A Creationist Puzzle
50,000-Year-Old-Fossils
Evolutionists aren’t the only ones who run into challenges when trying to reconcile radiocarbon dating with their view of history. How do creationists explain dates of 50,000 years?
Conventional geologists claim that fossils, coals, and diamonds are millions to billions of years old. Yet it has now been firmly established that they still contain measurable amounts of radiocarbon, which has a half-life (decay rate) of only 5,730 years.1
This creates a dilemma for conventional geology, as explained in Part 2 of this series.2 Absolutely no radiocarbon should be left in fossils, coals, and diamonds, because after just one million years it should have decayed away.
Carbon-14 Dating
Part 1 Understanding the BasicsPart 2 An Evolution Dilemma
Part 3 A Creationist Puzzle
Yet the radiocarbon in these fossils, coals, and diamonds equates to “ages” of up to 55,000 years. This is much older than the biblical time frame of earth history, which attributes most fossils and coals to the global Flood of Noah’s day, about 4,350 years ago. What should Bible-believing Christians think about this apparent discrepancy?
Assumptions Change Estimate of Age
To solve this puzzle it is necessary to review the assumptions on which radiocarbon dating is based. These include:
- The production rate of carbon-14 has always been the same in the past as now.
- The atmosphere has had the same carbon-14 concentration in the past as now.
- The biosphere (the places on earth where organisms live) has always had the same overall carbon-14 concentration as the atmosphere, due to the rapid transfer of carbon-14 atoms from the atmosphere to the biosphere.3
None of these assumptions is strictly correct, beyond a rough first approximation. Indeed, scientists have now documented that the atmosphere’s concentration of carbon-14 varies considerably according to latitude. They have also determined several geophysical causes for past and present fluctuations in carbon-14 production in the atmosphere.4
Specifically, we know that carbon-14 has varied in the past due to a stronger magnetic field on earth and changing cycles in sunspot activity. So when objects of known historical dates are dated using radiocarbon dating, we find that carbon-14 dates are accurate back to only about 400 BC.
The conventional scientific community ignores at least two factors that are crucial to recalibrating radiocarbon (so that it accounts for major changes in the biosphere and atmosphere that likely resulted from the Flood): (1) The earth’s magnetic field has been progressively stronger going back into the past, and (2) the Flood destroyed and buried a huge amount of carbon from the pre-Flood biosphere.
The Effect of a Past Stronger Magnetic Field
The evidence for the earth’s having a progressively stronger magnetic field in the past is based on reliable historical measurements5 and “fossil” magnetism trapped in ancient pottery.6, 7
A stronger magnetic field is significant because the magnetic field partly shields the earth from the influx of cosmic rays, which change nitrogen atoms into radioactive carbon-14 atoms. So a stronger magnetic field in the past would have reduced the influx of cosmic rays.
This in turn would have reduced the amount of radiocarbon produced in the atmosphere. If this were the case, the biosphere in the past would have had a lower carbon-14 concentration than it does today.
The best estimates indicate that the earth’s magnetic field was twice as strong 1,400 years ago, and possibly four times as strong 2,800 years ago. If this is true, the earth’s magnetic field would have been much stronger at the time of the Flood, and the carbon-14 levels would be significantly smaller.
So if you mistakenly assume that the radiocarbon levels in the atmosphere and biosphere have always been the same as they are today, you would erroneously estimate much older dates for early human artifacts, such as post-Babel wooden statuettes in Egypt. And that is exactly what conventional archaeology has done.
The Effect of More Carbon in the Pre-Flood Biosphere
An even more dramatic effect on the earth’s carbon-14 inventory would be the destruction and burial of all the carbon in the whole biosphere at the time of the Flood. Based on the enormous size of today’s coal beds, oil, oil shale, natural gas deposits, and all the fossils in limestones, shales, and sandstones, a huge quantity of plants and animals must have been alive when the Flood struck. It is conservatively estimated that the amount of carbon in the pre-Flood biosphere may have been many times greater than the amount of carbon in today’s biosphere.8
It is conservatively estimated that the amount of carbon in the pre-Flood biosphere may have been many times greater than the amount today.
We cannot yet know for certain how much radiocarbon (carbon-14) was in this pre-Flood carbon (a mixture of normal carbon-12 and carbon-14). Yet if the earth’s atmosphere started to produce carbon-14 (14C) at the Fall, then many radiocarbon atoms could have been in the pre-Flood biosphere by the time of the Flood, about 1,650 years after Creation.
However, if there was a whole lot more normal carbon (carbon-12, or 12C) in the pre-Flood biosphere, then the proportion of 14C to 12C would have been much less than the proportion in today’s biosphere.
So when scientists fail to account for so many more plants and animals in the pre-Flood biosphere and wrongly assume that plants buried in coal beds had the same proportion of carbon-14 as plants do today, their radiocarbon dating yields “ages” much higher than the true Flood age of about 4,350 years.
A Prediction Fulfilled
Now if this model of the earth’s past radiocarbon inventory is correct, then a logical prediction follows. Since all pre-Flood plants would have had the same low radiocarbon levels when they were buried, and they all formed into coal beds during that single Flood year, then those coal beds should all have the same low radiocarbon content.
They do! Samples from coal beds around the United States, ranging from Eocene to Pennsylvanian deposits, supposedly 40–320 million years old, all contain the same low radiocarbon levels equivalent to “ages” of 48,000–50,000 years.9
This makes sense only if these coal beds were all formed out of pre-Flood plants during the year-long Flood, about 4,350 years ago. Carbon-14 dates of the same value are expected in creation theory but contrary to the expectations of conventional old-earth theory.
The “Puzzle” Is Being Solved
So the radiocarbon “puzzle” can be solved, but only in the biblical framework for earth history. Research is therefore underway to find a means of recalibrating the radiocarbon “clock” to properly account for the Flood and its impact on dates for the post-Flood period to the present.
For example, conventional radiocarbon dating gives an age of “48,000 years” for a coal bed deposited during the Flood, about 4,350 years ago. This could be explained if the 14C/12C ratio at the time of the Flood was only 1/200th the ratio of the present world.
If scientists assume the ratio is 200 times greater than it really was, then their radiocarbon age estimate would be exaggerated by 43,650 years.10
In reality, calculations (described above) have led to estimates that the pre-Flood biosphere may have had more than 100 times the carbon-12 as the present earth. Using this information, we may be able to calculate how much carbon-14 was actually on the early earth at the Flood. This, in turn, would allow us to develop a proper interpretation of all carbon-14 dates.
Once the research is completed, one of the many exciting benefits is that it should be possible to begin more accurately dating any archeological artifact within the true chronology found in God’s Word.
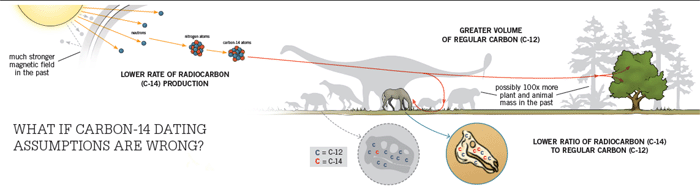
Radiocarbon dating of fossils compares the amount of radioactive carbon atoms (C-14) to regular carbon atoms (C-12). Conventional dating methods assume the past ratio based on current levels. But what if these assumptions are wrong?
Lower Rate of Radiocarbon (C-14) Production
Cosmic rays bombard the earth’s atmosphere and produce neutrons. These neutrons collide with nitrogen atoms, changing them into radioactive carbon atoms (C-14).
Conventional dating assumes radiocarbon (C-14) production has remained stable. But the earth’s magnetic field, which protects the earth from cosmic rays, was once several times stronger than it is today. So we would expect much less radiocarbon to be produced in the past. That would result in much less C-14 compared to C-12.
Greater Volume of Regular Carbon (C-12)
Plants absorb carbon atoms during photosynthesis (mostly regular C-12 and little radioactive C-14). With a limited amount of radiocarbon to go around, more plants would mean less radiocarbon per plant.
Coventional dating assumes the volume of plants and animals in the world has remained relatively stable. But the abundance of fossils indicates that the pre-Flood world’s shallow seas and temperate climate supported much more plants and animals (containing mostly C-12) than today.
Lower Ratio of Radiocarbon (C-14) to Regular Carbon (C-12)
Radiocarbon begins to break down after plants and animals die. The amount of radiocarbon remaining determines the time that has passed. Conventional dating assumes the ratio of C-12 to C-14 was the same in animals in the past. But if the ratio was much lower in the animals in the past, then those animals would have much less radiocarbon to break down after they died. This would result in much younger dates than conventional methods assume.
Related Videos
Radiometric Dating
Answers Magazine
April – June 2011
Continual attacks on God’s Word can cause even Christians to question the Bible. Are you prepared to answer the attacks, or more important, questions from those who honestly want to learn how we know God’s Word is true? This issue of Answers will burrow down to the heart of the matter, highlighting seven reasons we know the Bible is true!
Browse IssueFootnotes
- A. A. Snelling, “Radiocarbon Dating Part 1: Understanding the Basics,” Answers (Oct.–Dec. 2010), pp. 72–75.
- A. A. Snelling, “Radiocarbon Dating Part 2: Carbon–14 in Fossils and Diamonds—An Evolution Dilemma,” Answers (Jan.–Mar. 2011), pp. 72–75.
- S. Bowman, Interpreting the Past: Radiocarbon Dating (London: British Museum Publications, 1990), p. 14.
- Bowman, ref. 3, pp. 16–30; G. Faure and T. M. Mensing, Isotopes: Principles and Applications, 3rd edition (Hoboken, New Jersey: John Wiley & Sons, 2005), pp. 614–625; A. P. Dickin, Radiogenic Isotope Geology, 2nd edition (Cambridge: Cambridge University Press, 2005), pp. 383–398.
- T. G. Barnes, “Decay of the Earth’s Magnetic Field and the Geochronological Implications,” Creation Research Society Quarterly 8.1 (1971): 24–29.
- D. R. Humphreys, “Reversal of the Earth’s Magnetic Field during the Genesis Flood,” in Proceedings of the First International Conference on Creationism, ed. R. E. Walsh, C. L. Brooks and R. S. Crowell, (Pittsburgh, Pennsylvania: Creation Science Fellowship, 1986), 2:113–126. What is “fossil” magnetism? The clay used to make pottery often contains mineral grains that are slightly magnetic. When the clay is baked, the grain’s magnetic field imprint at the time is “locked in” or fossilized.
- The strength of the earth’s magnetic field was not affected by field reversals. The sun also regularly experiences field reversals without loss of strength in the magnetic field. D. R. Humphreys, “Physical Mechanism for Reversal of the Earth’s Magnetic Field during the Flood, in Proceedings of the Second International Conference on Creationism, ed. R. E. Walsh and C. L. Brooks (Pittsburgh, Pennsylvania: Creation Science Fellowship, 1990), 2: 129–142.
- R. H. Brown, “The Interpretation of C-14 Dates,” Origins 6 (1979): 30–44; J. R. Baumgardner, A. A. Snelling, D. R. Humphreys, and S. A. Austin, “Measurable 14C in Fossilized Organic Materials: Confirming the Young Earth Creation-Flood Model,” in Proceedings of the Fifth International Conference on Creationism, ed. R. L. Ivey, Jr. (Pittsburgh, Pennsylvania: Creation Science Fellowship, 2003), pp. 127–147.
- J. R. Baumgardner, “14C Evidence for a Recent Global Flood and a Young Earth,” in Radioisotopes and the Age of the Earth: Results of a Young-Earth Creationist Research Initiative, ed. L. Vardiman, A. A. Snelling, and E. F. Chaffin (El Cajon, California: Institute for Creation Research, and Chino Valley, Arizona: Creation Research Society, 2005), pp. 587–630; D. B. DeYoung, Thousands . . . Not Billions: Challenging an Icon of Evolution, Questioning the Age of the Earth (Green Forest, Arkansas: Master Books, 2005), pp. 45–62.
- These numbers are calculated in terms of half-lives, discussed in this series’ previous article. If the modern ratio is 200 times greater than the ratio at the Flood, the error ends up being 7.618 carbon-14 half-lives, or 43,650 years!

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis