Radiohalos—The Mysterious Vanishing Bullets
Part Two
Geologists have uncovered a great mystery in granite rocks. They find tiny black circles, known as radiohalos. They were caused by radioactive decay of polonium, but the source has disappeared. Where did it come from, and where did it go? The only answer is a global Flood.
Radiohalos—The Flood’s Smoking Gun
Part one of this three-part series described the mysterious “halos” found inside granite (the common speckled rocks used for kitchen countertops and tombstones). These microscopic halos look a lot like “bullet holes.” Why are they of interest to creationists?
Once you learn more about these features, you will see why they mystify geologists. They are hard to explain if granite formed slowly over millions of years, but they make perfect sense if molten material rose near the earth’s surface during the worldwide Flood and quickly hardened into granite.
But first, you will need to understand a bit more about radioactive decay and its effects on rocks.
The Well-Understood Radioactive Decay of Uranium
Geologists have a clear understanding of the radioactive decay of uranium atoms and the decay’s effects on surrounding rocks. We can observe this process today. It is not a mystery.
As explained in the first article, the nuclei of uranium atoms are so large that they are very unstable. As the atoms decay, subatomic alpha particles fly out like bullets, damaging the surrounding material. These bullets produce spherical halos called radiohalos (an abbreviation for “radioactive halos”).
After the first set of these particles is ejected, the smaller nucleus is still unstable. So more particles are ejected from the nucleus. This happens repeatedly until the atom is stable and no longer decays. In what is known as the uranium decay chain, the nucleus of the original uranium-238 atom typically undergoes eight alpha-particle changes until it becomes the stable lead-206 atom (see “The Uranium Decay Chain—The Source of Radiohalos” below).
The Uranium Decay Chain—The Source of Radiohalos
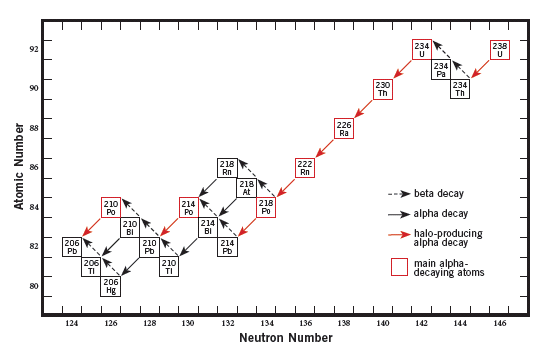
All the intermediate steps during the radioactive decay of uranium-238 are shown, resulting in the final, stable element lead-206. Where the arrows point downward, those changes result from alpha decay. The arrows pointing upward indicate beta decay. (Alpha decay involves the loss of two protons and two neutrons; beta decay involves splitting of a neutron, the loss of an electron, and the gain of a proton.)
The original (or parent) uranium nucleus contains 92 protons. That’s why uranium is element number 92 on the periodic chart. However, after two protons are ejected in an alpha particle, the nucleus then has only 90 protons, so the uranium has changed into element number 90, which is thorium.
The nucleus of thorium is still unstable, so it decays radioactively. However, it decays by successively splitting and ejecting two electrons, known as beta particles, while gaining two extra protons, giving it a total of 92 again. So the thorium has changed back into the element number 92, uranium.
But this uranium is now four neutrons lighter. Whereas the original parent uranium atom had 146 neutrons and 92 protons in its nucleus (called uranium-238), this uranium atom has only 142 neutrons and 92 protons (called uranium-234).
The nucleus of this uranium-234 atom is still unstable, so it decays. The process continues through what is known as the uranium decay chain, until the stable lead-206 atom is reached, the final end product.
The picture is a little more complicated at the bottom end of this decay chain. At polonium-218 there is “branching,” but most polonium-218 atoms decay to lead-214. Then they quickly decay to bismuth-214, then polonium-214. The main decay path is depicted with solid red arrows. And the main eight alpha-decaying atoms are shown in red.
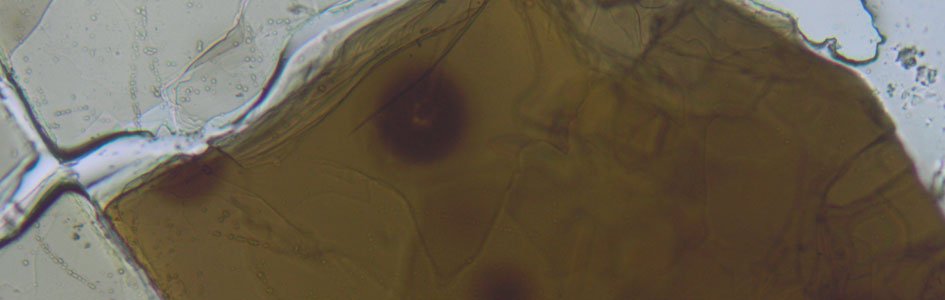
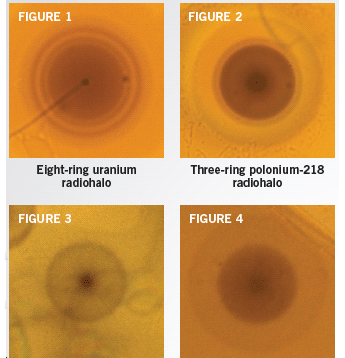
Figure 1 shows a sample radiohalo that resulted from this process. Notice the eight dark rings. Why eight and not just one? As each of the alphaparticles is “fired” from the nucleus, it has a different energy. Consequently each bullet travels a different distance into the surrounding material before it stops to leave a black mark.
The Mystery of Polonium Radiohalos
The origin of eight-ring radiohalos is not a mystery for geologists. They must have formed from uranium-238 decay. The mystery is the source of the one-ring, two-ring, and three-ring radiohalos that are found in the same rock specimens (Figures 2–4).
Photos courtesy Mark Armitage
Geologists find four types of radiohalos in granite. One was caused by the decay of uranium, and the others came from the decay of another radioactive element, polonium.
The source of eight-ring radiohalos is still there, but nothing is usually visible at the center of other types of radiohalos.
We can see the source of the eight-ring radiohalos because it is still there—a zircon crystal that still hosts uranium. But nothing is usually visible at the center of the other types of radiohalos. The source has vanished!
Fortunately, it is still possible to determine the source of the rings. By carefully measuring the distance from the center of the radiohalo to each ring, we can identify which type of nucleus formed each ring (Figure 5).1 In each case, the “smoking gun” was a variation of the radioactive element polonium.2 Polonium-218, polonium-214, and polonium-210 generate exactly the right amount of energy to produce the three-ring, two-ring, and one-ring radiohalos.
The problem is that polonium is never found alone in rocks. It is a rare, unstable element that appears quickly during the decay of uranium and then decays into stable elements, such as lead. The only possible source of the polonium was the decay of uranium. But we do not find a uranium source at the center of the one-, two-, and threering radiohalos! The crucial clue is the appearance of polonium during the uranium-238 decay chain. As the uranium atom’s nucleus becomes progressively smaller, three variations of polonium appear briefly in sequence at the end of the chain.
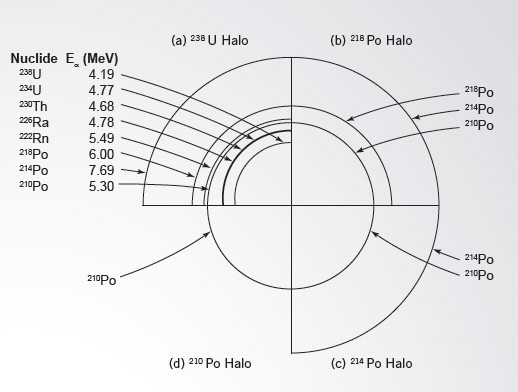
FIGURE 5—Composite schematic drawing of the four types of radiohalos: (a) a uranium-238 radiohalo, (b) a polonium-218 radiohalo, (c) a polonium-214 radiohalo, and (d) a polonium-210 radiohalo. Different types of atoms (nuclides) cause each ring in the radiohalos, and the particles that they send out have different energies (MeV).
A Possible Source of the Polonium Radiohalos
We can be certain that no other radioactive element appeared at the center of the polonium radiohalos. If any other elements had been there, such as uranium or radon, they would have formed other rings as they decayed.
So where did the polonium atoms come from?
The best possible answer is that the polonium traveled from a nearby source where uranium atoms were decaying. Is there such a nearby uranium source? Absolutely! The same flakes that host the polonium radiohalos usually contain uranium radiohalos, usually less than a fraction of an inch from polonium radiohalos (Figure 6).
Finding the Source of Polonium Radiohalos
The source of uranium radiohalos still survives in granite rocks: tiny zircon crystals. But geologists are mystified because the source of polonium radiohalos is missing. The answer lies only a tiny way up the biotite sheet . . . the zircons!
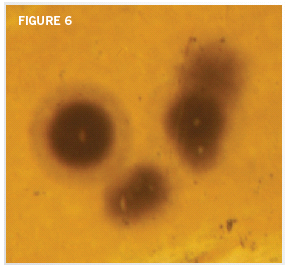
Photo courtesy Andrew Snelling
Biotite flakes often have numerous polonium radiohalos, in addition to uranium radiohalos.
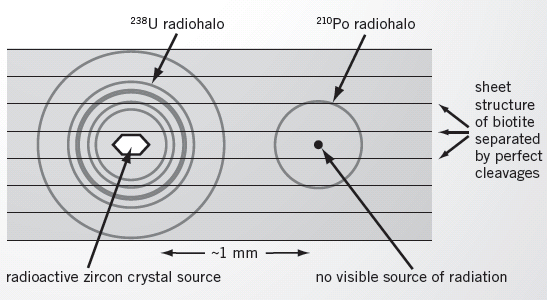
FIGURE 7—Diagram of a cross-section through a biotite flake where radiohalos are found. The uranium-238 in the zircon crystal generated the uranium-238 radiohalo. Water flowing past the crystal carried decaying atoms along the same sheet to a nearby location, where a polonium-210 radiohalo developed. Nothing remains at the center of this radiohalo, however, because it dissolved quickly.
Yet under normal conditions, like those we see in the earth today, that migration would be impossible. To understand the magnitude of the problem, you need to understand how many polonium atoms had to migrate and how quickly they had to travel.
Scientists have estimated that each radiohalo’s discoloration initially develops after 100 million alpha-particles have been emitted from its center. It does not become dark until 500 million particles, and it does not become very dark until one billion alpha particles have been ejected.3 This means that each polonium radiohalo needs at least 500 million polonium atoms. And many polonium radiohalos often appear in the same specimen, so many billions of polonium atoms had to move into position to explain the radiohalos we see.
Then there is the problem of how fast they must be moved. Polonium-214 atoms decay so quickly they are gone in the blink of an eye! Polonium-218 has a half-life (decay rate) of only 3.1 minutes, while polonium-214 decays in 164 microseconds!4 By comparison polonium-210 atoms are long-lived, with a half-life of 138 days. What unusual forces could have carried so many atoms away from the uranium source so quickly?
Another possibility is to move another element in the uranium-238 decay chain that appears before polonium. For example, the element immediately before the polonium-218, polonium-214, polonium-210 sequence is radon-222. If radon-222 moved into place, then the polonium would not have to be transported. But this presents two problems. First, the travel time is still short: the half-life of radon-222 is only 3.8 days. The radon would still have to be transported from the zircons to the polonium halo sites in only days.
Second, we don’t find a radon-222 decay halo in the polonium radiohalos. Somehow the polonium had to separate from the radon-222 and then concentrate in what would become the polonium radiohalo centers. The location of radiohalos and the different chemical properties of radon and polonium suggest a solution to these problems.
How Polonium Radiohalos Likely Formed
When uranium and polonium radiohalos are observed under the microscope, their radiocenters are actually on the same sheet.5 As explained in the first article, radiohalos are found in the dark flecks within granites, called biotite. These biotite flakes consist of layers and layers of crystal sheets, which can be pulled apart and examined under a microscope.
Water can easily travel down the spaces between these sheets (Figure 7). Changes in the biotite’s color are often evidence that water has seeped between the sheets. That’s the key to the creationist explanation for polonium radiohalos.
When granites crystallize and are cooling, hot waters are left over. This water is capable of seeping along the spaces between the stacked sheets of biotite. As they pass by the zircon, these waters could dissolve any radon-222 atoms that had leaked out of the zircon crystals and transport them between the sheets of the biotite flake.
Though they are very tiny, polonium radiohalos have a huge message that cannot be ignored.
Radon-222 atoms are chemically inert (they do not combine with other atoms). But once they decay, the newly formed polonium-218 atoms would readily combine with other atoms, such as chlorine or sulfur atoms that had dissolved in the hot water and were flowing between the biotite sheets. Polonium chlorides and polonium sulphides don’t dissolve well in water, so as soon as the polonium combines with these other atoms, the molecules drop out of the water. There the polonium would start forming polonium radiohalos.
The water would continue to move many radon-222 atoms past the forming radiocenters, providing a continual supply of new polonium atoms.
Ever since a geologist dismissed this evidence of the Flood, calling it “a very tiny mystery” at a 1981 trial on teaching creationism in Arkansas schools,6 scientists have ignored this mystery. The problem is that secular geologists believe the host granites took millions of years to form, but the process had to be much faster—only hours or weeks—for polonium radiohalos to appear. They shouldn’t exist, according to conventional wisdom! The next and final article will look at the profound implications for all of geology.
Though they are very tiny, polonium radiohalos have a huge message that cannot be ignored. These amazing testimonies to the Flood are found in granites all around the world. And they point to a catastrophic origin for granites, consistent with the biblical timeframe for earth history and God’s judgment during the Flood.
Related Videos
Radiohalos Excerpt
Answers Magazine
July – September 2012
It’s impossible to explain how even one creature could evolve by chance, but in fact all life needs a complete community of other organisms to survive. The web of life had to be in place from the very start for any of us to exist. What an amazing testimony to the Creator! Also learn about animals that seem to come back from the dead, robotics, and how archaeology in Jordan confirms the Bible’s history.
Browse IssueFootnotes
- R. V. Gentry, “Radioactive Halos,” Annual Review of Nuclear Science 23 (1973): 347–362.
- The element polonium is a rare metal discovered in 1898 by Marie Curie (with her husband, Pierre) and named by her after her homeland, Poland.
- R. V. Gentry, Creation’s Tiny Mystery (Knoxville, Tennessee: Earth Science Associates, 1988), p.19.
- A. A. Snelling, “Radiohalos” in Radioisotopes and the Age of the Earth: A Young-Earth Creationist Research Initiative, L. Vardiman, A. A. Snelling, and E. F. Chaffin, eds. (El Cajon, California: Institute for Creation Research, and St. Joseph, Missouri: Creation Research Society, 2000), pp. 381–468. A half-life of 3.1 minutes means that half of the material is lost in 3.1 minutes; the next half is lost in another 3.1 minutes. So in 6.2 minutes, only one-fourth of the original polonium-218 atoms are left. After ten half-lives, less than a thousandth of the original polonium-218 is left. So the polonium-218 has only ten half-lives to move before it all changes. (That means the polonium-218 has a bit over a half hour. Polonium-214 has 1.64 milliseconds or 1/500th of a second. Polonium-210 has a bit less than 4 years.)
- A. A. Snelling, “Radiohalos in Granites: Evidence for Accelerated Nuclear Decay,” in Radioisotopes and the Age of the Earth: Results of a Young-Earth Creationist Research Initiative, L. Vardiman, A. A. Snelling, and E. F. Chaffin, eds. (El Cajon, California: Institute for Creation Research, and Chino Valley, Arizona: Creation Research Society, 2005), pp. 101–207.
- Dr. G. Brent Dalrymple, then deputy director of the U.S. Geological Survey and more recently at the Berkeley Geochronology Center at the University of California Berkeley, dismissed the polonium radiohalos as “a very tiny mystery” while in the witness stand at the 1981 Arkansas “Creation Trial,” as reported by R. V. Gentry, Creation’s Tiny Mystery (Knoxville, Tennessee: Earth Science Associates, 1988), p. 122.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis