Special Tools of Life
Did you know that living things make their own tools? For example, they use enzymes to break down large molecules for parts (building blocks) and some for energy.
Have you ever tried to do a job, but not had the right tools? Many tools (like screw drivers, saws and wrenches) come in a variety of sizes and shapes so that different jobs can be done quickly and easily. Specialty tools are designed with unique shapes and bends so a person can do a special job very well. Trying to use a tool poorly suited for the job (like a slotted screw driver on a Phillips screw) often leads to poor results and frustration.
Did you know that living things make their own tools? For example, they use enzymes to break down large molecules for parts (building blocks) and some for energy. Other enzymes construct things, such as cell walls. These enzymes are like awesomely designed specialty tools with a very specific size and shape. Many do only one job and do it very well. There are several thousand known enzymes that are used to do all the different jobs necessary to keep living things alive and well.1
How do living things make these tools? They use the information from their DNA (the molecule in the nucleus that contains all the ‘programs’ a cell needs to function) and materials from the world around them. Since it would be wasteful for a creature to constantly be producing an enzyme even when it wasn’t needed, the DNA also contains an ON/OFF switch (repressor gene). This is normally switched OFF until the enzyme is needed.2

The unsuspecting soil dwelling bacteria were suddently whisked off to the lab and fed nothing but xylitol.
There are two competing worldviews as to how this information got on the DNA. Creationists believe that God put the information there when he created life, as recorded in the Bible. Evolutionists believe that it got there by mistake; actually by a lot of mistakes. That’s right! Before a cell divides, it copies the DNA so each new cell has a copy. Sometimes there is a copying error, called a mutation, that takes place. These mutations, according to the theory of evolution, are the source of new information. Natural selection then supposedly selects the useful mutations and eliminates the useless ones. This is how evolutionists account for new enzymes, hormones, organs, etc. as living things have (supposedly) developed upward from a single-celled beginning.
Most mutations that make a noticeable difference are harmful. A few might be considered beneficial—at least sometimes.3 But do these mutations add information to the DNA? A number of scientists thought so when they saw mutations in a bacterium that normally grows in the soil.4 This bacterium can grow well when it has one of several unusual sugars as an energy source: ribitol or D-arabitol.5 The scientists growing it in the lab tried giving it a very similar sugar, xylitol, as its only energy source. Xylitol is not normally found in the bacteria’s environment. The bacteria have no enzymes specifically designed for the first step in breaking it down.
The wild-type of the bacteria couldn’t grow. However, a mutant strain (X1) arose that could grow on xylitol, although very slowly. Later, a second mutant strain (X2) arose from X1 that grew faster on xylitol. From this, a third mutant strain (X3) developed that grew faster still. Evidence for evolution? A new enzyme evolving? Hardly.
Ribitol is broken down for energy by a series of steps, and each step is done by a different enzyme. The enzyme for the first step has a really cool name, ribitol dehydrogenase (RDH). If you’re ever stuck trying to come up with a name for a pet, this might be just the name. RDH, the enzyme, is like a specialty tool that is designed to take apart ribitol. Although xylitol is very similar to ribitol, RDH doesn’t take it apart very easily. When it does finally get it apart, the bacteria has all the right enzymes to break it down the rest of the way. The X1 was able to grow because the mutation destroyed the ON/OFF switch for RDH. Therefore, RDH was constantly produced and the large amounts of this enzyme broke down the xylitol that was able to get into the cell.
A New Enzyme, or a Defective One?6
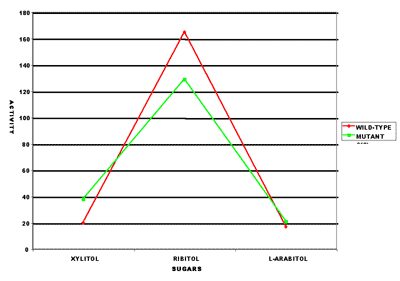
Living things use enzymes that are usually very specific. Ribitol dehydrogenase (RDH) is very specific in the wild-type bacteria (red) as seen by the tall spike in the graph. The mutant (X2, shown in green) has an enzyme that has lost some of its specificity as seen by a less pronounced peak and higher base.
If the theory of evolution were true, there should be many examples of new enzymes developing with increasing specificity. So far we know of none.
The second mutation affected the RDH enzyme and changed its shape slightly. This new shape caused RDH to be less effective on ribitol, but more effective on xylitol and L-arabitol (the mirror image of D-arabitol) as shown in the accompanying graph. This means RDH in X2 was less specific in its action. Is it beneficial for a mechanic to have bolts randomly loosened throughout the car when he wants to remove the starter? There are plenty of acids and bases that can take lots of molecules apart very well. However, living things use tools that are more specific so that important parts of the cell aren’t dismantled. A loss of specificity is a loss of information and usually not beneficial. Nevertheless, since the scientists insisted on growing the bacteria on only xylitol, the mutation was beneficial in this case. It allowed X2 to grow about 2½ times faster than X1, which was still slow, but it did grow faster.
The third mutation allowed more xylitol into the bacteria cell. Normally cells are very picky about what they let in. Some things that are important to the cells are pulled in. This bacteria has an enzyme that brings D-arabitol into the cell. When xylitol is present, it is brought in as well because it is so similar. This enzyme also has an ON/OFF switch in the DNA which is kept switched OFF unless D-arabitol is available. The third mutation destroyed this ON/OFF switch and xylitol ended up with a free ride into the cell. This enabled the X3 mutant to grow about twice as fast as X2.
If the theory of evolution were true, each step, on average, would add new information. This would explain the tremendous amount of information in bass, bison and birds compared to bacteria. In the length of time mankind has been studying genetics and mutations, many examples of information increase should have been documented. Yet, none have been found. In the example discussed here, the X3 mutant still only grew about half as well on xylitol as the wild-type did on ribitol. If the mutants were put back into a soil environment, they would quickly die out because they are less fit. Many otherwise intelligent people, in their quest for knowledge, have missed the obvious. Anyone who looks at a well designed tool should know that someone made it.
“For since the creation of the world God’s invisible qualities—his eternal power and divine nature—have been clearly seen, being understood from what has been made, so that men are without excuse. For although they knew God, they neither glorified him as God nor gave thanks to him, but their thinking became futile and their foolish hearts were darkened. Although they claimed to be wise, they became fools …” Romans 1:20–22.
Mutations: A Losing Change
Although it is extremely rare for a mutation to be beneficial, the following mutations were, as long as the bacteria were restricted to xylitol.
| Bacterium | Loss | Benefit |
|---|---|---|
| Wild-type | None | Grows well in soil |
| Mutant X1 | On/off switch for RDH destroyed; RDH constantly produced even when ribitol is absent (waste of resources; inefficient) | RDH available in large amounts; xylitol broken down slowly; X1 grows slowly on xylitol |
| Mutant X2 | RDH shape changed; loses specificity for ribitol (grows slower than wild type on ribitol) | RDH works better on xylitol; X2 grows faster on xylitol than X1 |
| Mutant X3 | On/off switch for ‘enzyme’ that brings in D-arabitol destroyed; enzyme constantly produced, even when D-arabitol absent (waste of resources) | Xylitol gets a free ride into the cell; more xylitol available in cell; X3 grows faster on xylitol than X2 |
Footnotes
- Koolman, J. and K.H. Rohm, Color Atlas of Biochemistry, Thieme, New York, p. 86, 1996.
- Most of this article is based on Spetner, L., Evolution, Randomness, and Hashkafa, <bex.nsstc.uah.edu/RbS/CLONE/VGS/spetner_evol1.html>.
- For example, see Are There Beneficial Mutations?
- Then known as Aerobacter aerogenes, this organism has been reclassified as one of several Klebsiella species. Return to text
- The word sugar is used somewhat simplistically in this article. The four sugars mentioned (ribitol, D-arabitol, xylitol, and L-arabitol) are actually sugar alcohols. All four have the same chemical formula (C5H12O5). The differences are due to how the hydrogen (H) and the hydroxyl (OH) groups are arranged on the five-carbon backbone.
- Graph adapted from bar graph at Is antibiotic resistance really due to increase in information?

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis