Eukaryote Without Mitochondria Is Not a Product of Evolution
News to Know
Abstract
Having no mitochondria, this protist is said to have “evolved beyond the known limits that biologists circumscribed.”1
News Sources
- NPR: “Look, Ma! No Mitochondria”
- Science Daily: “Change the Textbooks: This Eukaryote Completely Lacks Mitochondria”
Mitochondria are the energy factories that ordinarily generate most of the energy in nucleated (eukaryotic) cells. Bacteria have neither nuclei nor mitochondria, but textbooks say that all eukaryotic cells have mitochondria or some degenerate form of them. Now hiding in the low oxygen environment of a pet chinchilla’s gut, scientists have found a unicellular protist that doesn’t. (A protist is a eukaryotic microorganism; neither a plant, animal, or fungus, a protist can be unicellular or colonial.) Named Monocercomonoides, this unusual microorganism doesn’t have the slightest trace of mitochondria. How does it survive? And does its existence lend support to evolutionary notions about the origin of the eukaryotic cells that make up all multicellular organisms?
Not a Trace
Monocercomonoides is not the first eukaryote thought to be without mitochondria. Some other protists, such as the diarrhea-causing parasite Giardia, were once thought to lack them. But while further investigation found that those organisms have at least an abbreviated form of mitochondria or some mitochondrial proteins, Monocercomonoides does not.
This discovery is such a shock because even eukaryotes using an alternative energy-generating system need mitochondrial machinery to do other things. Dr. Anna Karnkowska, lead author of the report about Monocercomonoides published in Current Biology, explains, “In low-oxygen environments, eukaryotes often possess a reduced form of the mitochondrion, but it was believed that some of the mitochondrial functions are so essential that these organelles are indispensable for their life.”2 And a reduced mitochondrion, she points out, is “still a mitochondrion and it has some important function for the cell.”3
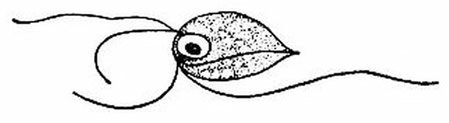
This is a species of Monocercomonoides, a flagellated protist. Monocercomonoides is the first eukaryote known to have no mitochondria at all. Image by Lois Roth and Edwin Willis, via Wikimedia Commons.
But Monocercomonoides has no mitochondria nor any evidence that it ever did. “We have characterized a eukaryotic microbe which indeed possesses no mitochondrion at all,” Dr. Karnkowska says.4 In other eukaryotes, the nuclear DNA contains some of the genes required to assemble mitochondria, but no such genes are present in Monocercomonoides. Neither does it contain any genes ordinarily found in mitochondrial DNA.5 It also contains no genes for making the energy-extracting enzymes present in mitochondria.6 “This amazing organism is a striking example of a cell which refuses to adhere to the standard cell biology text book, and we believe there may be many more similar examples in the so far hidden diversity in the world of microbial eukaryotes—the protists.”7
Energy and More
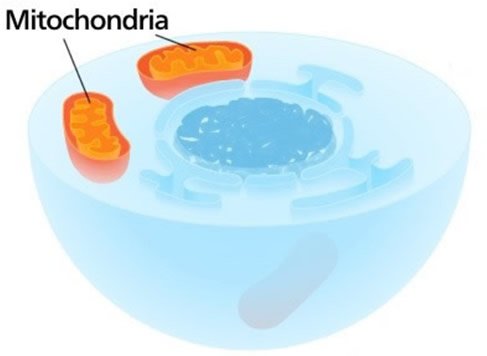
This cross-sectional illustration of a eukaryotic cell shows the dark nucleus in the center and two mitochondria, which generate a lot of energy. Mitochondria, which contain some DNA, also help synthesize iron-sulfur clusters, important components of many proteins. Image by Kelvinsong, via Wikimedia Commons.
While mitochondria are known as the powerhouses of eukaryotic cells, they are not the only way cellular biochemistry has of extracting energy from available fuels. Monocercomonoides gets some energy from glucose, using anaerobic metabolic pathways that operate in the cytoplasm of all sorts of cells. And it obtains a lot more energy by using a series of enzymes that break down the amino acid arginine.8
But mitochondria, which contain DNA, do more than generate energy. They also help synthesize some very important protein components called iron-sulfur clusters.
Iron-Sulfur Clusters
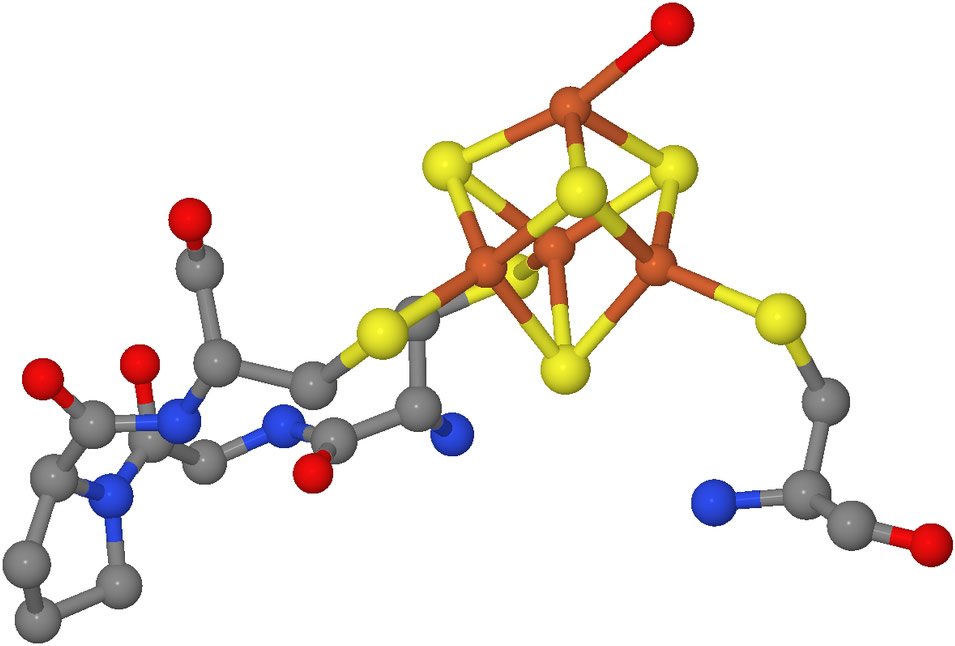
This is a molecular model of a portion of the enzyme aconitase with its attached iron-sulfur cluster, represented by the orange and yellow portion of the model. With the iron-sulfur cluster in place, this enzyme catalyzes the interconversion of citrate and isocitrate, a critical step in glucose metabolism. Although it still serves other functions, aconitase has no catalytic activity when the iron-sulfur cluster is not present. Image by Ayacop, via Wikimedia Commons.
Iron-sulfur clusters are molecular subunits containing iron and inorganic sulfur. They are vital components of a variety of proteins. Iron-sulfur clusters are essential for energy metabolism, DNA repair, and regulating gene expression in accord with a cell’s environmental conditions.9 Abnormalities in iron-sulfur cluster synthesis are associated with several debilitating diseases.10
Iron-sulfur clusters often function as cofactors—helpers in enzymatic processes. With their affinity for electrons, iron-sulfur clusters can readily bind electron-rich substrates for their companion enzymes.
Iron-sulfur clusters can readily receive or release single electrons. Therefore, they can facilitate both oxidation and reduction reactions and participate in electron transport systems. And by attracting key amino acids in a long peptide to itself, (like cysteine in the accompanying illustration, see caption) an iron-sulfur cluster is able to hold a protein in the folded shape (conformation) vital to the proper function of many sorts of proteins.
Because of their versatile chemical properties, iron-sulfur clusters are essential in all cell types. Therefore, iron-sulfur clusters are believed by evolutionists to have been an important stepping-stone in the origin of life. Many evolutionists imagine that life evolved thanks to the chemicals and conditions in hydrothermal vents where iron and sulfur would have been plentiful. However, the fact that some sort of molecule is ubiquitous in living things does not mean its presence would make possible that which is scientifically untenable—the spontaneous origin of life. (Learn more about problems with these notions in “Attempts to Trace Life Back to Chemical Origins Still Maps the Willful Ignorance of the Hunters.”)
Iron-sulfur clusters are produced by a series of steps. Naturally, in prokaryotic cells, all the steps occur in the cytoplasm, and the final stages in the assembly process are cytoplasmic in all cells. However, the initial steps are performed by mitochondrial proteins in every known eukaryotic cell. Even protists with abbreviated versions of mitochondria have the mitochondrial machinery to make iron-sulfur clusters. Yet Monocercomonoides’s genome contains no genes for these mitochondrial-type iron-sulfur-cluster-building enzymes. Instead, it contains four genes resembling those that produce iron-sulfur cluster assembly equipment in bacteria, archaea, and plastids.13 (Two other protists with abbreviated mitochondria also have genes like these, and scientists think they might have acquired them from bacteria, as we’ll discuss below.)
Mitochondrial Acquisition and Loss
Dr. Karnkowska and colleagues believe the unicellular protist’s ancestor evolved with mitochondria and then lost them when they were no longer needed. Therefore, Dr. Karnkowska declares, “This organism has evolved beyond the known limits that biologists circumscribed.”14
Monocercomonoides contains no detectable footprint that mitochondria were ever there.
Remember, Monocercomonoides contains no detectable footprint that mitochondria were ever there. So how do the scientists that introduced Monocercomonoides to the world know that the protist’s ancestors once had mitochondria? Actually they don’t.
The major reason these scientists are certain Monocercomonoides must have had mitochondria is that “it is now widely accepted that mitochondria or mitochondrion-related organelles (MROs) are essential compartments in all contemporary eukaryotes and that mitochondrial endosymbiosis took place before radiation of all extant eukaryotes.”15 In other words, evolutionists assume that mitochondria evolved in the common ancestor of all eukaryotes and therefore must have once been present in this eukaryote.
Evolution of Mitochondria
This is a model of viperin, a viral inhibitory protein produced by cells. Its anti-viral activity depends on an iron-sulfur cluster, and this model reveals why. The protein’s three-dimensional conformation—its tertiary structure—is ordinarily stabilized by bonds to an iron-sulfur cluster. The iron-sulfur cluster, bonded to three cysteine amino acids arrayed around a central pocket, holds viperin in its correct shape. The central pocket in this model is the space between the magenta-colored cysteine amino acids. Each iron atom attaches to a cysteine. Cysteine, because it has a side chain that is readily oxidized, binds in this way to the iron-sulfur cluster, holding it in position. (Attachment to cysteine seems to be the way iron-sulfur clusters are usually attached to their companion protein.11) The iron-sulfur cluster in turn holds the entire folded three-dimensional shape in position. If any of these cysteines is replaced due to a mutation, the iron-sulfur cluster is lost and the molecule partially unfolds. Loss of the cysteines results in loss of anti-viral activity, apparently due to this unfolding of the protein’s tertiary structure, the folded 3-dimensional shape that is essential to its activity.12 Image by Shubhasis Haldar et al., “The Presence of the Iron-Sulfur Motif Is Important for the Conformation Stability of the Antiviral Protein, Viperin,” PLOS One 7, no. 2 (2012): doi:10.1371/journal.pone.0031797.g001.
The popularly accepted evolutionary story to explain how the common eukaryotic ancestor got its mitochondria is called the serial endosymbiosis theory. This is the notion that a primitive ancestral cell engulfed bacteria and, instead of digesting them, drafted them into service as energy factories. Somewhere along the way, these evolving cells would have had to move some DNA around between their evolving nuclei and these engulfed energy factories, as actual mitochondria have their own DNA but also depend on genes in a cell’s nucleus. There are a lot of other problems with this story, and the more that scientists learn about the inner workings of cells, the more implausible this story becomes. (Read more about it in “Winding Back Life’s Story: Evolution of Mitochondria.”)
So far, no one is suggesting that Monocercomonoides is anything like that supposed primitive prokaryotic ancestor because this protist is clearly a nucleated cell. (It not only has a nucleus but also another membrane-bound organelle, the Golgi body.) In any case, evolutionists believe that the evolution of mitochondria was essential in the evolution of the common eukaryotic ancestor. Therefore, they assume that a eukaryote without mitochondria once had them and later lost them.
And while it is quite possible that Monocercomonoides once had mitochondria and over time lost them, even if it did, that scenario would not be an example fitting the needs of Darwinian evolution. The loss of a cellular organelle is not the same as the acquisition of the information to build something new. Loss of a structure or function at most represents a loss of genetic information, not sufficient evidence to prove that upward evolution of complexity ever occurred.
Borrowed Building Materials? . . .
Could Monocercomonoides of the past have acquired the genetic information to build iron-sulfur clusters from bacteria, as the authors assert? Horizontal gene transfer does occur between some bacteria, often involving the intermediary services of a bacteriophage, a sort of virus that affects bacteria. This is one of the ways in which antibiotic resistance and other traits that may be adaptive in certain environments are passed around in the microbial world. However, horizontal transfer of genes does not explain their origin, only the way they can be spread between bacteria. (Learn more in “Bacteria’s Unique Design—Pooling Resources” and “How Are New Genes Made?”)
Whether such a transfer of genetic material can occur between bacteria and protists has yet to be documented.16 In the case of Monocercomonoides, such a genetic acquisition has not been demonstrated, despite its discoverers’ assumption that it must have occurred. They assume such a genetic infusion made the loss of mitochondria possible, yet they can only assume the protist once had mitochondria. Furthermore, they cite phylogenetic analysis in support of their theory, yet phylogenetic analysis is only a comparison of the characteristics of many organisms, grouping them according to similarity and assuming that similarities map an evolutionary history. Their phylogenetic analysis assumes an unobserved history of evolutionary relationships on the basis of observable similarities.
Similar genes are not evidence that unobserved horizontal gene transfer occurred somewhere back in deep time and neither is “phylogenetic analysis.”
It is the similarity of the genes for encoding the iron-sulfur-cluster-building machinery in Monocercomonoides to those in prokaryotes that forms the basis of this phylogenetic analysis. Prokaryotes initiate iron-sulfur cluster construction with a multi-step, sulfur-mobilization (SUF) pathway. Although the enzymes are not identical, Monocercomonoides also uses a sulfur-mobilization pathway. (Monocercomonoides has a unique, fused form of two of the enzymes.)17 The scientists believe the genes to build a SUF system had to have come from prokaryotes. The anaerobic protists Pygsuia biforma and Blastocystis have mitochondrion-related organelles on which similar enzyme systems are active.18 Discoverers of each of these also believe that the protist acquired the genes to make these enzymes from bacteria or archaea because the protist’s genes contain some regions similar to those on homologous prokaryotic genes. However, similar genes are not evidence that unobserved horizontal gene transfer occurred somewhere back in deep time and neither is “phylogenetic analysis.”
Evolution. . . Or Original Design?
Monocercomonoides might have once had mitochondria.19 And it might have received and modified bacterial genes through transfer mechanisms not yet elucidated.20 However, the evolutionary scientists’ conclusion that this must have happened is a product of their evolutionary presuppositions. Furthermore, even if Monocercomonoides started out its existence with mitochondria, received genes for iron-sulfur cluster synthesis from bacteria, and eventually lost its mitochondria, then it only lost genetic information of one sort and borrowed genetic information of another sort. It never evolved any new information. And it remained a protist rather than evolving into some higher sort of organism. Nothing in this scenario—which is itself hypothetical—is an example of molecules-to-man evolution.
Monocercomonoides is well suited for life inside a mammalian intestine. Vladimir Hampl, the senior investigator, says, “It is very likely that the mitochondrion is absent in the whole group called oxymonads.”21 He expects to find other protists in its group without mitochondria.
With their unusual ways to power their lives and build the components needed for survival, are these protists showing us how evolution has worked things out? Not at all. Considering how perfectly equipped these organisms are for life without mitochondria, it actually makes sense to see them as the recipients of a great set of designs that equip them for their little corner of the biological world. Perhaps God in His wisdom designed them to live as they do.22
Think about it. They lack the mitochondria needed to perform highly efficient, oxygen-dependent energy metabolism. But they live in a low-oxygen environment. They utilize glycolysis, the same non-oxygen-requiring, energy-generating biochemical pathway found in the cytoplasm of all cells, to metabolize glucose. They don’t waste the products of glycolysis but contain the necessary enzymes to fully metabolize them, obtaining some energy from them, though not the amount mitochondria extract. But these protists make up for that. They get a huge energy boost from an enzymatic pathway that metabolizes a common amino acid (arginine). Monocercomonoides “contains a complete set of three genes for enzymes”23 used in this arginine deiminase pathway. The energy requirements of this protist are fully met by two complete sets of complex enzymes in a way suited to its low oxygen home.
Designs demand a designer, and the written record of our origins, God’s Word, lets us know that the Designer is the Creator God of the Bible.
As to their need for iron-sulfur clusters, Monocercomonoides meets this need using not one but four proteins with biochemical structures like those used by bacteria, archaea, and plastids during the first few steps of iron-sulfur cluster synthesis.24 However the genes that encode these proteins are structurally the sort of genes found in eukaryotes. And similar iron-sulfur assembly systems have been found in two other eukaryotes that get along with abbreviated sorts of mitochondria-like organelles.25 These four proteins lack any sort of markers suggesting they ever were part of a mitochondrial system.26 Thus they appear most consistent with an original design for cytoplasmic, iron-sulfur cluster production in a non-mitochondrial protist. Designs demand a designer, and the written record of our origins, God’s Word, lets us know that the Designer is the Creator God of the Bible.
The Limits of Evolution and Biology
The claim that this protist has “evolved beyond the known limits that biologists circumscribed” is ironic. Molecules-to-man evolution—by supposing that life can emerge from non-living raw materials through random chance processes—presumes that which goes beyond anything observational science supports. Furthermore, by supposing that mutations can provide the genetic information to evolve into different, more complex kinds of organisms, molecules-to-man evolution presumes the rampant occurrence of something biologists have never observed. The notion that all life shares a single ancestor is a tale told without the benefit of experimental mechanisms to show that such a scenario could happen. The popular endosymbiosis theory to explain the origin of all eukaryotes likewise fails to stand up to scrutiny.
Evolutionary biologists choose to draw the line on the limits of evolution not on the basis of observational science, but rather according to whatever suits their evolutionary presumptions. This protist is an observable surprise that will require textbooks to be updated, but it isn’t the protist that pushes the limits of what is actually known in biology: it is the whole story of molecules-to-man evolution!
Further Reading
- Seeing the Master Designer Through a Microbe’s Eye
- Winding Back Life’s Story: Evolution of Mitochondria
For More Information: Get Answers
Remember, if you see a news story that might merit some attention, let us know about it! (Note: if the story originates from the Associated Press, FOX News, MSNBC, the New York Times, or another major national media outlet, we will most likely have already heard about it.) And thanks to all of our readers who have submitted great news tips to us. If you didn’t catch all the latest News to Know, why not take a look to see what you’ve missed?
(Please note that links will take you directly to the source. Answers in Genesis is not responsible for content on the websites to which we refer. For more information, please see our Privacy Policy.)
Footnotes
- Cell Press, “Change the Textbooks: This Eukaryote Completely Lacks Mitochondria,” Science News, May 12, 2016, https://www.sciencedaily.com/releases/2016/05/160512124908.htm.
- Ibid.
- Nell Greenfieldboyce, “Look, Ma! No Mitochondreia,” NPR, May 12, 2016, http://www.npr.org/sections/health-shots/2016/05/12/477691018/look-ma-no-mitochondria.
- Cell Press, “Change the Textbooks: This Eukaryote Completely Lacks Mitochondria.”
- Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” Current Biology 26, no. 10 (May 2016) 26(10): 1275, doi:10.1016/j.cub.2016.03.053.
- Ibid., 1278.
- Cell Press, “Change the Textbooks: This Eukaryote Completely Lacks Mitochondria.”
- Glucose is metabolized anaerobically through glycolysis, and additional enzymes ferment the resulting breakdown products to obtain energy. These energy pathways are also present in the cytoplasm of cells with mitochondria. But this doesn’t match the energy that could be obtained using mitochondria in an oxygenated environment. However, in Monocercomonoides and in protists with diminished mitochondria, an abundance of energy is available from the metabolism of the amino acid arginine. Arginine is degraded in these protists by a series of three enzymes, collecting the energy from each chemical bond in turn. This arginine breakdown pathway produces eight times more energy than glucose metabolism. From Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” 1278.
- Robert Lill, “Function and Biogenesis of Iron-Sulphur Proteins,” Nature 460 (August 2009): 831–838, doi:10.1038/nature08301.
- Iron-sulfur clusters, vital for the function of many sorts of cellular operations, are synthesized and placed in their proper locations in a series of steps, each mediated by a particular sort of protein. Thus a mutation impairing any one of these proteins can be very detrimental. Such mutations in humans cause Friedreich’s ataxia, iron-sulfur cluster deficiency myopathy, a particular form of sideroblastic anemia, mitochondrial encephalopathy, and multiple mitochondrial dysfunctions syndrome. Learn more in Tracey A. Rouault “Biogenesis of Iron-Sulfur Clusters in Mammalian Cells: New Insight and Relevance to Human Disease,” Disease Models and Mechanisms 5 (2012): 155–164, doi:10.1242/dmm.009019.
- Tracey A. Rouault, “Mammalian Iron-Sulphur Proteins: Novel Insights Into Biogenesis And Function,” Nature Reviews: Molecular Cell Biology 16 (January 2015): 45, doi:10.1038/nrm3909.
- A protein’s primary structure is the amino acid sequence, shown by the green chain in this model. Interactions between adjacent strands of this chain cause the protein to fold into secondary and then tertiary structures. Secondary structures, shown by red and yellow ribbons in this model, are portions of the molecule held together by hydrogen bonds. The red ribbons are coiled and called alpha helices, while the yellow ribbons are sheet-like and called beta-sheets. The tertiary structure is a further folding of the entire molecule, which creates reactive regions and exposes the “business end” of the molecule to interact as needed with surrounding molecules. Therefore, the protein’s activity often depends on its tertiary structure. In viperin, an iron-sulfur cluster bonded to the cysteine amino acids around a central pocket holds its correct tertiary structure. These crucial cysteines are shown in magenta, and the iron-sulfur cluster, which is not shown in the image, fits into the pocket they create.
- Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” 1278.
- Cell Press, “Change the Textbooks: This Eukaryote Completely Lacks Mitochondria.”
- Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” 1274.
- The evidence that horizontal genetic transfer from prokaryotes to protists has occurred consists of the genetic similarities of homologous genes. In the case of another anaerobic parasitic protist, Entamoeba histolytica, similarities of other mitochondrial-associated genes for synthesizing iron-sulfur clusters were noted in 2004. The scientists who discovered these similarities assumed that horizontal gene transfer had occurred and was evidence for the presumed evolution of mitochondria through serial endosymbiosis. However, as discussed in “Winding Back Life’s Story: Evolution of Mitochondria,” there are a lot of insurmountable problems with this evolutionary belief. The presence of similar genes in different kinds of organisms is not evidence of evolution but is consistent with a history of a common Designer. (Entamoeba histolytica discussed in Mark van der Giezen, Siân Cox, and Jorge Tovar, “The Iron-Sulfur Cluster Assembly Genes iscS and iscU of Entamoeba histolytica Were Acquired by Horizontal Gene Transfer,” BMC Evolutionary Biology 4, no. 7 [February 2004]: doi:10.1186/1471-2148-4-7).
- Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” 1278.
- See Courtney Stairs et al., “A SUF Fe-S Cluster Biogenesis System in the Mitochondrion-Related Organelles of the Anaerobic Protist Pygsuia,” Current Biology 24, no. 11 (June 2014): 1176–1186, doi:10.1016/j.cub.2014.04.033 and Anastasios D. Tsaousis et al., “Evolution of Fe/S Cluster Biogenesis in the Anaerobic Parasite Blastocystis,” Proceedings of the National Academy of Sciences 109, no. 26 (June 2012): 10426–10431, doi:10.1073/pnas.11160671069.
- Because other protists that live in low-oxygen conditions have only abbreviated forms of mitochondria—called mitochondrion-related organelles (MROs)—and synthesize iron-sulfur clusters on them, it is not unreasonable to think that these protists might have once had MROs and lost them. But of course this would represent only loss of information within a particular kind of organism, not molecules-to-man evolution.
- Just as God may have designed these protists to be equipped as they are, so He may have designed eukaryotic microbes with a way to vary by sharing and shuffling genes in a way analogous to bacteria.
- Cell Press, “Change the Textbooks: This Eukaryote Completely Lacks Mitochondria.”
-
Could the acquisition of genes allowing these anaerobic protists to survive without mitochondria be associated with their ability to cause disease in this sin-cursed world? Probably not. While many of these anaerobic protists with abbreviated forms of mitochondria are parasites, there is no evidence that their lack of bona fide mitochondria or the availability of an alternate, iron-sulfur cluster synthetic pathway distinguishes disease-causing organisms from those varieties that remain harmless. In some cases, there are several varieties of the organism, and not all are pathogenic. In fact, though quite at home in a chinchilla’s bowels, Monocercomonoides is not a pathogen.
Thus it is reasonable to consider the unusual traits of these protists as possible designs compatible with God’s creation of a very good world. Read more about the development of pathogenic microbes in the wake of sin’s curse in “The Genesis of Malaria,” “The Genesis of Pathogenic E. coli,” and “The Role of Genomic Islands, Mutation, and Displacement in the Origin of Bacterial Pathogenicity.”
- Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” 1278.
- Ibid.
- The anaerobic protists Pygsuia biforma and Blastocystis have mitochondrion-related organelles on which similar enzyme systems are active. Discoverers of each believe that the protists acquired the genes to make these enzymes from bacteria or archaea because their genes have some regions similar to those on homologous genes in those organisms. (See Courtney Stairs et al., “A SUF Fe-S Cluster Biogenesis System in the Mitochondrion-Related Organelles of the Anaerobic Protist Pygsuia,” and Anastasios D. Tsaousis et al., “Evolution of Fe/S Cluster Biogenesis in the Anaerobi Parasite Blastocystis.”)
- Anna Karnkowska et al., “A Eukaryote Without a Mitochondrial Organelle,” 1280–1281.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis