Radiometric Dating: Back to Basics
Radiometric dating is often used to “prove” rocks are millions of years old. Once you understand the basic science, however, you can see how wrong assumptions lead to incorrect dates.
Radiometric Dating 101
PART 1: Back to Basics
PART 2: Problems with the Assumptions
PART 3: Making Sense of the Patterns
This three-part series will help you properly understand radiometric dating, the assumptions that lead to inaccurate dates, and the clues about what really happened in the past.
Most people think that radioactive dating has proven the earth is billions of years old. After all, textbooks, media, and museums glibly present ages of millions of years as fact.
Yet few people know how radiometric dating works or bother to ask what assumptions drive the conclusions. So let’s take a closer look and see how reliable this dating method really is.
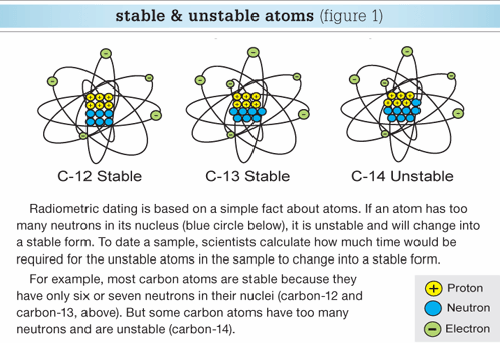
Atoms—Basics We Observe Today
Each chemical element, such as carbon and oxygen, consists of atoms. Each atom is thought to be made up of three basic parts.
The nucleus contains protons (tiny particles each with a single positive electric charge) and neutrons (particles without any electric charge). Orbiting around the nucleus are electrons (tiny particles each with a single negative electric charge).
The atoms of each element may vary slightly in the numbers of neutrons within their nuclei. These variations are called isotopes of that element. While the number of neutrons varies, every atom of any element always has the same number of protons and electrons.
So, for example, every carbon atom contains six protons and six electrons, but the number of neutrons in each nucleus can be six, seven, or even eight. Therefore, carbon has three isotopes (variations), which are specified carbon-12, carbon-13, and carbon-14 (Figure 1).
Radioactive Decay
Some isotopes are radioactive; that is, they are unstable because their nuclei are too large. To achieve stability, the atom must make adjustments, particularly in its nucleus. In some cases, the isotopes eject particles, primarily neutrons and protons. (These are the moving particles measured by Geiger counters and the like.) The end result is a stable atom, but of a different chemical element (not carbon) because the atom now has a different number of protons and electrons.
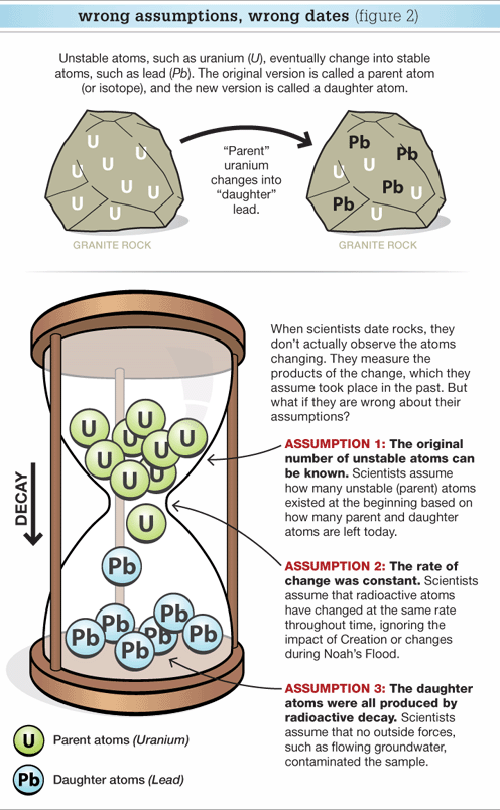
This process of changing one element (designated as the parent isotope) into another element (referred to as the daughter isotope) is called radioactive decay. The parent isotopes that decay are called radioisotopes.
Radiometric dating is based on an observable fact of science: unstable atoms will break down over a measurable period of time.
Actually, it isn’t really a decay process in the normal sense of the word, like the decay of fruit. The daughter atoms are not lesser in quality than the parent atoms from which they were produced. Both are complete atoms in every sense of the word.
Geologists regularly use five parent isotopes to date rocks: uranium-238, uranium-235, potassium-40, rubidium-87, and samarium-147. These parent radioisotopes change into daughter lead-206, lead-207, argon-40, strontium-87, and neodymium-143 isotopes, respectively. Thus geologists refer to uranium-lead (two versions), potassium-argon, rubidium-strontium, or samarium-neodymium dates for rocks. Note that the carbon-14 (or radiocarbon) method is not used to date rocks because most rocks do not contain carbon.
Chemical Analysis of Rocks Today
Geologists can’t use just any old rock for dating. They must find rocks that have the isotopes listed above, even if these isotopes are present only in minute amounts. Most often, this is a rock body, or unit, that has formed from the cooling of molten rock material (called magma). Examples are granites (formed by cooling under the ground) and basalts (formed by cooling of lava at the earth’s surface).
The next step is to measure the amount of the parent and daughter isotopes in a sample of the rock unit. Specially equipped laboratories can do this with accuracy and precision. So, in general, few people quarrel with the resulting chemical analyses.
It is the interpretation of these chemical analyses that raises potential problems. To understand how geologists “read” the age of a rock from these chemical analyses, let’s use the analogy of an hourglass “clock” (Figure 2).
In an hourglass, grains of fine sand fall at a steady rate from the top bowl to the bottom. After one hour, all the sand has fallen into the bottom bowl. So, after only half an hour, half the sand should be in the top bowl, and the other half should be in the bottom bowl.
Suppose that a person did not observe when the hourglass was turned over. He walks into the room when half the sand is in the top bowl, and half the sand is in the bottom bowl. Most people would assume that the “clock” started half an hour earlier.
By way of analogy, the sand grains in the top bowl represent atoms of the parent radioisotope (uranium-238, potassium-40, etc.) (Figure 2). The falling sand represents radioactive decay, and the sand at the bottom represents the daughter isotope (lead-206, argon-40, etc).
When a geologist tests a rock sample, he assumes all the daughter atoms were produced by the decay of the parent since the rock formed. So if he knows the rate at which the parent decays, he can calculate how long it took for the daughter (measured in the rock today) to form.
But what if the assumptions are wrong? For example, what if radioactive material was added to the top bowl or if the decay rate has changed? Future articles will explore the assumptions that can lead to incorrect dates and how the Bible’s history helps us make better sense of the patterns of radioactive “dates” we find in the rocks today.
Answers Magazine
July – September 2009
How do I deal with cancer? Why did God allow evil in the world? Why do snakes appear designed to kill? God’s curse is a historical reality, and we can see its results in everyday life. Don’t miss this special issue, which will give you the tools to tackle questions about evil and its true origin.
Browse IssueRecommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis