
1.3 The Origin of Life: DNA and Protein
Evidence of Creation?
The two basic parts of the tumbled pebble and the arrowhead we considered are hard and soft rock. Two basic parts of every living system are DNA and protein.
DNA is the famous molecule of heredity. It’s a focus of crime scene investigations, and we often hear news stories about it. This is the molecule that gets passed down from one generation to the next. Each of us starts off as a tiny little ball about the size of a period on a printed page. In that tiny ball, there are over six feet (2m) of DNA all coiled up. All of our physical characteristics (height, skin color, etc.) are “spelled out” in that DNA.
What are proteins? Proteins are the molecules of structure and function. Hair is mostly protein; skin cells are packed full of proteins; the enzymes that break down food and build it up are proteins; the filaments that slide together to make muscles work are proteins.
So DNA and protein are two basic “parts” of every living system. When you get down to a virus, that’s all you find—DNA and protein. (In some viruses, RNA substitutes for DNA.) The DNA molecules code for the protein molecules that make us what we are. That same principle applies to all life forms: viruses, plants, and animals, as well as human beings.
My students study all of the details,1 but DNA and protein molecules are really quite simple in their basic structure. If you can picture a string of pearls, you can picture DNA: it is a chain of repeating units. Figure 2-A is a diagram of a DNA molecule. The parts that look like railroad boxcars are sugar and phosphate groups, and the parts that stick out from each boxcar in the chain are groups called bases.
Proteins are built in about the same way. Proteins are also chains of repeated units. As shown in Figure 2-B, the links in protein chains are called amino acids. In all living things, inherited chains of DNA bases are used to line up chains of amino acids. These amino acid chains are the protein molecules responsible for structure and function. For example, chains of several hundred DNA bases tell the cell how to make a protein called hemoglobin, and that protein functions as the oxygen carrier in red blood cells. In short form, DNA → protein → trait, and that relationship is the physical basis of all life on earth.
Now, what about that relationship between DNA and protein? How did it get started? Evolutionists picture a time long ago when the earth might have been quite different. They imagine that fragments of DNA and fragments of protein are produced. These molecules are supposed to “do what comes naturally” over vast periods of time. What’s going to happen? Will time, chance, and chemical reactions between DNA and protein automatically produce life?
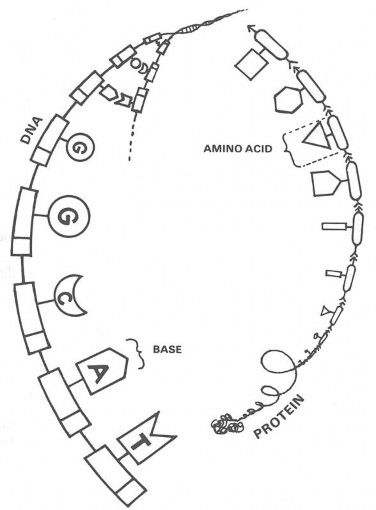
Figure 2-A. DNA is built like a string of pearls, whose links (specifically the bases G, C, A, and T) act like alphabet letters that “spell out” hereditary instructions.
Figure 2-B. Proteins are chains of amino acids. Each chain coils into a special shape that has some special function: muscle contraction, digestion, oxygen transport, holding skin together, etc.
At first, you might think so. After all, nothing is more natural than a reaction between acids and bases. Perhaps you’ve used soda (a base) to clean acid from a battery. The fizz is an acid-base reaction. So is using “Tums” to neutralize stomach acid. Nothing is more common than reactions between acids and bases. If you just wait long enough, acid-base reactions will get DNA and protein working together, and life will appear—right? Wrong! Just the opposite.
The problem is that the properties of bases and acids produce the wrong relationship for living systems. Acid-base reactions would “scramble up” DNA and protein units in all sorts of “deadly” combinations. These reactions would prevent, not promote, the use of DNA to code protein production. Since use of DNA to code protein production is the basis of all life on earth, these acid-base reactions would prevent, not promote, the evolution of life by chemical processes based on the inherent properties of matter.
These wrong reactions have produced serious problems for Stanley Miller, Sidney Fox, and other scientists trying to do experiments to support chemical evolution. Almost all biology books have a picture of Miller’s famous spark chamber (Figure 3). In it, Miller used simple raw materials and electric sparks to produce amino acids and other simple molecules—the so-called “building blocks of life.” Some newspapers reported that Miller had practically made “life in a test tube.”
Miller’s experiment was brilliant, and I loved to tell my students about it. Then I came to see there were just three little problems: he had the wrong starting materials, used the wrong conditions, and got the wrong results.
What do I mean by “wrong starting materials”? Miller left out oxygen. Why? Because of the scientific evidence? No. He left it out because he knew oxygen would destroy the very molecules he was trying to produce. It’s hard for us to realize how “corrosive” oxygen is, since most modern living things depend on it. But oxygen is so valuable to life precisely because it’s so chemically reactive, and aerobic living things today have systems to protect themselves against the harmful effects of oxygen, while using its chemical power to their advantage. (Anaerobic organisms and some viruses are quickly destroyed by contact with oxygen.)
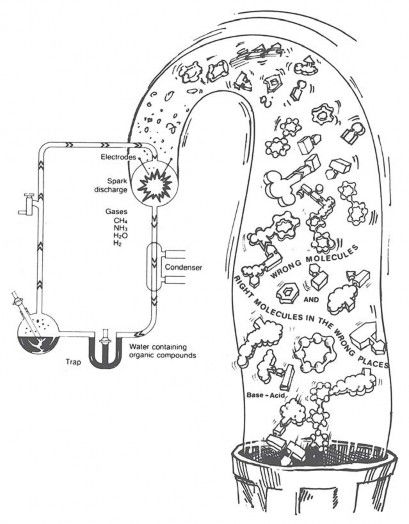
Figure 3. Left to time, chance, and their chemical properties, the bases of DNA and amino acids of proteins would react in ways that would prevent, not promote, the evolution of life. In the same way, reactions among molecules in Miller’s famous “spark chamber” would destroy any hope of producing life. Living systems must constantly repair the chemical damage done to them, and when biological order loses out to inherent chemical processes, death results—even though a dead body has all the right molecules in the right places in the right amounts at the right times (almost!).
A.I. Oparin, the Russian biochemist who “fathered” modern views of spontaneous generation or chemical evolution, knew oxygen in the atmosphere would prevent evolution. He also “knew,” by faith in Engels’ materialistic philosophy (the view that matter is the only reality), that creation was impossible (there was no spiritual dimension). As an act of faith, then, Oparin believed evolution must have occurred, and as a concession to his faith, he left oxygen out. Science has not been kind to that belief. We find oxidized rocks, suggesting an oxygen atmosphere, as deep as we can dig.
Furthermore, methane (CH4) and ammonia (NH3), two prime gases in the Miller spark chamber, could not have been present in large amounts. The ammonia would be dissolved in the oceans, and the methane should be found stuck to ancient (deep) sedimentary clays. It’s not there! Those who still believe in chemical evolution are aware of these problems (as is Miller himself), so they are simply trying (as yet unsuccessfully) to simulate the origin of life using different starting materials. (Carbon monoxide and hydrogen cyanide are two popular, if unlikely, gases being used today.)
Wrong conditions? Miller used an electric spark to get the gas molecules to combine, and that works. Problem: The same electric spark that puts amino acids together also tears them apart, and it’s much better at destroying them than making them, meaning that few, if any, amino acids would actually accumulate in the spark chamber. Miller, a good biochemist, knew that, of course, so he used a common chemist’s trick. He drew the products out of the spark chamber and into a “trap” that would save the amino acids from destruction by the same electric spark that made them. Using product removal (the principle of LeChatelier or law of mass action) to increase yield is ordinary chemical practice, but it depends on intervention by informed intelligence. Miller was supposed to be demonstrating that the gases could make the “building blocks of life” all by themselves without any outside help, yet his outside, intelligent help was necessary to save the molecules from their destructive chemical fate. (Moreover, creating life in a test tube as a consequence of intelligent design would offer more support to creation than to evolution.)
Wrong results? How could that be? Miller wanted to make amino acids, and he got amino acids (along with sugars and a few other things). How could those results be wrong?
The proteins in living cells are made of just certain kinds of amino acids: those that are “alpha” (short) and “left-handed.” Miller’s “primordial soup” contained many long (beta, gamma, delta) amino acids and equal numbers of both right- and left-handed forms. Problem: Just one long or right-handed amino acid inserted into a chain of short, left-handed amino acids would prevent the coiling and folding necessary for proper protein function. What Miller actually produced was a seething brew of potent poisons that would absolutely destroy any hope for the chemical evolution of life.
The “left-handed amino acid problem” is particularly well-known to evolutionists, and several have been trying to solve it. One brilliant researcher, after working unsuccessfully for years on the problem, just smiled and chuckled when asked about it: “Perhaps God is left-handed.” He may have been closer to the truth than he realized. From what we know about the chemistry of the molecules involved, it really looks like the molecules could never put themselves together into living cells apart from the careful selection, engineering genius, and deliberate design of the transcendent, creative intelligence we call God!2
Chemistry, then, is not our ancestor; it’s our problem. When cells lose their biological order and their molecules start reacting in chemical ways, we die. A dead body contains all the molecules necessary for life and approximately the right amount of each, but we never see a “road kill” get up and walk off because sunlight energy shining on the carcass made all the molecules of life start working together again. What’s lost at death are balance and biological order that otherwise use food to put us together faster than chemistry tears us apart! (See Bliss and Parker3; Illustra Media4; and Thaxton, Bradley, and Olsen5 for details.)
Time and chance are no help to the evolutionist either, since time and chance can only act on inherent chemical properties. Trying to throw “life” on a roll of molecular dice is like trying to throw a “13” on a pair of gaming dice. It just won’t work. The possibility is not there, so the probability is just plain zero.
The relationship between DNA and protein required for life is one that no chemist would ever suspect. It’s using a series of bases (actually taken three at a time) to line up a series of R-groups (Figure 4). R-groups are the parts of each amino acid that “stick out” along the protein chain. “R” stands for the “variable radical,” and variable it is! An R-group can be acid; it can be a base; it can be a single hydrogen atom, a short chain, a long chain, a single ring, a double ring, fat-soluble, or water-soluble!
The point is this: There is no inherent chemical tendency for a series of bases (three at a time) to line up a series of R-groups in the orderly way required for life. The base/R-group relationship has to be imposed on matter; it has no basis within matter.
The relationship between hard and soft rock in the arrowhead in Figure 1 had to be imposed from the outside. All of us could recognize that matter had been shaped and molded according to a design that could not be produced by time, chance, and weathering processes acting on the hard and soft rock involved. In the same way, our knowledge of DNA, protein, and their chemical properties should lead us to infer that life also is the result of plan, purpose, and special acts of creation.
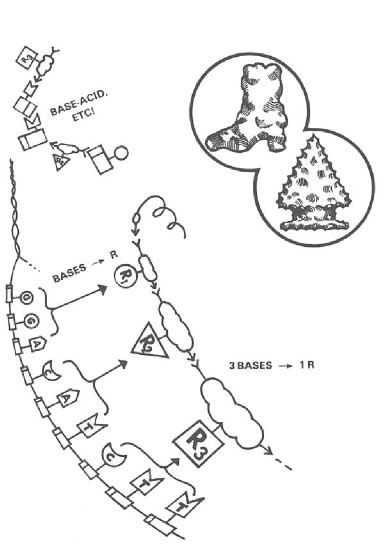
Figure 4. All living cells use groups of three DNA bases as code names for amino acid R-groups. But all known chemical reactions between these molecules (e.g., base-acid) would prevent, not promote, development of this coding relationship. Is the hereditary code, then, the logical result of time, chance, and the inherent properties of matter (like the water-worn pebble), or does it have the irreducible properties of organization (like the arrowhead) that scientists ordinarily associate with plan, purpose, and creative acts?
Let me use a simpler example of the same kind of reasoning. Suppose I asked you this question: Can aluminum fly? Think a moment. Can aluminum fly? I’m sure that sounds like a trick question. By itself, of course, aluminum can’t fly. Aluminum ore in rock just sits there. A volcano may throw it, but it doesn’t fly. If you pour gasoline on it, does that make it fly? If you pour a little rubber on it, that doesn’t make it fly, either. Suppose you take that aluminum, stretch it out in a nice long tube with wings, a tail, and a few other parts. Then it flies; we call it an airplane.
Did you ever wonder what makes an airplane fly? Try a few thought experiments. Take the wings off and study them; they don’t fly. Take the engines off, study them; they don’t fly. Take the pilot out of the cockpit; the pilot doesn’t fly. Don’t dwell on this the next time you’re on an airplane, but an airplane is a collection of non-flying parts! Not a single part of it flies!
What does it take to make an airplane fly? The answer is something every scientist can understand and appreciate, something every scientist can work with and use to frame hypotheses and conduct experiments. What does it take to make an airplane fly? Creative design and organization.
Take a look at the features of a living cell diagrammed in Figure 5. Don’t worry; I am not going to say much about this diagram. Just notice the DNA molecule in the upper left circle and the protein in the lower right. What are all the rest of those strange looking things diagrammed in the cell? Those represent just a few of the molecules that a cell needs to make just one protein according to the instructions of just one DNA molecule. A cell needs over 75 “helper molecules,” all working together in harmony, to make one protein (R-group series) as instructed by one DNA base series. A few of these molecules are RNA (messenger, transfer, and ribosomal RNA); most are highly specific proteins.6
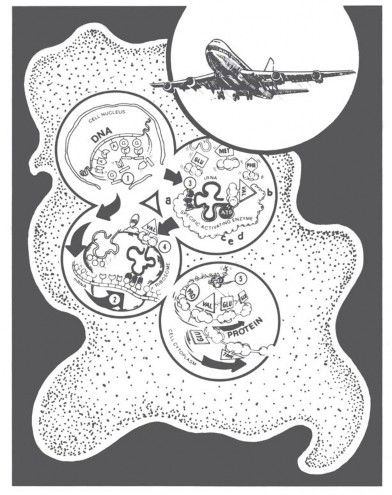
Figure 5. Living cells use over 75 special kinds of protein and RNA molecules to make one protein following DNA’s instructions. What we know about airplanes convinces us that their flight is the result of creative design. What scientists know about the way living cells make protein suggests, just as clearly, that life also is the result of creative design. The real “heroes,” the molecules that establish the non-chemical, grammatical/linguistic coding relationship between triplet base codons and amino acid R-groups are the set of specific activating enzymes I call “translases.” (Drawing from Bliss and Parker, Origin of Life [Green Forest, AR: Master Books, 1979]).
Contrary to popular impression, DNA does not even possess the genetic code for making protein, but only the genetic alphabet. The “alphabet letters” of DNA (the four bases, abbreviated GCAT) are used in groups of three (triplet codons) as code names for the 20 different amino acids of proteins. But bases are equally spaced along DNA; there’s nothing in the structure or chemistry that even hints why or which bases should be grouped as triplet codons. Three letter groupings are not inherent in base sequences; they are imposed on the base series by huge cellular particles called ribosomes.
Ribosomes don’t act directly on DNA, but on expendable “base pair copies” of DNA called messenger RNA, or mRNA. The production of mRNA, and of more DNA for reproduction, is magnificently profound, but it’s a simple consequence of interlocking base shapes and ordinary chemical attraction (mediated by enzymes). The way ribosomes establish the genetic coding system, however, completely transcends the inherent properties of DNA bases.
Ribosomes are “molecular machines” each consisting of about 50 specific proteins and three large RNA molecules. Its overall 3-D shape gives a ribosome two adjacent slots each precisely shaped to hold three and only three bases, thus establishing the triplet coding system. This coding system is not based on time, chance, and the properties of the bases, but on plan, purpose, and intelligent design. In the structure of the ribosome, however, as in the arrowhead, nothing supernatural, complex, or even unusual is involved, and the function of the ribosome is easy to understand and explain. In both the ribosome and the arrowhead, the evidence of creation is not in what we can’t see and don’t know; it’s in the pattern of order (“exherent”) that we do see and can explain: matter shaped and molded to accomplish the purpose of its Creator, not to satisfy inherent chemical properties.
Besides the above, the ribosomes which establish the amino acid code names for making proteins are themselves made of 50 or more specific proteins. It takes specific proteins to establish the code for making specific proteins, so how did the system get started? Evolutionists admit that’s a problem for them because they insist evolution based on time, chance, and the properties of matter is a blind process that can’t plan ahead or work toward a goal. On the other hand, creationists see the goal-oriented function of ribosomes as another evidence of creation. Like batteries can be used to start car engines that then recharge the batteries, so proteins can be used to code for the production of proteins that can then “recharge” the coding proteins.
And there’s more. Even after ribosomes establish triplet codon names for amino acids, the protein building blocks have no chemical way to recognize their code names! All sorts of wrong chemical reactions between amino acids and base triplets are possible, but these would destroy the code. It falls to transfer RNA (tRNA) molecules to pick up amino acids and base pair them with their codons on the ribosome slots. The base pairing of tRNA and mRNA triplets is based on interlocking shapes and ordinary chemical attraction, but the proper pairing of tRNAs with amino acids requires much more than ordinary chemistry.
When it comes to “translating” DNA’s instructions for making proteins, the real “heroes” are the activating enzymes that unite specific tRNA/amino acid pairs. Enzymes are proteins with special slots for selecting and holding other molecules for speedy reaction. As shown in Figure 5 (circle 3), each activating enzyme has five slots: two for chemical coupling (c, d), one for energy (ATP), and, most importantly, two to establish a nonchemical three-base “code name” for each different amino acid R-group (a, b). You may find that awe-inspiring, and so do my cell biology students!
The living cell requires at least 20 of these activating enzymes I call “translases,” one for each of the specific R-group/code name (amino acid/tRNA) pairs. Even so, the whole set of translases (100 specific active sites) would be (1) worthless without ribosomes (50 proteins plus rRNA) to break the base-coded message of heredity into three-letter code names; (2) destructive without a continuously renewed supply of ATP energy to keep the translases from tearing up the pairs they are supposed to form; and (3) vanishing if it weren’t for having translases and other specific proteins to re-make the translase proteins that are continuously and rapidly wearing out because of the destructive effects of time and chance on protein structure!
Most enzymes are proteins that select and speed up chemical reactions that would occur slowly without them. Translases are an entirely different category of enzymes. They impose a relationship that transcends the chemistry of base triplets and amino acids, a code that would not occur at all, slowly or otherwise, in their absence.
Let’s forget about all the complexity of the DNA-protein relationship and just remember two simple points. First, it takes specific proteins to make specific proteins. That may remind you of the chicken-and-egg problem: how can you get one without the other? That problem is solved if the molecules needed for “DNA-protein translation” are produced by creation.
Second, among all the molecules that translate DNA into protein, there’s not one molecule that is alive. There’s not a single molecule in your body that’s alive. There’s not a single molecule in the living cell that’s alive. A living cell is a collection of nonliving molecules! What does it take to make a living cell alive? The answer is something every scientist recognizes and uses in a laboratory, something every scientist can logically infer from his observations of DNA and protein. What does it take to make a living cell alive? Creative design and organization!
Only creative acts could organize matter into the first living cells, but once all the parts are in place, there is nothing “supernatural” or “mysterious” in the way cells make proteins. If they are continually supplied with the right kind of energy and raw materials, and if all 75-plus of the RNA and protein molecules required for DNA-protein “translation” are present in the right places at the right times in the right amounts with the right structure, then cells make proteins by using DNA’s base series (quite indirectly!) to line up amino acids at the rate of about two per second. In ways scientists understand rather well, it takes a living cell only about four minutes to “crank out” an average protein (500 amino acids) according to DNA specifications.
Scientists also understand how airplanes fly. For that very reason, no scientist believes that airplanes are the result of time, chance, and the properties of aluminum and other materials that make up the airplane. Flying is a property of organization, not of substance. A Boeing 747, for example, is a collection of 4.5 million non-flying parts, but thanks to design and creation (and a continuous supply of energy and of repair services!), it flies.
Similarly, “life” is a property of organization, not of substance. A living cell is a collection of several billion non-living molecules, and death results when a shortage of energy or a flaw in the operational or repair mechanisms allows inherent chemical processes to destroy its biological order.
It’s what we do know and can explain about aluminum and the laws of physics that would convince us that airplanes are the products of creation, even if we never saw the acts of creation. In the same way, it’s what we do know and can explain about DNA and protein and the laws of chemistry which suggests that life itself is the result of special creation.
My point is not based on design per se, but on the kind of design we observe. As creationists point out, some kinds of design, such as snowflakes and wind-worn rock formations, do result from time and chance—given the properties of the materials involved. Even complex relationships, such as the oxygen-carbon dioxide balance in a sealed aquarium, can result from organisms “doing what comes naturally,” given the properties of living things. But just as clearly, other kinds of design, e.g., arrowheads and airplanes, are the direct result of creative design and organization giving matter properties it doesn’t have and can’t develop on its own. What we know about the DNA-protein relationship suggests that living cells have the created kind of design. It’s not so much the molecular complexity as it is the transcendent simplicity.
In the well-known Scientific American book, Evolution, Dickerson7 seems to support my point (without meaning to, I’m sure). After describing the problems in producing the right kinds of molecules for living systems, he says that those droplets that by “sheer chance” contained the right molecules survived longer. He continues, “This is not life, but it is getting close to it. The missing ingredient is . . . .”
What will he say here? The “missing ingredient” is . . . one more protein? . . . a little more DNA? . . . an energy supply? . . . the right acid-base balance? No, he says: “The missing ingredient is an orderly mechanism . . . .” An orderly mechanism! That’s what’s missing—but that’s what life is all about! As I stated before, life is not a property of substance; it’s a property of organization. The same kind of reasoning applies to the pyramids in Egypt, for example. The pyramids are made of stone, but studying the stone does not even begin to explain how the pyramids were built. Similarly, until evolutionists begin to explain the origin of the “orderly mechanism,” they have not even begun to talk about the origin of life.
When it comes to the evolutionary origin of that orderly mechanism, Dickerson adds, we have “no laboratory models; hence one can speculate endlessly, unfettered by inconvenient facts.” With “no laboratory models” to provide data, the case for the evolution of life must be based on imagination. But, as Dickerson admits, “We [evolutionists] can only imagine what probably existed, and our imagination so far has not been very helpful.”
The case for creation, however, is not based on imagination. Creation is based instead on logical inference from our scientific observations, and on simple acknowledgment that everyone, scientists and laymen alike, recognize that certain kinds of order imply creation.
Let me give you another example of the same sort of reasoning. Imagine that you have just finished reading a fabulous novel. Wanting to read another book like it, you exclaim to a friend, “Wow! That was quite a book. I wonder where I can get a bottle of that ink?” Of course not! You wouldn’t give the ink and paper credit for writing the book. You’d praise the author, and look for another book by the same writer. By some twist of logic, though, many who read the fabulous DNA script want to give credit to the “ink (DNA base code) and paper (proteins)” for composing the code.
In a novel, the ink and paper are merely the means the author uses to express his or her thoughts. In the genetic code, the DNA bases and proteins are merely the means God uses to express His thoughts. The real credit for the message in a novel goes to the author, not the ink and paper, and the real credit for the genetic message in DNA goes to the Author of life, the Creator, not to the creature (Rom. 1:25).
The message conveyed by DNA is the kind called “specified complexity” in contrast to randomness or “mere” order. It takes only a simple program or algorithm, for example, to generate a random sequence of letters: (1) Print any letter; (2) Repeat step 1. An ordered, repeat pattern, such as ABCABCABC, could be generated by an algorithm nearly as simple: (1) Print ABC; (2) Repeat step 1. A program ENORMOUSLY larger and more sophisticated would be required to specify, for example, the letter sequence in the first volume of an encyclopedia set! The letter sequence is complex and specific (“specified complexity”), like the base letter sequence in human DNA—except that the DNA contains more information than a thousand volumes of literary works!8
Occasionally, naïve evolutionists argue that crystal formation demonstrates that order can appear spontaneously, without “supernatural” help. Crystal order, yes; specified complexity, no. A crystal is a beautiful but simple repeat pattern produced by the shape and charge of its constituents. At 32°F (O°C), for example, the areas of partial plus and minus charges on water molecules attract them with a force greater than the thermal motion that keeps them apart at higher temperatures. The exquisite shape of the ice crystal is an automatic consequence of the shape and charge distribution (“design features”) of the water molecules. (Incidentally, ice crystal formation is driven by decreasing electrostatic potential, an illustration—not a contradiction—of the famed second law of thermodynamics.)
The “specified complexity” in a DNA sequence is nothing like the “ordered simplicity” or repeat pattern in the ice crystal. Breaking a big ice crystal produces little ice crystals, each with structures and properties like the original. Breaking a DNA chain produces fragments that are dissimilar in structure and lose their function entirely. A child at home can make ice crystals; it takes a team of chemists using expensive equipment to produce a specific DNA sequence from scratch.
The specified complexity in a DNA gene sequence has very high information content. Scientists know two things about information. First, information is independent of the material that carries it. The phrase “In God We Trust” can be written in pen or pencil, typed onto paper or a computer screen, embroidered in lace, etched in stone, impressed on American coins, etc. The message is the same in any case, and it is obviously not produced by the material that conveys it. In other words, informational messages—including genetic messages—have the “exherent” kind of design, reflecting plan, purpose, and special acts of creation. Thus, the meaning of a message lies with its Creator, not its carrier.
Second, information comes only from pre-existing information. Much more information on information can be found in the landmark9 book by internationally respected information theorist Werner Gitt, In the Beginning Was Information. Biblically, that concept is expressed as “In the beginning, God . . .” (Gen. 1:1) and as “In the beginning was the Word” (John 1:1). The word “Word,” identified as Jesus Christ in John 1:14, is the Greek word “Logos.” Logos is a grand word in Greek, connoting divine plan, reason for being, etc., and means “study of” as the suffix “ology” attached to the various academic disciplines. Wow! Our DNA ties us back to the ultimate source of meaning and purpose for the whole universe!
Creation thus stands between the classic extremes of mechanism and vitalism. Mechanists, including evolutionists, believe that both the operation and origin of living things are the result of the laws of chemistry which reflect the inherent properties of matter. Vitalists believe that both the operation and origin of living systems depend on mysterious forces that lie beyond scientific description. According to creation, living things, including their DNA codes, operate in understandable ways that can be described in terms of scientific laws, but such observations include properties of organization that logically imply a created origin of life.
In this sense, the Bible proved to be, as it often has, far ahead of its time. Into the 1800s, most scientists and philosophers believed living things were made of something fundamentally different from non-living. Genesis 1–2 tells us living things, human beings included, were just made of “dust of the ground.” Indeed, scientists now recognize that living cells are composed of only a few simple elements. It’s not the stuff (“dust”) we’re made of that makes us special; it’s the way we’re put together. It’s not the metal and glass that make an airplane fly, nor the ink and paper that write a novel. Similarly, it’s not the “dust” that makes life, but the way it’s put together with creative design and organization. When that organization is lost, we return to “dust,” the simple elements that we are made of, just as other created objects break down into their simpler parts when left to the ravages of time, chance, and chemistry.
The creationist, then, recognizes the orderliness that the vitalist doesn’t see, but he doesn’t limit himself to only those kinds of order that result from time, chance, and the properties of matter, as the evolutionist does. Creation introduces levels of order and organization that greatly enrich the range of explorable hypotheses and turn the study of life into a scientist’s delight. Science requires an orderliness in nature. One of the real emotional thrills of my changing from evolution to creation was realizing both that there are many more levels of order than I had once imagined and that order in nature, and a mind in tune with it, were guaranteed by God himself. It’s no wonder that explicit biblical faith gave initial success to the founding fathers of modern experimental science (a couple of centuries before evolution came along to shift the basis toward time and chance).
If the evidence for the creation of life is as clear as I say it is, then other scientists, even those who are evolutionists, ought to see it—and they do.
I once took my students to hear Francis Crick, who shared a Nobel Prize for the discovery of DNA’s structure. After explaining why life could not and did not evolve on earth, he argued instead for “directed panspermia,” his belief that life reached earth in a rocket fired by intelligent life on some other planet. Crick admitted that his view only moved the creation-evolution question back to another time and place, but he argued that different conditions (which he did not specify) might have given life a chance to evolve that it did not have on earth.10
Creationists are pleased that Crick recognized the same fatal flaws in chemical evolution that they have cited for years, but creationists also point out that the differences between “chemical chemistry” and “biological chemistry” are wrapped up with the fundamental nature of matter and energy and would apply on other planets as well as on earth.11
That opinion seems to be shared in part by famed astronomer Sir Fred Hoyle,12 who made the news under the heading: “There must be a God.” Hoyle and his colleague, Chandra Wickramasinghe, independently reached that conclusion after their mathematical analyses showed that believing that life could result from time, chance, and the properties of matter was like believing that “a tornado sweeping through a junkyard might assemble a Boeing 747 from the materials therein.”
Drawing the logical inference from our scientific knowledge, both scientists concluded that “it becomes sensible to think that the favorable properties of physics on which life depends are in every respect deliberate” (emphasis Hoyle’s). Both were surprised by their results. Hoyle called himself an agnostic, and, in the same article, Wickramasinghe said he was an atheistic Buddhist who “was very strongly brainwashed to believe that science cannot be consistent with any kind of deliberate creation.”
My purpose in quoting these scientists (and others later on) is not, of course, to suggest that they are creationists who would endorse all my views.13 Rather, it is simply to show that experts in the field, even when they have no preference for creationist thinking, at least agree with the creationists on the facts, and when people with different viewpoints agree, we can be pretty sure what the facts are. I also want to show that scientists who are not creationists are able to see that creation is a legitimate scientific concept, whose merits deserve to be compared with those of evolution.
In that light, I’d like to call your attention to a fascinating and revolutionary book, Evolution: A Theory in Crisis, by a prominent molecular biologist, Dr. Michael Denton.14 In a television program we did together, and in our extensive personal conversations, Dr. Denton describes himself as a child of the secular age who desires naturalistic explanations when he can find them. When it comes to the origin of life, Dr. Denton explains with authority and stark clarity that evolutionists are nowhere near a naturalistic explanation at present. After comparing the genetic programs in living things to a library of a thousand volumes encoding a billion bits of information and all the mathematically intricate algorithms for coordinating them, Dr. Denton refers to the chemical evolution scenario as “simply an affront to reason,” i.e., an insult to the intelligence! (p. 351).
He openly and frankly states that the thesis of his book is “anti-evolutionary” (p. 353), but it seems to me that he is cautiously taking a step even further. The first chapter of his book is titled “Genesis Rejected,” and he would react very strongly against being called a creationist, but in his honest analysis of the creation-evolution controversy through history, Dr. Denton freely admits that many of the scientific views of the early creationists have been vindicated by modern discoveries in science.
Take William Paley’s classic argument that design in living things implies a Designer just as clearly as design in a watch implies a watchmaker. In The Blind Watchmaker,15 discussed later, Richard Dawkins argues—incorrectly—that Paley was wrong. Denton states, “Paley was not only right in asserting an analogy between life and a machine, but also remarkably prophetic in guessing that the technological ingenuity realized in living systems is vastly in excess of anything yet accomplished by man” (emphasis added). Then Denton goes on to summarize his thinking on life’s origin (p. 341) as follows:
The almost irresistible force of the analogy has completely undermined the complacent assumption, prevalent in biological circles over most of the past century, that the design hypothesis can be excluded on the grounds that the notion is fundamentally a metaphysical a priori concept and therefore scientifically unsound. On the contrary, the inference to design is a purely a posteriori induction based on a ruthlessly consistent application of the logic of analogy. The conclusion may have religious implications, but it does not depend on religious presuppositions (emphasis added).
Now that’s quite an admission! Even though he would deny any leaning toward a Christian concept of creation, this leading molecular biologist sees quite plainly that a scientific concept of creation can be constructed, just as I’ve said, using the ordinary tools of science, logic, and observation. In fact, Denton intimates that creation scientists have shown more respect than evolutionists for empirical evidence and a “ruthlessly consistent” application of logic!
It’s also true, as Denton concludes, that creation may have religious implications, but so does evolution, and that should not prevent our evaluating their scientific merits on the basis of logic and observation alone.
In a short but thought-provoking article, British physicist H.S. Lipson16 reached the same conclusion. First he expressed his interest in life’s origin, then his feeling—quite apart from any preference for creation—that, “In fact, evolution became in a sense a scientific religion; almost all scientists have accepted it and many are prepared to ‘bend’ their observations to fit with it.”
After wondering how well evolution has stood up to scientific testing, Lipson continues: “To my mind, the theory [evolution] does not stand up at all.” Then he comes to the heart of the issue: “If living matter is not, then, caused by the interplay of atoms, natural forces, and radiation [i.e., time, chance, and chemistry], how has it come into being?” After dismissing a sort of directed evolution, Lipson concludes: “I think, however, that we must go further than this and admit that the only acceptable explanation is creation” (emphasis his).
Like Hoyle and Wickramasinghe, Lipson is a bit surprised and unhappy with his own conclusion. He writes, “I know that this [creation] is anathema to physicists, as indeed it is to me.” But his sense of honesty and scientific integrity forces him to conclude his sentence thus: “. . . but we must not reject a theory that we do not like if the experimental evidence supports it.”
By the way, let me assure you that not all who see the evidence of creation are unhappy about it! Witness Dr. Dean Kenyon. Dr. Kenyon is a molecular biologist whose area of research interest is specifically the origin of life. His book on life’s origin, Biochemical Predestination, opened with praises for Darwinian evolution, and he taught evolution at San Francisco State University for many years.
A couple of students in Dr. Kenyon’s class once asked him to read a book by Dr. Duane Gish on creation science. He didn’t want to, but thanks to their polite persistence (1 Pet. 3:15), he resolved to read it and refute it, but, as I heard him tell it, he read it and couldn’t refute it. Instead, Dr. Kenyon got interested in creation science and began a long re-evaluation of the scientific evidence, which finally led him to the happy conclusion that life, including his, is here as a result of creation, the deliberate plan and purpose of a personal Creator God!17
Creation: Facts of Life
Dr. Parker, a leading creation scientist and former AiG speaker, presents the classic arguments for evolution used in public schools, universities, and the media, and refutes them in an entertaining and easy-to-read style. A must for students and teachers alike! This is a great book to give to a non-Christian as a witnessing tool.
Read OnlineFootnotes
- Gary E. Parker, W. Ann Reynolds, and Rex Reynolds, DNA: The Key to Life, Rev. ed., Programmed Biology Series (Chicago, IL: Educational Methods, Inc., 1977).
- Gary E. Parker, “The Origin of Life on Earth” Creation Science Research Quarterly (September 1970); Gary E. Parker and Thomas R. Mertens, Life’s Basis: Biomolecules (New York: John Wiley and Sons, 1977), available in Spanish as Biomoleculas: Basa de la Vida.
- Richard B. Bliss and Gary E. Parker, Origin of Life, Two Models Creation-Evolution Series (Green Forest, AR: Master Books, 1979).
- Illustra Media, Unlocking the Mystery of Life, DVD/VHS, 2002.
- Charles Thaxton, Walter Bradley, and Roger Olsen, The Mystery of Life’s Origin: Reassessing Current Theories (New York: Philosophical Library, 1984).
- Parker, Reynolds, and Reynolds, DNA: The Key to Life; Parker and Mertens, Life’s Basis: Biomolecules; Bliss and Parker, Origin of Life; Thaxton, Bradley, and Olsen, The Mystery of Life’s Origin.
- Richard E. Dickerson, “Chemical Evolution and the Origin of Life,” Scientific American, and Scientific American book Evolution (September 1978).
- Jonathan Sarfati, Refuting Evolution (Green Forest, AR: Master Books, 1999).
- Werner Gitt, In the Beginning Was Information (Green Forest, AR: Master Books, 2006).
- Francis Crick, “The Seeds of Life,” Discover (October 1981).
- Michael Denton, Nature’s Destiny (New York: The Free Press, 1998).
- Sir Fred Hoyle and Chandra Wickramasinghe, as quoted in “There Must Be a God,” Daily Express (August 14,1981); and “Hoyle on Evolution,” Nature (November 12, 1981).
- Duane T. Gish, Creation Scientists Answer Their Critics (Green Forest, AR: Master Books, 1993).
- Michael Denton, Evolution: A Theory in Crisis (London: Burnctt Books, 1985).
- Richard Dawkins, The Blind Watchmaker (New York: W.W. Norton, 1986).
- H.S. Lipson, “A Physicist Looks at Evolution,” Physics Bulletin (May 1980): page 138.
- Percival Davis and Dean Kenyon, Of Pandas and People, 2nd edition (Dallas, TX: Haughton Pub. Co., 1999).

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis

