
Chapter 1
Can Natural Processes Explain the Origin of Life?
When considering how life began, there are only two options. Either life was created by an intelligent source (God) or it began by natural processes. The common perception presented in many textbooks and in the media is that life arose from nonlife in a pool of chemicals about 3.8 billion years ago. The claim by evolutionists is that this formation of life was the result of time, chance, and natural processes. One widely used example of how life could have formed by natural processes is the Miller-Urey experiment, performed in the early 1950s.
Miller’s objective was not to create life but to simulate how life’s basic building structures...might have formed in the early earth.
Miller’s objective was not to create life but to simulate how life’s basic building structures (amino acids1) might have formed in the early earth. In the experiment, Miller attempted to simulate the early atmosphere of earth by using certain gases, which he thought might produce organic compounds necessary for life. Since the gases he included (water, methane, ammonia, and hydrogen) do not react with each other under natural conditions, he generated electrical currents to simulate some form of energy input (such as lightning) that was needed to drive the chemical reactions. The result was production of amino acids. Many textbooks promote this experiment as the first step in explaining how life could have originated. But there is more to this experiment than what is commonly represented in textbooks.
The Rest of the Story—Some Critical Thinking
When we examine the purpose, assumptions, and results of the Miller experiment, there are three critical thinking questions that can be raised:
- How much of the experiment was left to chance processes or how much involved intelligent design?
- How did Miller know what earth’s early atmosphere (billions of years ago) was like?
- Did Miller produce the right type of amino acids used in life?
The Method Used
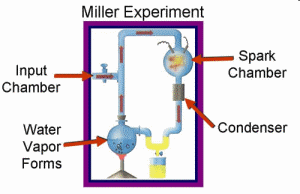
In the experiment, Miller was attempting to illustrate how life’s building blocks (amino acids) could have formed by natural processes. However, throughout the experiment Miller relied on years of intelligent research in chemistry. He purposely chose which gases to include and which to exclude. Next, he had to isolate the biochemicals (amino acids) from the environment he had created them in because it would have destroyed them. No such system would have existed on the so-called primitive earth. It appears Miller used intelligent design throughout the experiment rather than chance processes.
The Starting Ingredients
How did Miller know what the atmosphere was like billions of years ago? Miller assumed that the early earth’s atmosphere was very different from today. He based his starting chemical mixture on the assumption that the early earth had a reducing atmosphere (an atmosphere that contains no free oxygen). Why did Miller and many other evolutionists assume there was no free oxygen in earth’s early atmosphere? As attested below, it is well known that biological molecules (specifically amino acid bonds) are destroyed in the presence of oxygen, making it impossible for life to evolve.
Oxygen is a poisonous gas that oxidizes organic and inorganic materials on a planetary surface; it is quite lethal to organisms that have not evolved protection against it.2
In the atmosphere and in the various water basins of the primitive earth, many destructive interactions would have so vastly diminished, if not altogether consumed, essential precursor chemicals, that chemical evolution rates would have been negligible.3
Therefore, in order to avoid this problem, evolutionists propose that earth’s first atmosphere did not contain any freestanding oxygen. We must ask ourselves, “Is there any evidence to support this claim, or is it based on the assumption that evolution must be true?” As it turns out, the existence of a reducing atmosphere is merely an assumption not supported by the physical evidence. The evidence points to the fact that the earth has always had oxygen in the atmosphere.
There is no scientific proof that Earth ever had a non-oxygen atmosphere such as evolutionists require. Earth’s oldest rocks contain evidence of being formed in an oxygen atmosphere.4
The only trend in the recent literature is the suggestion of far more oxygen in the early atmosphere than anyone imagined.5
If we were to grant the evolutionists’ assumption of no oxygen in the original atmosphere, another fatal problem arises. Since the ozone is made of oxygen, it would not exist; and the ultraviolet rays from the sun would destroy any biological molecules. This presents a no-win situation for the evolution model. If there was oxygen, life could not start. If there was no oxygen, life could not start. Michael Denton notes:
What we have is sort of a “Catch 22” situation. If we have oxygen we have no organic compounds, but if we don’t have oxygen we have none either.6
Because life could not have originated on land, some evolutionists propose that life started in the oceans. The problem with life starting in the oceans, however, is that as organic molecules formed, the water would have immediately destroyed them through a process called hydrolysis. Hydrolysis, which means “water splitting,” is the addition of a water molecule between two bonded molecules (two amino acids in this case), which causes them to split apart. Many scientists have noted this problem.
Besides breaking up polypeptides, hydrolysis would have destroyed many amino acids.7
In general the half-lives of these polymers in contact with water are on the order of days and months—time spans which are surely geologically insignificant.8
Furthermore, water tends to break chains of amino acids apart. If any proteins had formed in the oceans 3.5 billion years ago, they would have quickly disintegrated.9
Scientifically, there is no known solution for how life could have chemically evolved on the earth.
On the Other Hand . . .
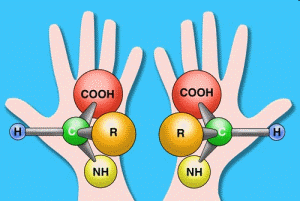
Because the scientific evidence contradicts the origin of life by natural processes, Miller resorted to unrealistic initial conditions to develop amino acids in his experiment (no oxygen and excessive energy input). However, there is more to the story. Producing amino acids is not the hard part. The difficult part is getting the right type and organization of amino acids. There are over 2,000 types of amino acids, but only 20 are used in life. Furthermore, the atoms that make up each amino acid are assembled in two basic shapes. These are known as left-handed and right-handed. Compare them to human hands. Each hand has the same components (four fingers and a thumb), yet they are different. The thumb of one hand is on the left, and the thumb of the other is on the right. They are mirror images of each other. Like our hands, amino acids come in two shapes. They are composed of the same atoms (components) but are mirror images of each other, called left-handed amino acids and right-handed amino acids. Objects that have handedness are said to be chiral (pronounced “ky-rul”), which is from the Greek for hand.
Handedness is an important concept because all amino acids that make up proteins in living things are 100 percent left-handed. Right-handed amino acids are never found in proteins. If a protein were assembled with just one right-handed amino acid, the protein’s function would be totally lost. As one PhD chemist has said:
Many of life’s chemicals come in two forms, “left-handed” and “right-handed.” Life requires polymers with all building blocks having the same “handedness” (homochirality)—proteins have only “left-handed” amino acids. . . . But ordinary undirected chemistry, as is the hypothetical primordial soup, would produce equal mixtures of left- and right-handed molecules, called racemates.10
A basic chemistry textbook admits:
This is a very puzzling fact. . . . All the proteins that have been investigated, obtained from animals and from plants from higher organisms and from very simple organisms—bacteria, molds, even viruses—are found to have been made of L-amino [left-handed] acids.11
The common perception left by many textbooks and journals is that Miller and other scientists were successful in producing the amino acids necessary for life. However, the textbooks and media fail to mention that what they had actually produced was a mixture of left- and right-handed amino acids, which is detrimental to life. The natural tendency is for left- and right-handed amino acids to bond together. Scientists still do not know why biological proteins use only left-handed amino acids.
The reason for this choice [only left-handed amino acids] is again a mystery, and a subject of continuous dispute.12
Jonathan Wells, a developmental biologist, writes:
So we remain profoundly ignorant of how life originated. Yet the Miller-Urey experiment continues to be used as an icon of evolution, because nothing better has turned up. Instead of being told the truth, we are given the misleading impression that scientists have empirically demonstrated the first step in the origin of life.13
Despite the fact that the Miller experiment did not succeed in creating the building blocks of life (only left-handed amino acids), textbooks continue to promote the idea that life could have originated by natural processes. For example, the following statement from a biology textbook misleads students into thinking Miller succeeded:
By re-creating the early atmosphere (ammonia, water, hydrogen and methane) and passing an electric spark (lightning) through the mixture, Miller and Urey proved that organic matter such as amino acids could have formed spontaneously.14
First, note the word proved. Miller and Urey proved nothing except that life’s building blocks could not form in such conditions. Second, the textbook completely ignores other evidence, which shows that the atmosphere always contained oxygen. Third, the textbook ignores the fact that Miller got the wrong type of amino acids—a mixture of left- and right-handed.
The Miller experiment (and all experiments since then) failed to produce even a single biological protein by purely naturalistic processes. Only God could have begun life.
Information
Another important component of life is information. The common factor in all living organisms is the information contained in their cells. Where and how did all this coded information arise? Proteins are amazingly versatile and carry out many biochemical functions, but they are incapable of assembling themselves without the assistance of DNA. The function of DNA is to store information and pass it on (transcribe) to RNA, while the function of RNA is to read, decode, and use the information received from DNA to make proteins. Each of the thousands of genes on a DNA molecule contains instructions necessary to make a specific protein that, in turn, is needed for a specific biological function.
Any hypothesis or model meant to explain how all life evolved from lifeless chemicals into a complex cell consisting of vast amounts of information also has to explain the source of information and how this information was encoded into the genome. All evolutionary explanations are unable to answer this question. Dr. Werner Gitt, former physics professor and director of information processing at the Institute of Physics and Technology in Braunschweig, Germany, and Dr. Lee Spetner both agree that information cannot arise by naturalistic processes:
There is no known law of nature, no known process and no known sequence of events which can cause information to originate by itself in matter.15
Not even one mutation has been observed that adds a little information to the genome. This surely shows that there are not the millions upon millions of potential mutations the theory [evolution] demands.16
The DNA code within all plant and animal cells is vastly more compact than any computer chip ever made. DNA is so compact that a one-square-inch chip of DNA could encode the information in over seven billion Bibles. Since the density and complexity of the genetic code is millions of times greater than man’s present technology, we can conclude that the originator of the information must be supremely intelligent.
Two biologists have noted:
DNA is an information code. . . . The overwhelming conclusion is that information does not and cannot arise spontaneously by mechanistic processes. Intelligence is a necessity in the origin of any informational code, including the genetic code, no matter how much time is given.17
God, in His Word, tells us that His creation is a witness to himself and that we do not have an excuse for not believing (Romans 1:19–20). The fact that the information encoded in DNA ultimately needs to have come from an infinite source of information testifies to a Creator. And as we saw above, the only known way to link together left-handed amino acids is through purposeful design.
Since no human was present to assemble the first living cell, it is further testimony to an all-wise Creator God.
Given Enough Time . . .
Nobel prize-winning scientist George Wald once wrote:
However improbable we regard this event [evolution], or any of the steps it involves, given enough time, it will almost certainly happen at least once. . . . Time is the hero of the plot. . . . Given so much time, the impossible becomes possible, the possible becomes probable, the probable becomes virtually certain. One only has to wait; time itself performs miracles.18
In the case of protein formation, the statement “given enough time” is not valid. When we look at the mathematical probabilities of even a small protein (100 amino acids) assembling by random chance, it is beyond anything that has ever been observed.
What is the probability of ever getting one small protein of 100 left-handed amino acids? (An average protein has at least 300 amino acids in it—all left-handed.) To assemble just 100 left-handed amino acids (far shorter than the average protein) would be the same probability as getting 100 heads in a row when flipping a coin. In order to get 100 heads in a row, we would have to flip a coin 1030 times (this is 10 x 10, 30 times). This is such an astounding improbability that there would not be enough time in the whole history of the universe (even according to evolutionary time frames) for this to happen.
The ability of complex structures to form by naturalistic processes is essential for the evolution model to work. However, the complexity of life appears to preclude this from happening. According to the laws of probability, if the chance of an event occurring is smaller than 1 in 10-50, then the event will never occur (this is equal to 1 divided by 1050 and is a very small number).19
What have scientists calculated the probability to be of an average-size protein occurring naturally? Walter Bradley, PhD, materials science, and Charles Thaxton, PhD, chemistry,5 calculated that the probability of amino acids forming into a protein is:
4.9 x 10-191
This is well beyond the laws of probability (1x10-50), and a protein is not even close to becoming a complete living cell. Sir Fred Hoyle, PhD, astronomy, and Chandra Wickramasinghe, professor of applied math and astronomy, calculated that the probability of getting a cell by naturalistic processes is:
1 x 10-40,000
No matter how large the environment one considers, life cannot have had a random beginning. . . . There are about two thousand enzymes, and the chance of obtaining them all in a random trial is only one part in (1020)2000 = 1040,000, an outrageously small probability that could not be faced even if the whole universe consisted of organic soup.20
Conclusion
As we have seen, the scientific evidence confirms that “in the beginning, God created.” Life cannot come from nonlife; only God can create life. True science and the Bible will always agree. Whether in biology, astronomy, geology, or any other field of study, we can trust God’s Word to be accurate when it speaks about these topics. Let us stand up for the truth of Genesis and take back our culture.
The New Answers Book 2
In The New Answers Book 2 you’ll find 31 more great answers to big questions for the Christian life.
Read Online Buy BookFootnotes
- The basic building blocks of all living systems are proteins, which consist of only 20 different types of amino acids. The average number of amino acids in a biological protein is over 300. These amino acids must be arranged in a very specific sequence for each protein.
- P. Ward and D. Brownlee, Rare Earth (New York: Copernicus, 2000), p. 245.
- C. Thaxton, W. Bradley, and R. Olsen, The Mystery of Life’s Origin: Reassessing Current Theories (New York: Philosophical Library, 1984), p. 66.
- H. Clemmey and N. Badham, “Oxygen in the Atmosphere: An Evaluation of the Geological Evidence,” Geology 10 (1982): 141.
- Thaxton, Bradley, and Olsen, The Mystery of Life’s Origin, p. 80.
- M. Denton, Evolution: A Theory in Crisis (Bethesda, MD: Adler & Adler, 1985), p. 261.
- Encyclopedia of Science and Technology, Vol. 1, 1982: p. 411–412.
- K. Dose, The Origin of Life and Evolutionary Biochemistry (New York: Plenum Press, 1974), p. 69.
- R. Morris, The Big Questions (New York: Times Books/Henry Holt, 2002), p. 167.
- J. Sarfati, In Six Days, (Green Forest, AR: Master Books, 2000), p. 82.
- Linus Pauling, General Chemistry, 3rd ed. (San Francisco, CA: W.H. Freeman & Co., 1970), p. 774.
- Robert Shapiro, Origins: A Skeptic’s Guide to the Creation of Life on Earth (New York: Summit Books, 1986), p. 86.
- J. Wells, Icons of Evolution (Washington, DC: Regnery Pub., 2000), p. 24.
- Kenneth Miller and Joseph Levine, Biology, 5th ed. (Upper Saddle River, NJ: Pearson Prentice Hall, 2000).
- W. Gitt, In the Beginning Was Information (Green Forest, AR: Master Books, 2006).
- L. Spetner, Not by Chance (New York: Judaica Press, 1997), p. 160.
- L. Lester and R. Bohlin, The Natural Limits to Biological Change, (Dallas, TX: Probe Books, 1989), p. 157.
- G. Wald, “The Origin of Life,” Scientific American 191 (August 1954): 45.
- Probability expert Emile Borel wrote, “Events whose probabilities are extremely small never occur. . . . We may be led to set at 10-50 the value of negligible probabilities on the cosmic scale.” (E. Borel, Probabilities and Life, [New York: Dover Publications, 1962], p. 28.)
- F. Hoyle and C. Wickramasinghe, Evolution from Space (New York: Simon and Schuster, 1984), p. 176.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis



