Winding Back Life’s Story: Evolution of Mitochondria
News to Know
Abstract
When mitochondria evolved: another just-so story about cellular origins.
News Sources
Among the great, unsolved evolutionary mysteries, the origin of eukaryotic cells—cells with nuclei—ranks high. Nucleated cells are the building blocks of all multicellular organisms, including us. And they are powered by mitochondria.1 Mitochondria use oxygen and a series of enzymes to extract the maximum possible energy from sugar and to package it in a usable form. Where did cells get these energy factories?
Endosymbiosis Theory
The most popular evolutionary story to explain how the last eukaryotic common ancestor (LECA) got mitochondria is that it ate them. Or rather that it ate small non-nucleated cells (prokaryotes, such as bacteria)—and that then those prokaryotes developed a symbiotic relationship with the host cell, supplied it with energy, and turned into mitochondria. That story, popularized in the 1970s by the late Dr. Lynn Margulis, is called the “serial endosymbiosis theory.” Because single-celled organisms and other cells like our white blood cells engulf debris and smaller microorganisms—an observable process called endosymbiosis—the serial endosymbiosis theory for the origin of eukaryotic cells seemed reasonable to evolutionists. After all, they reasoned, mitochondria and prokaryotes have some superficial similarities. Both are small. And while most of a eukaryotic cell’s DNA is in its nucleus, mitochondria have some DNA of their own as well as ribosomes to translate its genes into proteins. Nuclear DNA is in the form of double helical strands, but mitochondrial DNA is not. Though the mitochondrial genome is much smaller than a bacterial genome, both mitochondria and prokaryotes have circular DNA.
Because multicellular organisms are made of eukaryotic cells, many think the evolution of mitochondria was the stepping-stone that fueled multicellular evolution. However, there are a lot of problems with the story of mitochondrial evolution. Therefore, exactly when mitochondria evolved, the fate of the proto-mitochondria’s missing genes, and the identity of their ancestral bacteria have remained controversial.
Ancestry Anonymous
Because genomic analysis fails to point to a single sort of ancestral bacteria—as confirmed by a group of French scientists in 2014—evolutionists think eukaryotes branched from their common ancestor very early and diversified quickly, obscuring the LECA’s identity. Unsure then how to explain the ubiquitous presence of mitochondria in eukaryotes, the French team suggested that mitochondria either evolved very early—before eukaryotes evolved or diversified—or very often.2 The research team of Alexandros Pittis and Dr. Toni Gabaldón announced in February 2016 in Nature that they have solved this mystery. They examined mitochondrial genomes from lots of living organisms and concluded that mitochondria evolved fairly late in the cell’s evolutionary history.
Pittis and Gabaldón compared the genomes of many bacteria thought to resemble the ancestral cell (LECA), identifying how much their genes for common proteins and structures differ.3 They believe bacteria inherited their oldest genes from a common ancestor. And they believe that genes differing the most are the oldest, having had longer to evolve their differences. Genes that are more similar, they believe, were acquired by the ancestral cells sometime later. The genes that differ most among different sorts of bacteria, they found, are those that code for proteins used by diverse parts of complex cells—organelles other than mitochondria and the nucleus—and for proteins unrelated to energy production.4 Genes related to the mitochondria, the nucleus, and proteins unrelated to energy production tended to vary less across different families of prokaryotes. Therefore, they conclude that the ancestral cell was quite complex, having cobbled together genetic components from many sorts of bacteria, before finally adopting genes from proto-mitochondria into its genome. The LECA, they maintain, was well along its evolutionary pathway before it acquired power-producing mitochondria and thus the energy to evolve into all of us.
“Our work demonstrates that the acquisition of mitochondria occurred late in cell evolution, and that the host cell already had a certain degree of complexity. Our study makes it possible to break down the steps of what is considered the greatest evolutionary leap after the origin of life,” says Pittis.5 “Like archeologists,” Gabaldón explains, “we are trying to reconstruct something that existed in the past based on the evidence we have today. Specifically, we’ve tracked down proteins common to all complex organisms, and reconstructed their evolution. We found that the proteins related with mitochondria acquisition arrived later than those related with other parts of the cell.”6
In reality, however, this study has not shown when a hypothetical ancestral cell acquired mitochondria, or that such an event ever occurred. Neither does anything about their observations indicate different kinds of organisms evolved from a common ancestor. Their comparisons only show that, in the bacteria examined, the genes associated with mitochondria and energy production vary less than the genes coding for other cellular components. The authors’ determination to translate the differences between genes from different kinds of organisms into a molecular clock timeline is merely an unverifiable evolutionary presupposition superimposed upon the genetic data.
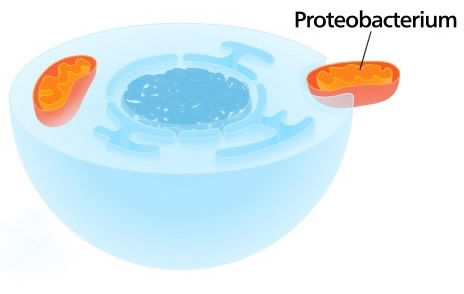
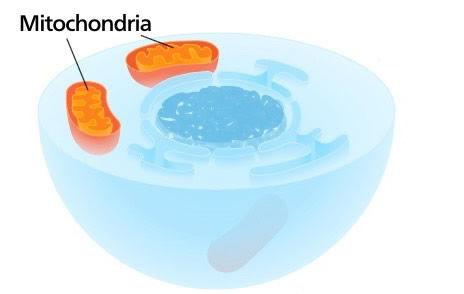
These cross-sectional illustrations of a cell show how many evolutionists think mitochondria, which are only found in nucleated cells, evolved. They think that an early cell—either before or after it wrapped its DNA in a membrane to form a nucleus (shown on the left here, with its nucleus in blue)—engulfed proteobacteria. Those organisms supposedly evolved into mitochondria, the cell’s energy-generating powerhouses (shown in orange in the illustration on the right). Faith in this theory persists among evolutionists despite its many inconsistencies. Images from Wikipedia.
Cellular Archaeology
Gabaldón’s comparison of his work to archeology is telling. Archaeologists have to build a story about the past around the artifacts they find in the present. And that can be especially challenging when contemporaneous written accounts of the artifacts’ historical context are unavailable. Then the story can vary considerably depending on the archaeologists’ historical presuppositions. But when an archaeologist finds written parchments or tablets with the eyewitness accounts of events in the past, he or she should be able to come up with a more accurate story. (This is well illustrated by the conundrum of Egyptian chronology. Early Egyptologists developed a chronology on the basis of limited data and their own presuppositions. Traditional Egyptian dating was later pronounced a confusing and inaccurate mess after written records from other ancient nations were found to contradict the story they had constructed. Read about it in “Doesn’t Egyptian Chronology Prove That the Bible Is Unreliable?,” and learn how this drama plays out in the search for the historical Exodus, documented in the recent book and film documentary Patterns of Evidence: The Exodus.)
These evolutionary authors want to know about life’s origins. But when they perform laboratory comparisons in the present and simply assume that whatever homologous genes differ the most must have evolved earliest in a common ancestor, they are making conjectures in a vacuum that ignores our Creator’s historical eyewitness account. God’s Word, recorded in the Bible, lets us know that life did not evolve, but that the Creator spoke all the different kinds of living things into existence about 6,000 years ago. From the Bible we understand and that all the varieties of life we see today are variations within the created kinds of things He made to reproduce “after their kinds.” And what we observe through the eyes of science affirms what we read in God’s historical account.
The Tale of the Missing Genes
Ascribing significance of their evolutionary conclusions, Pittis claims, “Understanding how complexity originated and evolved is important to better understand the mechanisms that govern cells, and by extension, the functioning of all living organisms.”7 Indeed, a great deal has been learned in recent years about cellular components like mitochondria and ribosomes and the mechanisms that regulate them. Those discoveries are being made without any need to know about their unobservable—and fictional—evolutionary origin. In fact, the more scientists observe, the more untenable evolutionary claims—like the just-so story of serial endosymbiosis—become.
Have scientists, as media reports consistently claim, really shown that the mitochondria we find in our cells are just stripped-down bacteria? Not at all! For one thing, the genome of a mitochondrion is much smaller than a bacterial genome, and many genes essential for mitochondrial function and reproduction are found in their encompassing cells’ nuclei. To account for this discrepancy, evolutionists long ago embellished their story with the explanation that the proto-mitochondrion entrusted most of its genes to its host cell’s nucleus. This sounded promising at one level because horizontal gene transfer does occur between plant nuclei and chloroplasts—an energy-capturing cellular organelle. However, the need to produce an evolutionarily superior cell better equipped to survive than its predecessor while dumping the majority of the eaten-but-not-consumed bacteria’s genes into the host cell’s nucleus and transforming them into functional components of the host DNA would be irreducibly complex. Fitness of cells in transition would be compromised. Until all the components of this new system were in place, the new mitochondria could not function.
The Ribosomal Story
Nevertheless, evolutionists have long held that the similarity of mitochondrial and bacterial ribosomes provides strong support for their story of mitochondrial evolution.8 Mitochondria have their own ribosomes, just as they have some DNA. Ribosomes build proteins. They transform the information in genes into the proteins they designate. Bacterial ribosomes are smaller than those in the cytoplasm of eukaryotic cells. And mitochondrial ribosomes seemed closer in size to prokaryotic ribosomes.
Evolutionists have long held that the similarity of mitochondrial and bacterial ribosomes provides strong support for their story of mitochondrial evolution.
Modern research has revealed, however, that the internal structure and density of mitochondrial and bacterial ribosomes differ greatly. The size of structures like ribosomes is measured as a “sedimentation coefficient,” abbreviated S. This measurement is obtained by observing how these cellular components settle when spun in a centrifuge. Not surprisingly, the sedimentation coefficient is affected by many factors, not only the size but also the density and other characteristics of the particular molecules comprising the ribosomes. Therefore, the sedimentation coefficient has not told the whole story.
Prokaryotes have 70S ribosomes, and the ribosomes in the cytoplasm of mammalian eukaryotic cells are 80S.9 The mammalian mitochondrial ribosome has an even lower sedimentation coefficient, 55S, but rather than being smaller, the mitochondrial ribosome is actually larger and heavier. And while there are some similar components—not a surprise given the fact that the process of translating genetic information into protein structure occurs in all living things—there are dramatic structural and functional differences at the molecular level. (Learn more about these distinctly different creations and how they work in “A Review of Mitoribosome Structure and Function Does not Support the Serial Endosymbiotic Theory.”) It is not reasonable to think that even over billions of years the ribosomal derangements and loss of function caused by mutations could somehow add up to produce the very different and irreducibly complex intracellular structure of eukaryotic cells.
Leaky Sheds and Broken Cisterns
Furthermore, if the proto-mitochondrial endosymbiont gave most of its genes into the keeping of its host cell’s nucleus, why not all? Another research team, Iain Johnston and Ben Williams, reported their investigation of this conundrum in the February 24, 2016, issue of Cell Systems. “The hypotheses underlying potential reasons for mitochondria to keep their own genes have been debated for decades,” Johnston says.10 Like Pittis and Gabaldón, evolutionists Johnston and Williams looked for answers about ancient unobservable origins in the genomes of present-day mitochondria, hoping to retrace the steps that created them back through deep evolutionary time. “This is the first data-driven approach to address this question,” says Johnston. “It’s facilitated by the fact that there are thousands of mitochondrial genomes from across a very wide diverse set of taxa available so now we can harness the data and let it speak for itself.”11
Why mitochondrial genomes are so much smaller than the genomes of their ancestral bacteria? Williams says, “It’s not that the ‘lost’ genes no longer exist in many cases, it’s that the nucleus produces the proteins and the proteins go into the mitochondria, but why bother having anything in the mitochondria when you could have it all in the nucleus? It’s like saying you have a central library with all your books in it, but we’re going to keep 10 of them off site in a leaky shed.”12
Williams compares the mitochondrion to a leaky shed because the energy-producing processes constantly going on there produce a lot of free radicals, by-products that tend to cause mutations and cellular damage. Why would billions of years of evolution leave something as important as the genetic blueprints for components essential for an organisms’ mitochondrial function in such an environment? For that matter, casting aside the evolutionary fairy tale, why would our wise Creator leave the DNA for building critical mitochondrial components in the one place it would seem most vulnerable to damage?
Most genes in the mitochondrial genome, Johnston and Williams found, are used for building mitochondrial ribosomes. As we said above, the protein-building ribosome is the great link between the genes in DNA and the proteins for which they carry the blueprints. The proteins manufactured in the mitochondria are predominantly those in the electron transport chain—a series of enzymes at the heart of a mitochondrion’s energy-producing function. Thus, each of the many mitochondria in a cell has local control of its energy production because it has the ribosomal genes to do so, rather than having to rely on central control from the nucleus. This would seem to be a great advantage. Johnston and Williams also found that mitochondrial genes have a composition that seems unusually resistant to damage.13
Does the data speak for itself?
Does the data speak for itself? Nothing about this data supports evolution; the evolutionary conclusions spring from the presuppositions of the researchers. Just as it turns out that the mitochondria are not really leaky sheds, so the evolutionary story of how mitochondria evolved—though propped up by claims about the complexity of the cells that adopted them and the remarkable design that evolved again and again—is like a broken cistern that can hold no water.14 What we see in their discoveries is not how mitochondria evolved. Instead, we gain greater insight into the irreducible complexity that shouts against the possibility of their ever having evolved, much less of having evolved over and over again. We have here a glimpse into the wonderful design of mitochondria by a wise Creator.
What researchers are learning about how mitochondria are designed—not about how they supposedly evolved—may ultimately help medical researchers discover ways to help people with mitochondrial diseases. The cellular processes that keep us alive—and that go so very wrong sometimes in this sin-cursed world—were created by God in the fully functional “very good” (Genesis 1:31) world in the beginning, about 6,000 years ago, without evolution. God created all things good, but in the wake of man’s rebellion against Him, sickness and death entered our world. Yet on balance, most of the systems God designed mercifully still work quite well. The more we learn about cellular structures like mitochondria, the more we should be able to see the wisdom and creativity of our Creator.
Further Reading
- Does “Loki” Show How Humans Evolved from Single-Celled Organisms?
- Seeing the Master Designer Through a Microbe’s Eye
- “Non-Evolution” of the Appearance of Mitochondria and Plastids in Eukaryotes: Challenges to Endosymbiotic Theory
- Just Endosymbiosis
- A Review of Mitoribosome Structure and Function Does not Support the Serial Endosymbiotic Theory
For More Information: Get Answers
Remember, if you see a news story that might merit some attention, let us know about it! (Note: if the story originates from the Associated Press, FOX News, MSNBC, the New York Times, or another major national media outlet, we will most likely have already heard about it.) And thanks to all of our readers who have submitted great news tips to us. If you didn’t catch all the latest News to Know, why not take a look to see what you’ve missed?
(Please note that links will take you directly to the source. Answers in Genesis is not responsible for content on the websites to which we refer. For more information, please see our Privacy Policy.)
Footnotes
- Though some unusual eukaryotic cells without mitochondria have been discovered, those cells contain organelles that appear to be degenerate forms of mitochondria. Thus it is reasonable to say that all eukaryotic cells have mitochondria or some degenerate form of them.
- N. Rochette et al., “Phylogenomic Test of the Hypotheses for the Evolutionary Origin of Eukaryotes,” Molecular Biology and Evolution 31, (2014): 832–845, doi:10.1093/molbev/mst272.
- Different kinds of organisms, and different kinds of cells too for that matter, have in common a lot of genes coding for essential structures and proteins. This is no surprise, given that many biochemical and cellular processes are essential to virtually all forms of life. Often those common genes differ somewhat in their nucleotide sequences. Evolutionists assume that these variant forms developed over time after divergence from a common ancestor. But homologous genes are not evidence that all cells and all kinds of life share a common ancestor. Neither are their differences a measure of how long ago they evolved from this hypothetical ancestor. Instead, they point to the common solutions for common needs designed by the common Designer of all life, the Creator God, who has given us the reliable eyewitness account of life’s history in His Word, the Bible.
- Alexandros Pittis and Toni Gabaldón, “Late Acquisition of Mitochondria by a Host With Chimaeric Prokaryotic Ancestry,” Nature 531 (2016): 101–104, doi:10.1038/nature16941.
- Center for Genomic Regulartion, “Team Sheds Lights on a Crucial Moment in the Evolution of Life: When Cells Acquired Mitochondria,” Phys.org, February 3, 2016, http://phys.org/news/2016-02-team-crucial-moment-evolution-life.html.
- Ibid.
- Ibid.
- To learn more about how ribosomes work, see the study guide we developed in response to the claims made in Cosmos: A SpaceTime Odyssey.
- A ribosome is made of two major components that come together in order to synthesize proteins. The prokaryote 70S ribosome, for instance, is made of a smaller 30S subunit and a larger 50S subunit.
- Cell Press, “Why Do We Still Have Mitochondrial DNA?,” Phys.org, February 18, 2016, http://phys.org/news/2016-02-mitochondrial-dna.html.
- Ibid.
- Ibid.
- The researchers found that the mitochondrial DNA in each species they examined, when compared with its nuclear DNA, is composed of an unusually high percentage of those nucleic acids that chemically seem most resistant to oxidative damage. From Iain G. Johnston and Ben P. Williams, “Evolutionary Inference Across Eukaryotes identifies Specific pressures Favoring Mitochondrial Gene Retention,” Cell Systems 2 (February 24, 2016): 101–111, http://dx.doi.org/10.1016/j.cels.2016.01.013.
- This apt analogy is drawn from Jeremiah 2:12–13: “Be appalled, O heavens, at this; be shocked, be utterly desolate, declares the Lord, for my people have committed two evils: they have forsaken me, the fountain of living waters, and hewed out cisterns fore themselves, broken cisterns that can hold no water.”

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis