary to reconsider whether these small amoebas were as innocuous as they had been thought to be. At first, they were unknown organisms that appeared in monkey kidney tissue cultures used in the production of the polio virus (Marquardt, Demaree, and Grieve 2000). Some of these were found to produce disease in experimentally inoculated lab rodents. Then, in the early 1960s, isolates of amoebas were found causing problems in humans. In 1965, the brain-eating amoebas were found in humans in Australia, Florida, Texas, and Europe.
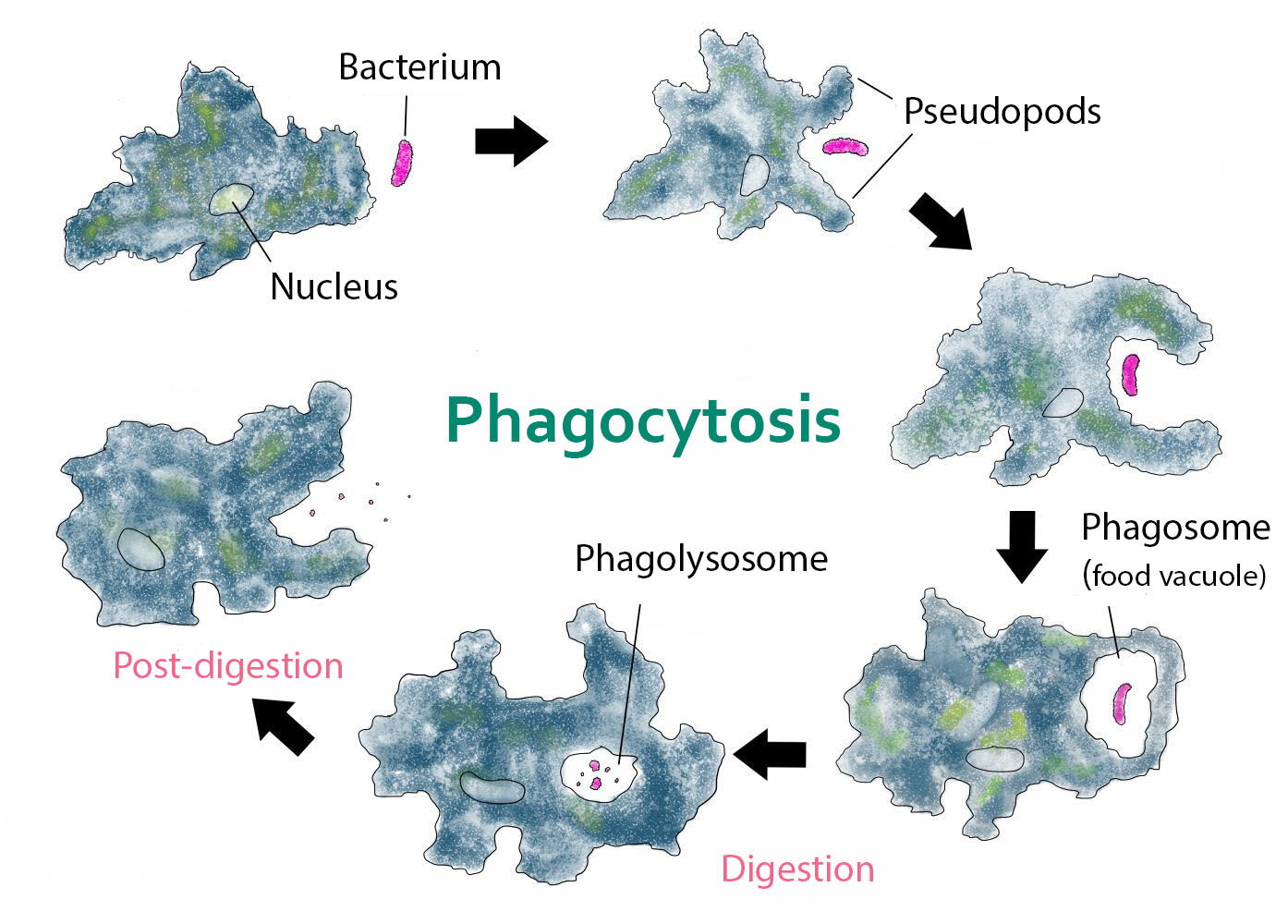
Figure 7. Process of Phagocytosis by amoebas. The amoeba engulfing a bacterium is in pink. Image credit: Kate Taylor [CC0], via Wikimedia Commons.
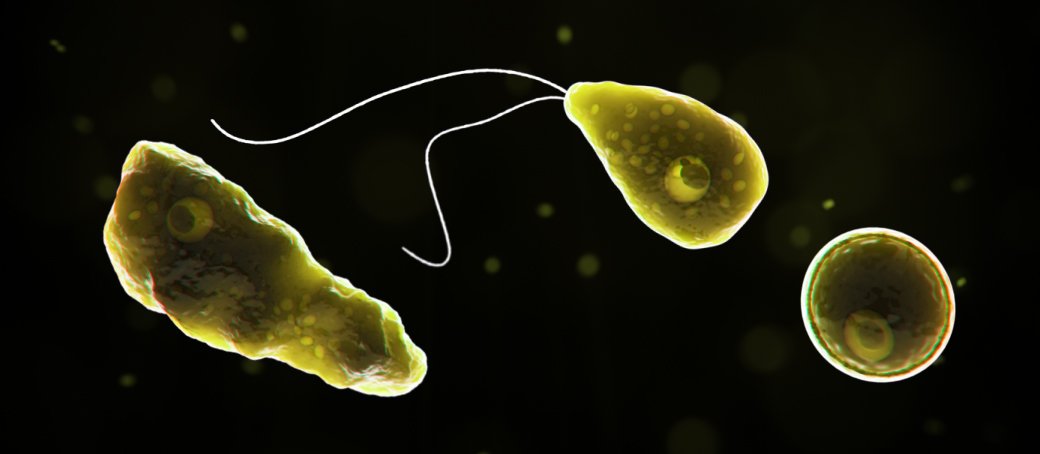
Figure 8.Three forms of Naegleria fowleri: trophozoite, cyst, and biflagellated form illustrated in 3-D form. Image credit: CDC (page archived 2019).
Pathogenic, free-living amoebae such as Acanthamoeba spp., Balamuthia mandrillaris, and Naegleria fowleri—popularly termed “brain-eating amoebae”—have been known to cause infections of the human central nervous system. There were about 144 cases reported for brain-eating amoebae through the end of 2018. They are all facultative, opportunistic parasites, meaning the amoebae are not always parasites since they are not physiologically dependent on a host to live but can become a parasite if the opportunity should arise.
Pathogenesis of Brain-Eating Amoebas
There are 3 stages: trophozoite, cyst, and biflagellated form. Their numbers rise when water temperature rises. The biflagellated form (or trophozoite) can be inhaled into the nasal cavity while swimming, diving, or using Neti pots. Some infections have occurred in people with no water exposure; this may indicate the possibility of infection by cysts. Once inside the nasal cavity, the flagellated form transforms into trophozoites. The trophozoites attach to the olfactory epithelium, then follow the olfactory cell axon through the cribriform plate to the olfactory bulb (brain). These trophozoites ingest brain tissue and multiply. The result is typically death in one to two weeks; there are minimal effective treatments. Formally, this disease is known as primary amebic meningoencephalitis (PAM). There are several cases in the U.S. annually which occur in the summer time due to increased swimming activity. It also occurs in warm lakes and thermal pools.
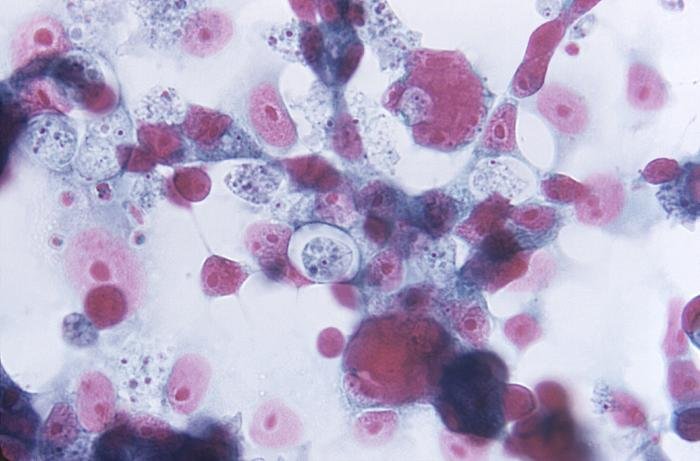
Figure 9. Naegleria gruberia is a well-studied nonpathogenic amoeba that may share a common ancestor to Naegleria fowleri. Image credit: CDC/Dr. George R. Healy.
- Domain Excavata Class Heterolobosea, Family Vahlkampfiidae
- Ameboid trophozoite or biflagellated cell in fresh water, eating bacteria
- Actively grow at 85ºF (30ºC), but can live up to 115ºF (46ºC) possible
- Long-lived cyst if dried
- All 3 stages infective if inhaled into nasal cavity
- Water activities (main route of infection), Neti pots, ritual ablution
- Diagnosis: trophozoites or biflagellated cell in CSF (no cysts present)
Some of the most devastating infectious diseases are those that affect the nervous system or include the brain and spinal cord. Because of the critical importance of the brain and spinal cord, the body’s design includes bone barriers (Gillen 2009). Even pathogens and parasites that are circulating in the blood cannot enter the brain due to the blood brain barrier (Tortora, Funke, and Case 2019). Sometimes trauma disrupts the barrier and often is associated with serious consequences. The cerebrospinal fluid (CSF) of the central nervous system lacks many of the defenses of the blood and makes the entry of any pathogen/parasite especially dangerous. In the case of Naegleria fowleri, it enters the brain from the olfactory nerve in the nose and passes through the cribriform plate. It invades the bottom of the cerebrum, travels to the front of the cerebellum, and gradually spreads throughout the brain. Like the free-living Amoeba proteus consuming bacteria and other protozoans through phagocytosis, these Naegleria do in fact eat the brain.
This microbe ultimately kills 97% of the people it infects. There are a few treatments that if administered quickly work, and a number of treatments are currently being studied. The name Naegleria fowleri may misguide people as to why this microbe’s infections are so deadly. Some data indicate that it is our own immune system’s response to the amoeba is responsible for much of the brain’s damage. Experiments on cells in the lab offer clues. It appears the amoeba damages tissue directly by releasing harmful proteins. When N. fowleri attacks the brain, it starts a domino effect. The initial inflammation triggers dangerous swelling. The amoeba produces enzymes that digest mucus and protein, which it swallows up with its “feeding cups” or amoebastomes. N. fowleri is attracted to chemicals released by nerve cells. All amoebas have enzymes that digest bacteria and yeast in the present world. The olfactory nerves (nerves of smell) travel from the roof of the nasal cavity through openings in the skull (cribriform plate) into the base of the brain. The amoeba consumes the nerve cells while migrating along these tracts until it reaches the brain. The brain is an especially rich food source for the amoeba due to its high levels of oxygen, glucose, and living cells. Damage to the brain is caused by severe inflammation, direct injury, and bleeding. Death is caused by the resulting severe brain swelling (Roberts, Janovy, and Nadler 2013). This swelling is the reason for its name, the “brain-eating” amoeba. New data demonstrates that is not the whole story. Many are exposed to the amoeba; however, they often do not get sick.
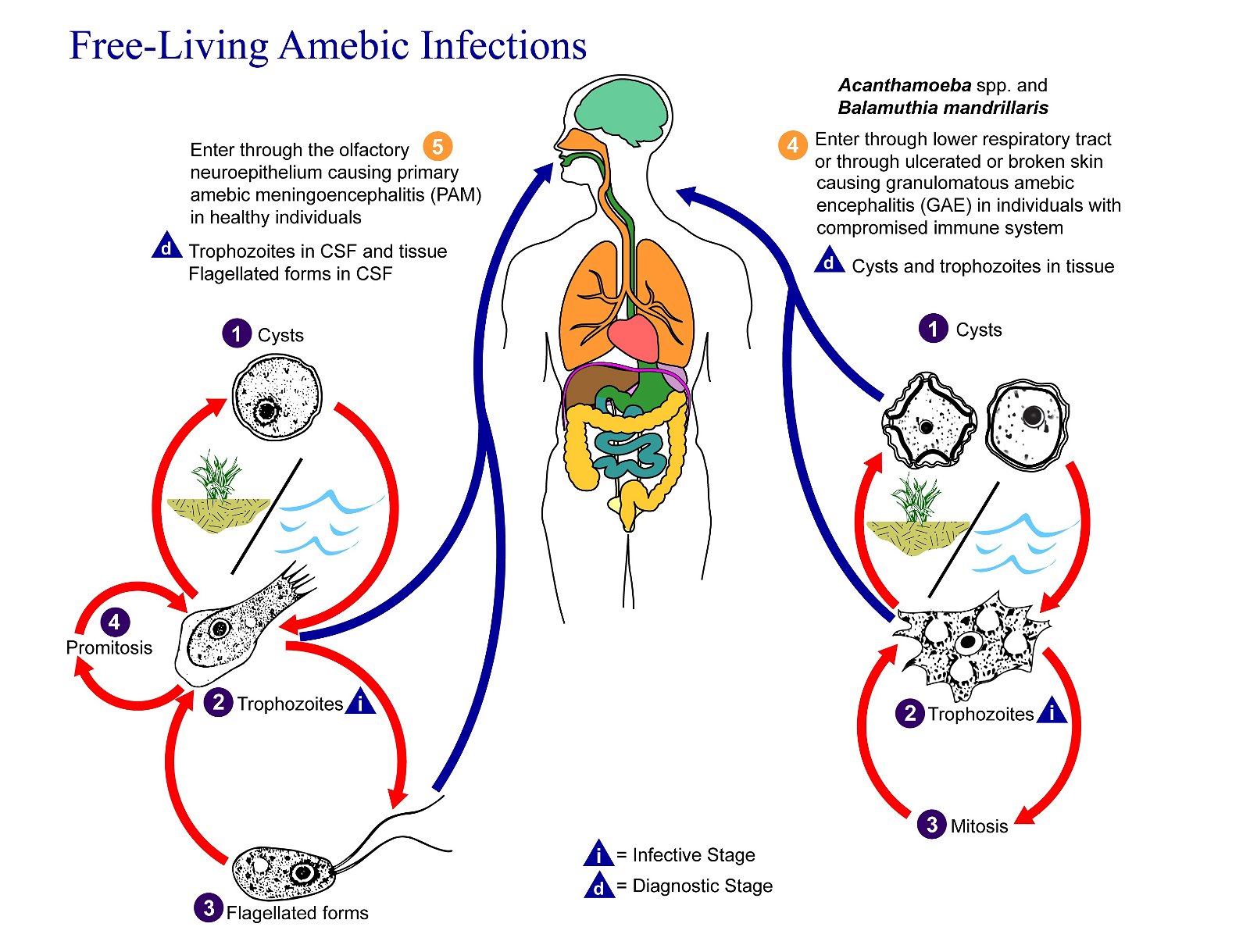
Figure 10. Naegleria fowleri Life Cycle. Naegleria fowleri has three stages in its life cycle: Number 1, ameboid trophozoites, number 2, flagellates, and number 3, cysts. The only infective stage of the amoeba is the amoeboid trophozoite. Trophozoites are 10–35 µm long with a granular appearance and a single nucleus. The trophozoites replicate by binary division during which the nuclear membrane remains intact (a process called promitosis) The number 4. Trophozoites infect humans or animals by penetrating the nasal tissue. The number 5 shows the amoeba migrating to the brain. The number 6 indicates the movement via the olfactory nerves causing primary amebic meningoencephalitis (PAM). Image credit: CDC/Alexander J. da Silva, PhD; Melanie Moser.
Figure 11. Thermal Waters near Aiken, SC. The large SRL thermal lakes and streams on the SRL plant represents a location where N. fowleri could have first emerged. Image credit: Google Earth.
Origins of Virulence and a Pathogen of High Consequence
The changes from nonpathogen to pathogen are fairly minor.
There is strong indication that the pathogenic N. fowleri emerged from the nonpathogenic amoeba, Naegleria lovaniensis, in America and spread throughout the globe. Evidence for this “evolving” comes from the fact that N. fowleri shares 9547 genes with N. lovaniensis. The changes from nonpathogen to pathogen are fairly minor. Analysis of the genomes of both of these species of Naegleria showed the similarity of N. lovaniensis and N. fowleri along with the minor changes involved with the transition from nonpathogenic to pathogenic (Liechti et al. 2018). “There is no evidence of virulence differences between the types of N. fowleri. Two other Naegleria spp. are pathogenic for mice, but human infections due to these two other Naegleria spp. are not known” (DeJonckheere 2011). The different types of N. fowleri arose from each other by the addition of a few base pairs. The progression from a nonpathogen to a pathogen (proteolytic enzyme-producing strain) results from repeats in the internal transcribed spacer (ITS)1 sequence and a C to T transition at location 31 in the 5.8S rDNA (ribosomal DNA). The latest evolved types have only been detected in Europe until now. Two more pathogenic Naegleria spp. have been described, but human infections due to those have not been reported yet.
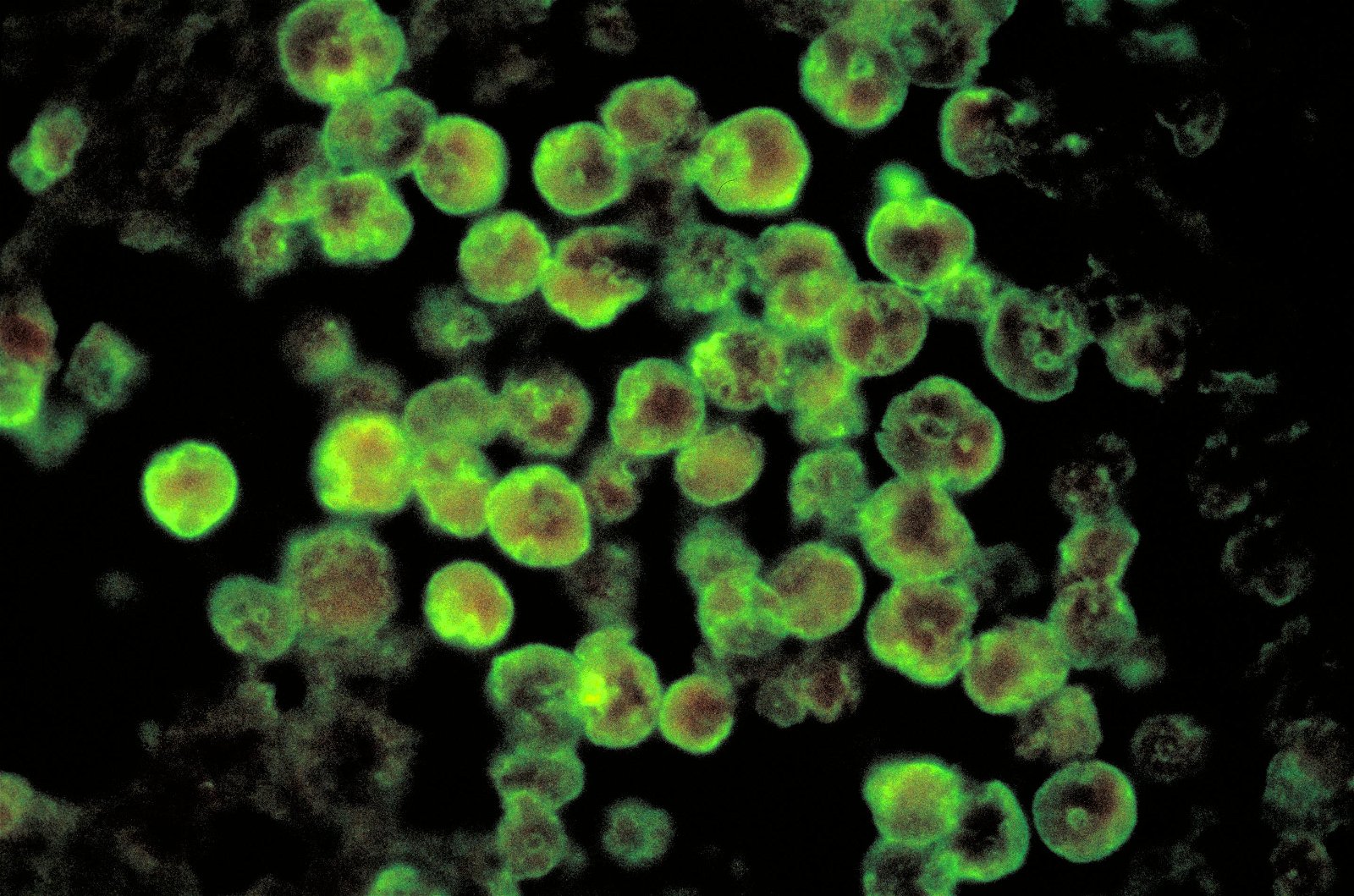
Figure 12. Histopathology of PAM due to N. fowleri direct florescent antibody stain. Image credit: CDC/Dr. Govinda S. Visvesvara.
Researchers have established that there are several types of N. fowleri, which can be distinguished based on the length of the ITS1 and a one-base-pair transition in the 5.8S rDNA (gene for rRNA). Biologists are referring to identification based on 18S rDNA (gene for rRNA) sequence data to identify the amoeba species. The ITS is a region of the ribosomal DNA that is typically used to delineate between strains or closely related species. That is why they say the different “types” can be distinguished using ITS and the 5.8S rDNA sequence. There is strong indication that the pathogenic N. fowleri differentiated (“evolved”) from the nonpathogenic Naegleria lovaniensis on the American continent. The genetic differences between a nonpathogen and pathogen appear to be relatively small and possibly coding for stealth/entry from nose to the brain via the olfactory nerve. Pathogenic N. fowleri appears to have emerged from a prolonged heat spell: it lost DNA and an adaptive mutation may have occurred in recent times.
There appears to be a difference in the origin of the water samples with amoebas from America and Europe and the water samples from other continents. In America and Europe, most of the water samples came from thermally polluted industries, mainly electricity power plants, while on other continents most of the samples were taken from naturally heated water.
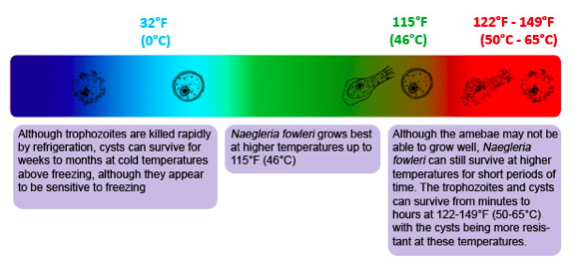
Figure 13. Temperature Ranges for Naegleria fowleri are wide ranging in hot water. The amoeba is a heat-loving (thermophilic) and able to grow and survive at very high temperatures, such as those found in hot springs and in the human body. Image credit: CDC (page archived 2019).
Most of these N. fowleri types are found in thermally polluted water (Fig. 11). This could be an indication that prolonged heat spells create excellent conditions for rapid adaptation. It has been postulated that at the hot surfaces in thermally polluted water (46℃ or 115ºF), competition would favor the pathogenic species (Delattre and Oger, 1981). It could also be that these hot surfaces give an excellent opportunity to N. fowleri to change into different types quickly. Higher temperatures usually increase growth rates and cause more genetic errors. These errors lead to more mutations in other microbial and protozoan species. This action may be a result of altered proofreading of the DNA. It appears there has been a loss of genetic information as shown in Table 3. According to Mike Behe (2019), breaking a gene will lead to diversification; this is devolution not evolution.
| N. gruberia Nonpathogen |
N. lovaniensis Nonpathogen |
N. fowleri Pathogen |
|
|---|---|---|---|
| Genome Size (MB) | 40.9 | 30.8 | 29.6 |
| Repeat Content (%) | 5.1 | 3.5 | 2.5 |
| Temperature Range Habitat reference | Wide Thermointolerant |
Intermediate Thermotolerant |
Narrow Very Thermotolerant (Fig. 13) |
| Speed of Transformation: Cyst, flagellate, amoeboid stages | Fast | Intermediate | Slower |
| Metabolism | Diverse, Adaptive | Intermediate | Specific, Parasitic |
| Environment/Niche | Generalist | Intermediate | Specialist |
N. fowleri has a distinction among Naegleria species. The attachment to a mammalian host cell membrane or defending against lysis are important mechanisms of pathogenesis. Vesicular transport and regulation of protein secretion are important points in the pathogenesis of N. fowleri. Beside membrane vesiculation, recent studies identified secreted cysteine and metalloproteinases, which are involved in the degradation of the host extracellular matrix and enable the penetration of the blood-brain barrier through cribriform plate via olfactory nerve (Liechti et al. 2018).
Local Research and Practical Implications
Microbes change over time, and these changes can happen quickly.
Microbes change over time, and these changes can happen quickly. The microbes appear to be preadapted for the changes seen. This preadaptation has practical implications for public health. In our local research, these parasites are widespread and cause intestinal diseases, which are especially harmful to immunocompromised individuals. We are exploring local surface waters to discover the factors, such as rainfall, that influence the growth of amoebas, coliforms, Serratia marcescens, Giardia, and Cryptosporidium. During the time of high rainfall, we saw amoebas flourish in soil/leaf matter, as well as Giardia, E. coli, and other bacteria. Extreme environments appear to induce change, or variation, of microbes within a kind due to adaptive mutations. We have seen this first hand.
Summary and Conclusions
There is strong indication that the pathogenic N. fowleri differentiated from the nonpathogenic Naegleria lovaniensis on the American continent, then migrated to Europe and the rest of the world. It appears there has been an overall loss of genetic information. Nonpathogenic N. gruberia (40.9 MB, Fig. 9) may have been closer to the original free-living amoeba “kind.” Over time, more than 10 MB of DNA may have been lost in the transition, then another MB from Naegleria lovaniensis (30.8 MB) to N. fowleri (29.6 MB). Most of these N. fowleri types are found in thermally polluted water. This could also be an indication that prolonged heat spells create excellent conditions for rapid adaptation. It has been postulated that at the hot surfaces in thermally polluted water (46℃ or 115ºF) competition would favor the pathogenic species, but it could also be that these hot surfaces give an excellent opportunity to N. fowleri to change into different types quickly. The genetic differences between a nonpathogen and pathogen are small, and coding for stealth/entry from nose to the brain via the olfactory nerve seems to be the only difference.
Mike Behe (2019) states, “The first rule of adaption evolution: Break or blunt any functional gene whose loss would increase the number of offspring.” N. fowleri appears to have emerged in thermal lakes to survive higher temperatures. It lost genes (becoming host dependent), and the altered DNA allowed it to penetrate the blood-brain barrier. Pathogenic N. fowleri appears to have emerged from a loss of genetic information (possibly making it more dependent), a prolonged heat spell, and an adaptive mutation in recent times. Rapid multiplication, diversification, and adaptation to prolonged changes in new environments appear to be themes in the Creator’s plan for life. Changes in these amoeboflagellates have occurred, but they are still amoebas.
Compared to free-living eukaryotes, obligate parasites, including trypanosomes and amoebas, show a functional reduction of their gene repertoire in adaption to the life cycle in the host, resulting, for example, in reduced metabolic capabilities. In addition, minor DNA mutations appear to help survival in novel environments. Adaptive mutations can readily fit within a creation model where variation mechanisms are a designed feature of microbes and protozoans. Adaptation, variation, and mediated design are creation concepts. God designed created kinds with genes that could be turned on to help adapt to new environments, similar to campers who take Swiss army knives with them to face unknown challenges in the wilderness. The diversity of tools serves different functions for specialized needs in camping, fishing, and survival. Some adaptive design traits can only be expressed under certain conditions to allow microbes, plants, animals, andhumans to fill the earth as environments change over time (Genesis 1 and 8). God programmed organisms with mechanisms that would be triggered under certain conditions to allow organisms to survive, thrive, and to function in new environments (Purdom 2013). Microbes may have internal genetics to adapt to future environmental changes and make changes to adjust to a world that has varying conditions over time. Biological adaptation systems offer an alternative view to chance evolutionary models that are blind to future needs. An omniscient awesome Creator could provide for creatures’ needs in a changing world (Gillen 2019).
Amoebas were designed to live in fresh water and moist soil, eating bacteria and protozoans. If they were preadapted to withstand extreme heated environments or were capable of subsequent adaptation to it, they have become progressively more dependent upon the new environment or might have migrated to other hospitable environments (i.e. the brain) to meet their new metabolic needs after losing information. It is evident that amoebas already eat other protozoans and have some predisposition to “eat” proteinaceous material via phagocytosis. Now, the amoebas devastatingly eat the brain in a fallen world. The genesis of germs is best explained by a “Creation, Curse, and Contagion” worldview. For the Christian, we have the hope that someday creation will be restored and there will be an exodus of the germs through Jesus Christ. Then the angel[a] showed me the river of the water of life, bright as crystal, flowing from the throne of God and of the Lamb (Revelation 22:1).
References
Behe, M. Darwin Devolves: The New Science about DNA that Challenges Evolution. New York, NY Haper One, 2019.
Bogitsch, B. J., et al. Human Parasitology, 5th ed. Waltham, MA: Academic Press, 2018.
De Jonckheere, J. F. “Origin and Evolution of the Worldwide Distributed Pathogenic Amoeboflagellate Naegleria fowleri.” Infection, Genetics and Evolution 11, 7 (2011): 1520–1528, https://doi.org/10.1016/j.meegid.2011.07.023.
Gillen, A. L. Body by Design: Fearfully & Wonderfully Made. Green Forest, AK: Master Books, 2009.
Gillen, A. L. The Genesis of Germs: Disease and the Coming Plagues in a Fallen World. Green Forest, AK: Master Books. 2019.
Gillen, A. L. and F. Sherwin. “The Genesis of Malaria: the Origin of Mosquitoes and Their Protistan Cargo, Plasmodium falciparum.” Answers in Genesis. June 19, 2013, https://answersingenesis.org/biology/disease/the-genesis-of-malaria//.
Gillen, A. L. and F. Sherwin. “The Design of Giardia and the Genesis of Giardiasis.” Answers in Genesis. July 19, 2017. https://answersingenesis.org/biology/microbiology/design-giardia-and-genesis-giardiasis/.
Gillen, A. L. et al. “Serratia marcescens, the ‘Flame’ Strain: the Genesis of a New Variant. A Newly Described Strain with Prolific Pigment Produced at High Temperature.” Answers in Genesis. June 19, 2018. https://answersingenesis.org/biology/microbiology/serratia-marcescens-flame-strain-genesis-new-variant/.
Liechti, N., et al. “The Genome of Naegleria lovaniensis, the Basis for a Comparative Approach to Unravel Pathogenicity Factors of the Human Pathogenic Amoeba N. fowleri.” BMC Genomics 19 (2018): 654, https://doi.org/10.1186/s12864-018-4994-1.
Marquardt, W. C., et al. Parasitology and Vector Biology 2d ed., San Diego, CA: Harcourt Academic Press, 2000.
Tortora, G.J., et al. Microbiology, an Introduction, 13th ed. San Francisco: Pearson Benjamin/Cummings, 2018.
Answers in Depth
2019 Volume 14
Answers in Depth explores the biblical worldview in addressing modern scientific research, history, current events, popular media, theology, and much more.
Browse VolumeAcknowledgement: Funding for the materials (in part) were made possible by Provost Research Initiative grant.
Footnotes
- MB or Mb = Mbp = mega base pairs (bp) = 1,000,000 bp
Most creation biologists believe that God created all life as being “very good” (Genesis 1:31). Even today, a vast majority of life forms are beneficial to man and nature. Many larger life forms live in a beneficial relationship with smaller life forms that biologists call symbiosis (Greek sym, “together”; bios, “life”). In general, symbiosis in nature involves a large organism (the host) and a smaller organism (the symbiont). There are three types of symbiosis in nature. Mutualism is a type of symbiosis where both host and symbiont have a beneficial relationship. Commensalism is a type of symbiosis where one life form is benefitted and the other is unharmed. In these two symbiotic relationships, two different organisms live together in harmony.
After creation was corrupted by man’s sin in the Garden of Eden, some of the original created animals and other life forms degenerated into parasites. Thus, parasitism is the third type of symbiosis in which one organism (the parasite) benefits at the expense of the host. Parasites are usually smaller than their host, exhibit a high degree of specialization, and reproduce quickly and in vast numbers when compared to their host. In parasitism, the parasite’s “goal” is to produce more progeny (offspring) into the next generation. Parasites are extremely “successful” organisms in the world today. Current estimates place the number of children in the U.S. infected with various types of parasitic worms at about 55 million. Worldwide 4–5 billion people are infected with parasites. Classic examples of human parasitism include interactions between man and diverse protozoans (e.g., Giardia and Plasmodium), worms (e.g., tapeworms and flukes), or ectoparasites (e.g., fleas, ticks, and mosquitoes). Parasites have a complexity of structure and function. Parasites frequently have complex life cycles. There is an intricacy with their host. Parasites might be seen as having a “broken fellowship” with their host. They may have started good, but something bad happened between them. Parasites are a secondary state in nature—one that God did not start in original creation.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis


