
Chapter 14
What About Cloning and Stem Cells?
There are few issues in our society that raise as many emotional and ethical concerns as cloning and stem cells.
There are few issues in our society that raise as many emotional and ethical concerns as cloning and stem cells. Scientists, journalists, special interest groups, and even patients themselves regularly bombard us with their particular views on this issue. How are we to know what to think regarding these issues? How do we separate fact from fiction? Since cloning and stem cells are two separate (but related) issues, we will deal with them individually.
What Is Cloning?
Cloning is a process by which a genetically identical copy of a gene, an entire cell, or even an organism is produced. For this chapter, we will confine the discussion primarily to the cloning of an entire organism. This is a topic about which there is much misinformation. It is also a subject that raises some very serious ethical issues.
Cloning as usually understood is an artificial process, meaning it is carried out in a laboratory setting. It can and does, however, occur regularly in nature. There are organisms (e.g., bacteria, protists, and some plants) that typically reproduce by asexual reproduction. Here a genetically identical copy of the parent is produced by the splitting of a single cell (the parent cell).
Identical twins are also clones. In fact, identical twins have been called “natural clones” since splitting of a fertilized egg causes this, producing two copies of the same organism.

Dolly was the result of cloning a mammary cell from a mature sheep, but at what cost?
It is the issue of artificial cloning that has captured the interest of so many in our society. This process has garnered much attention in recent years, especially with the birth of the famous sheep, Dolly. Actually, many different types of animals have been cloned including tadpoles, mice, cats, sheep, cattle, a horse, and others.
How Is a Clone Made?
The simplest method for making a clone is to remove the nucleus (containing the organism’s DNA) from a somatic (body) cell in the animal you want to clone. You then take an egg cell (from the same type of animal) that has had its own nucleus removed, and you place the donor nucleus into the egg cell. This is called somatic cell nuclear transfer (SCNT). This egg is grown briefly in a test tube and then implanted into the womb of an adult animal. If there are no complications, at the end of gestation an animal is born with an identical genetic makeup of the donor animal. As one might imagine, the process is technically quite difficult. Let’s use the aforementioned Dolly as an example. It took 277 eggs that ultimately produced 29 embryos and only one living sheep to create Dolly.1 This is consistent with the failure rate for other animals. As can be seen, many embryos are wasted in these attempts.
It should also be noted that Dolly died at age six. She apparently died of a respiratory infection. Some have suggested that she exhibited signs of premature aging, but others have disputed these reports. The strongest speculation is that her early death was due to shortening of telomeres. Telomeres are segments of DNA that exist on the ends of chromosomes. They progressively shorten with age due to repeated cell division until they reach a point that no further replication of the chromosome can occur. Since Dolly was cloned from a 6-year-old sheep, it could be said that Dolly’s DNA when she died was actually 12 years old. The telomere issue remains a significant problem for those involved in cloning research.
How Can Cloning Be Used?
There are two main purposes for cloning: to produce an identical organism (reproductive cloning) or to produce a cloned embryo for the purpose of obtaining embryonic stem cells (therapeutic cloning). There are those who promote reproductive cloning in many different areas. For example, cloning of certain animals used for food or for animals used in specific work environments has been suggested. This has also been proposed as a possible solution for the rescue of many endangered species. Although it is far beyond our present technology, some have theorized that the extinct woolly mammoth or even dinosaurs might in the future be produced through reproductive cloning!
Therapeutic cloning is aimed at producing cloned embryos from which embryonic stem cells may be obtained. This is done ostensibly to use the stem cells to treat disease or illness. While this is laudable in one sense, there are serious ethical issues that arise (see the following stem cell section).
The obvious next step would be to consider cloning a human being. There are those who advocate therapeutic cloning of humans to provide an adequate supply of embryonic stem cells. It has even been suggested by some that humans should be reproductively cloned in order to provide a ready reserve of tissues and organs should they ever be needed. The clone would simply be “spare parts,” to be used at the discretion of the “parent” human.
So What’s the Problem?
If man is just another animal, just a higher form of pond scum, there really is no problem. Cloning a person is totally justifiable. Just make copies of ourselves and chop them up as we please. People are nothing special.
But those of us who trust in God’s Word know there is a problem here. We are not just a higher form of pond scum. We are not just animals. We are made in the image of the Creator.
And God said, “Let us make man in our image, after our likeness . . .” (Genesis 1:26, ASV).
Therefore, humans are not to be created at man’s whim. Rather, we are a special creation of our Father in heaven.
What Is a Stem Cell?
Simply put, a stem cell is a cell in the body (or in an embryo) that has the capability of turning into many specialized cell types. At the time of conception, when the sperm and egg unite, we consist of only one cell. Ultimately, as this cell divides into two cells, then four, then eight, and so on, the roughly 200 different cell types in the body must be produced. This process can occur because of stem cells.
A stem cell is a cell in the body (or in an embryo) that has the capability of turning into many specialized cell types.
The very earliest cells produced after fertilization are called totipotent because they have the capability of turning into any other cell type in the body as well as extra-embryonic cell types such as those which form the placenta. As cells divide and begin to specialize (a process called differentiation), the stem cells along these pathways lose the ability to produce certain types of cells. At this point they are called pluripotent in that they can still develop into all the tissue types of the body but not the extra-embryonic tissue types. After further differentiation, the cells become multipotent, meaning the number of potential cell types that can be derived from them has been reduced. This process continues until cells are only able to produce cells of one type.
Along the way, some stem cells stop differentiating and merely reproduce themselves, thereby giving the body a reservoir of stem cells. These cells then provide a source of new cells for tissue replacement and repair.
Why Are Stem Cells Important?
Medical researchers are interested in stem cells for their potential to treat various diseases. For example, stem cells that could be induced to change into insulin-secreting cells could help cure those with diabetes. Patients with damage to the spinal cord could benefit from new nerve tissue generated from stem cells. Those suffering from heart muscle damage after a heart attack might be able to have new heart muscle derived from stem cells. Think about Parkinson’s disease, multiple sclerosis, Alzheimer’s disease . . . the list of potential interventions based on stem cell therapy seems almost endless.
This research is very important. It is certainly one of the most exciting medical advances in our lifetime. Physicians strive to relieve the suffering of their patients. Who among them would not want to have available a means to cure many horrible diseases? This research has so much potential. However, that potential comes with a grave concern. That concern revolves around how we obtain these stem cells.
How Are Stem Cells Obtained?
In order to understand the basis of the debate over stem cell research, one must understand that there are two basic types of stem cells: embryonic stem cells (ESC) and adult stem cells (ASC).
Embryonic stem cells are, as you would expect from their name, derived from embryos. Four to five days after fertilization, the embryo consists of a hollow ball of cells called a blastocyst. It is from this ball of cells that all the body’s tissues are ultimately derived. To harvest embryonic stem cells, the embryo is disrupted (killed) and the cells collected. As this cell harvest occurs very early in development, very little differentiation of the stem cells would have taken place. These cells would be considered pluripotent.
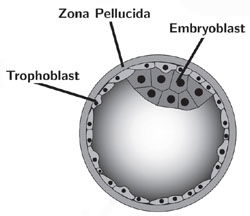
While an embryo must be killed in order to harvest embryonic stem cells, harvesting adult stem cells does not lead to the death of the donor.
Adult stem cells, on the other hand, are not necessarily derived from “adults.” This is somewhat confusing to many as adult stem cells can be obtained from any fully formed person, whether newborn, infant, child, or adult. These cells can be found in many tissues in the body: bone marrow, skin, teeth, liver, brain, intestines, blood vessels, skeletal muscle, among others.
The concern regarding adult stem cells is that these cells could have limitations on the types of tissues that could be obtained from them. In theory, these stem cells are further down the path of differentiation (they would be multipotent rather than pluripotent), thus limiting their potential usefulness in medical research and intervention.
How Are Stem Cells Used?
After being isolated, stem cells are then grown in laboratory culture. They are placed in dishes containing a special culture medium. The cells divide and multiply. The initial phase of the process would be designed to grow an adequate supply of the stem cells themselves.
So how are different tissue types generated from stem cells? There are many different methods used to cause a stem cell to differentiate into a specific cell type. Manipulation of the culture medium can guide this process. Other research techniques include hormonal stimulation or genetic modification of the stem cells. This is still an area of intensive investigation, with new techniques becoming available seemingly every few months.
As an example, let’s select a patient who has suffered a heart attack. A portion of the heart muscle has been damaged or killed as a result of this event. It would certainly be to the patient’s benefit to be able to repair the heart muscle. In this situation, the patient’s medical team might choose to intervene with stem cell therapy. Stem cells could be induced to differentiate into heart muscle cells. These cells could then be administered to the patient in hopes of improving the function of the damaged heart. This type of intervention is, in fact, taking place at this time.2 The results have been very promising thus far.
Depending on the particular situation, the stem cells might be given intravenously,3 by injection or direct deposit of the cells into the target site, given by intracoronary injection (for cardiac intervention), or injection into the spinal fluid (for neurologic problems). The hope for the future is that by using stem cells, entire organs might be grown for transplant. Again, research in this area is promising.
Are There Problems with Stem Cell Treatments?
As with any new research endeavor, there are pitfalls associated with stem cell research. While we tout the successes, we should also be aware of the problems and limitations.
Several major issues have limited embryonic stem cell therapy. First of all, in laboratory animals, embryonic stems cell have shown a tendency to form tumors. The reasons for this are unclear, although some have speculated that ESC can form tumors due to their tendency to associate with each other rather than with the target tissue.4 Obviously, this is an area of intense investigation at present.
The other major issue is that of tissue rejection. As with any transplant, foreign tissue is recognized by the body as “non self.” So an embryonic stem cell transplant from a random donor would be no different than a heart or a kidney transplant. After all, these embryonic stem cells would come from another person. These cells would be seen as foreign tissue by the body. Thus, anti-rejection drugs would be needed to prevent rejection of the new tissue. However, it should be noted that therapeutic cloning using a person’s own cells would avoid this problem.
Adult stem cells apparently do not have the problem of tumor formation. Therapy with ASC would also not have the problem of tissue rejection as long as the stem cells are harvested from the patients themselves.5
That is not to say that intervention with adult stem cells is without problems. The main problem is that even though ASC can be found in many body tissues, they occur in very small numbers and can be quite difficult to isolate. Thus, obtaining an adequate supply of ASC for a given therapeutic intervention can be difficult. The most often claimed problem with adult stem cells is the supposed limitation on the number of tissue types that can be derived from them. Since embryonic stem cells have undergone less initial differentiation, there is the potential to derive all needed cell types from ESC. Thus, it would stand to reason that ASC are more limited.

Image courtesy of Martin Green/Dreamstime.com
Although adult stem cells have their problems, they have produced far more success than embryonic trials. Alzheimer’s and other degenerative diseases are prime candidates for such research.
However, actual research does not bear out these claims. Adult stem cells have, to date, been used to generate almost every different cell type in the body.6 It has been shown that adult stem cells of one cell lineage can be induced to produce cells in another. For example, blood-forming stem cells in bone marrow can differentiate into cardiac muscle cells, etc.7 Some multipotent adult stem cells have been induced to revert to an apparent pluripotent state, and have subsequently produced many more cell types than would have been predicted. So while this may have seemed a problem in theory, it has not been a problem in practice.
Should Both ESC and ASC Research and Treatments Be Pursued?
So should we not pursue any avenue we can to help the sick and the dying? Do we not want stem cell therapy to succeed? Certainly we want to help the sick. We want medical science to progress. But we must also examine the facts and ask the question, “At what cost?”
First of all, what can be said for adult stem cell research? Simply this: adult stem cell therapy has been used to treat over 70 diseases to date. Some stem cell therapies have been used for over 40 years.8 ASC have a proven track record with the hope of greater successes to come. Thus far the only significant clinical interventions available are from using adult stem cells. For some reason the media, when reporting on these issues, has consistently downplayed these successes and even implied that these successes are the result of embryonic stem cell research rather than adult stem cell research.
Embryonic stem cells, on the other hand, despite the regularly reported theoretical benefits, have yet to achieve any significant clinical success. Those in favor of ESC argue that given time these advances will come. Perhaps this is so, but again we need to ask, “At what cost?”
What Is the Cost of Embryonic Stem Cell Research?
Although one could point to the lack of success of embryonic stem cell research, in spite of the years and countless dollars invested in it, and say, “It hasn’t been worth the cost,” the biggest cost is yet to be counted. The cost is that of human life.
As has been noted, embryonic stem cells are obtained by the destruction of an embryo. An embryo is fully human. So in order to get these stem cells to help one person, another person must be killed. This is simply morally unacceptable.
Scripture tells us that life begins at conception (here defined as the moment of fertilization). In His Word, God tells us:
Then the word of the Lord came to me, saying: “ Before I formed you in the womb I knew you; Before you were born I sanctified you; I ordained you a prophet to the nations” (Jeremiah 1:4–5).
Behold, I was brought forth in iniquity, And in sin my mother conceived me (Psalm 51:5).
For You formed my inward parts; You covered me in my mother’s womb. I will praise You, for I am fearfully and wonderfully made; Marvelous are Your works, And that my soul knows very well. My frame was not hidden from You, When I was made in secret, And skillfully wrought in the lowest parts of the earth (Psalm 139:13–15).
We are told that the Lord knew us before we were conceived. We have a sin nature in the womb. How could this be if we are not fully human at the time of fertilization?
What’s the Ethical Solution?
As has been shown, stem cell therapy has the potential to alleviate much suffering. It is an avenue of medical research that should be pursued in hopes of building on the successes already achieved. However, in our haste to help the sick, we must not neglect those who cannot speak for themselves. Adult stem cell therapy can allow us to fight disease without the destruction of human life.
Although everyone wants to see such devastating diseases come to an end, we all must realize our work will only lead to a temporary alleviation. Jesus Christ, the true conqueror of disease and death, will create a new heaven and a new earth where the effects of sin have been removed. That is the cure we eagerly await.
The New Answers Book 3
Do you have answers to the big questions about the Christian faith, evolution, creation, and the biblical worldview?
Read Online Buy BookFootnotes
- www.en.wikipedia.org/wiki/Cloning
- Medical News Today, “First Human Receives Cardiac Stem Cells in Clinical Trial to Heal Damage Caused By Heart Attacks,” July 1, 2009, www.medicalnewstoday.com/articles/155915.php.
- This has the problem of the so-called “first pass effect” where the cells given are filtered out of the circulation by the lungs.
- Joseph Panno, Stem Cell Research (New York, NY: Facts on File, 2005), p. 9.
- ASC therapy using cells from another person would encounter that same rejection potential as ESC.
- “Adult Stem Cell Pluripotency,” www.stemcellresearch.org/facts/ASCRPlasticity.pdf.
- “Stem Cell Basics,” National Institutes of Health, Stem Cell Information, https://web.archive.org/web/20140721004642/http://stemcells.nih.gov/info/basics/pages/basics4.aspx.
- Specifically those involving blood-forming stem cells for bone marrow diseases.

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis

