The Mystery of the Exploding Beetle
When Dr. Andy McIntosh investigated the bombardier beetle, he discovered explosive evidence of God’s intricate design.
In 1903 the Wright brothers succeeded with controlled powered flight because they asked the right question: “How do birds use their wings?” Steve Jobs, founder of Apple, wondered how we could hold a computer in the palm of our hand. He succeeded with the iPhone because he asked the right question.
History is filled with engineers who asked the right question. From airplanes to smartphones, we couldn’t imagine our lives without these modern inventions. While the names and technology above might be familiar to you, our story involves someone you might not know—Dr. Andy McIntosh. And the mystery he is trying to solve is on a much smaller scale than airplanes or smartphones.
Andy has spent over 40 years in the fields of thermodynamics and engineering. For nearly 20 of those years, he’s focused his attention on a little insect whose explosive tendencies have inspired exciting research and discoveries, all pointing to the Creator.
How Can That Be?

Photo courtesy Dr. Andy McIntosh
As an engineer, Dr. Andy McIntosh studies the bombardier beetle from a unique perspective.
A seemingly small incident in 2001 changed Andy’s life. He was sitting in his office at Leeds University in England where he had been conducting research for 15 years (and would continue for another decade). While reading a copy of the Proceedings of the Natural Academy of Sciences, he noticed an article about the bombardier beetle, an insect that blows bursts of boiling water and chemicals out its rear end.
Looking at high-speed photos of a bug blasting chemicals from its behind might fascinate most of us for only a few minutes, but not Andy. Someone with a doctorate in combustion theory doesn’t look at the world the way we do. He knew that there must be more to the story.
Biologists have known about the beetle since the early 1800s, when the first reports were published about beetles shooting “artillery.” Later in the 1960s and 1970s, the world’s leading expert on the bombardier beetle, entomologist Thomas Eisner, made some exciting discoveries about the beetle’s complex chemistry, but many mysteries remained.
What caught Andy’s attention in the new report was the obvious evidence of combustion, his area of expertise. Something amazing must be going on for an insect to set off a series of explosions and then to machine gun its enemies.
Andy wasn’t interested in the bombardier beetle like a biologist might be. He was interested in the engineering and physics. As those who believe in creation, we know that God made this sophisticated chemical system (which includes specialized chemicals that make the reaction go faster), along with a combustion chamber, a moveable exhaust turret (more versatile than a tank turret), the inlet and exhaust valves, and a sensory mechanism to determine from which direction the attack may be coming. Andy wondered if it were possible that the Designer God had implemented some unique engineering solutions to miniaturized explosions that human industry might learn from and imitate (biomimicry) for the good of our fellow man.
We may not know the explosive jet’s purpose in the perfect world before Adam’s fall, but Andy wanted to know more about the engineering applications today. He visited his university’s biology department to see if anyone would be interested in learning more about the mechanics of this beetle’s unique weapon system.
To his surprise, one of the biologists was not only uninterested but also questioned why Andy would bother. “What do you hope to learn, since the beetle is still evolving?”
“It was precisely because I believed in creation that I was spurred to ask the right questions.”
The lack of curiosity shocked Andy. “I am interested in how things work. Since I knew the master Designer designed animals, I expected to discover new insights into combustion and engineering. Rather than being a hindrance, my belief in God’s creation opened a new research field. It was precisely because I believed in creation that I was spurred to ask the right questions.”
He reached out to the author of the bombardier beetle paper, Dr. Eisner, who was working at Cornell University. Little did Andy know what doors this decision would open for major research, which continues to this day, including new discoveries and patents.
The Work Begins
For about six years, Andy worked off and on with Eisner at his Cornell University laboratory in Ithaca, New York. Eisner had access to electron microscopes that could take detailed images of the beetle’s internal organs. Andy still remembers the light-bulb moment he had during a visit with Eisner in March 2004.
Biologists had long known that the beetle has an inlet valve controlling the flow of chemicals into the reaction chamber where the explosion occurs. But the mystery was that the chemicals don’t naturally produce an explosion as strong as the blast captured in Eisner’s images. The jet of steam and noxious chemicals (benzoquinones) fire repeatedly through nozzles, at a speed of up to 65 feet (20 m) per second.
Then in discussions with Eisner in his laboratory, Andy discovered the secret. When Eisner showed him scanning electron microscope images of the beetle’s anatomy, Andy realized that it had another valve at the outlet. If the beetle can keep the chamber closed long enough, the pressure will build up without the water turning to steam (sort of like a pressure cooker).
There it sat, where it had been all along—the membrane that served as an outlet valve. But nobody had realized its function before. It is generally limp under a microscope, like a deflated balloon. Up until this point, even Eisner hadn’t considered that this was a valve. But after seeing the detailed imaging, Eisner agreed that the membrane was functioning in this way. So Andy had discovered that the beetle’s blaster is a two-valve system, not a one-valve system, “an example of exquisite engineering,” as he called it.
The other missing piece of the puzzle was whether a special kind of explosion, called a steam explosion, could account for the rapid ejection of the spray that Eisner had found. To uncover that answer, Andy needed to identify the precise nature of the mixture the beetle released. How much of it was steam, and how much was liquid water and the noxious chemicals?
The beetle’s basic chemical cocktail has long been known: hydrogen peroxide and hydroquinone. And scientists know that these chemicals don’t react without a catalyst (a substance that speeds up a chemical reaction). The beetle has these catalysts in abundance: catalase and peroxidase. But how much will turn into steam before the concoction is released as an explosive spray?
Here’s where Andy’s engineering came in, but he needed help from someone who could do advanced computer modeling. So he applied for a grant to hire an assistant, and to his delight the grant was approved. The computer modeler analyzed what should happen if the outlet valve opens at 1.1 bar (1 bar is the atmospheric pressure at sea level). At that pressure the water will reach 221°F (105°C) without boiling. (Water normally boils at 212°F [100°C].) When the pressure is suddenly released, the water will instantly turn to steam in what’s called flash evaporation.
In the computer model, the steam explosion drove the water and steam combination within only two thousandths of a second. At that rate, the spray would eject about 500 cycles per second—exactly what Eisner had found in his experimental observations. So Andy and his assistant knew the computer had correctly simulated the insect’s explosion.
Beetle Bombardiers in Every Clime and Place
Nearly 1,000 known bombardier beetle species come in every shape and color, with an array of arsenals. They are found in clusters under rocks on every continent except Antarctica. Bombardier beetles fall into two subfamilies: “exploding” and “nonexploding” types.
“Exploding” Bombardier Beetles (Brachininae, over 500 species)
These common beetles produce precisely aimed sprays shot through rotating turrets on their rear. These are the most familiar and studied bombardier beetles.

“Nonexploding” Bombardier Beetles (Paussinae, around 400 species)
These beetle species release their hot chemicals through two rearward pipes (flanges) sticking out the sides. The chemicals do not explode and usually come out as a spray.

Mimicking the Beetle Blaster
Andy and his partner published a technical paper and shared their astonishing findings at a conference. So intrigued by their work, an entrepreneur in the audience offered to continue funding the research if they were prepared to build an experimental rig to mimic the beetle’s actions. The entrepreneur’s special interest was biomimicry, and he believed Andy and his team could invent new technology if they took their research a step further.
Andy was delighted, but he needed help. He was a theoretical engineer, more comfortable with mathematical calculations on a chalkboard. He explains, “I never built stuff before, but we had some brilliant staff in the engineering department who were able to both design and build prototypes.”
Andy’s team got to work. Their objective was to build a two-valve delivery system. The main aim was to demonstrate how the spray system worked. They also looked at the effects of varying the pressure, the timing of the spray, the distance traveled, and size of the spray droplets. Their goal was to produce a machine that sent an explosive jet as far as the beetle’s. And in time, they were successful.
Unlike the bombardier beetle’s passive valve system, which automatically opens and closes when the pressure reaches a certain point, Andy’s rig uses advanced electronically controlled valves that a computer opens and closes on command.
Their experimental chamber was about 1 inch (2 cm) long, 20 times bigger than the beetle’s reaction chamber (which is only 5 hundredths of an inch [1 mm]). The beetle can spray 200 times the length of its chamber, easily hitting a nearby ant on the forest floor. Andy and his team were delighted when their rig could spray 200 times its size—13 feet (4 m) across the room.

Photo ©SATOSHI KURIBAYASHI/MINDEN PICTURES
The bombardier beetle (Pheropsophus jessoensis) protects itself by ejecting a noxious chemical spray.
In 2010, Andy and his lab partner received the prestigious Times Higher Education award for “The Outstanding Contribution to Innovation and Technology” in London.
Andy is still pursuing possible applications to industry and has three patents for the three main applications of this invention: injectors for fuel additives in engines (for more efficient burning), pharmaceutical sprays, and fire extinguishers.
Now in retirement of sorts, Andy is working with students in the US at Liberty University’s engineering department to develop a fire protection system that could better protect fire fighters during a wildfire. Andy’s plan is to develop backpacks filled with water that could shoot steam and water spray up to 50 feet (15 m). (See bombardierbeetle.org for the latest on this creationist research project.)
Irreducible Complexity
This beetle cannot blast predators unless all its parts are present and working together.
Back in the 1970s, creationists latched onto the bombardier beetle as a premier example of irreducible complexity, even before Dr. Michael Behe invented the term in his 1996 book Darwin’s Black Box. It refers to a system in which all the parts must be present and working together or else the system fails. Just as a mousetrap won’t snap shut unless all the pieces are working together, this beetle cannot blast predators unless all its parts are present and working together.
Evolutionists try to argue that each individual part can be built stage by stage, but they must show how each of the chemicals offers an advantage by itself. Yet the hydrogen peroxide and hydroquinone are no use as explosives without the catalysts (peroxidase and catalase) to help the chemistry work fast enough.
Well-known atheist Richard Dawkins mocked creationists back in the 1980s and 1990s for popularizing the bombardier beetle (as he still does today). In a lecture to children in 1991, he famously claimed that the bombardier beetle could have easily evolved by gradually adding more and more hydrogen peroxide. This could produce bigger and bigger explosions. But he set aside the hydroquinone, saying it was unimportant. And sure enough, hydrogen peroxide can be mildly explosive in small quantities and with the right catalyst.
But in doing this, Dawkins failed to explain the chemistry of the beetle. The catalytic reaction of hydroquinone is critically important for an effective explosion. No one has shown how the system could evolve slowly. The chemistry is complex, but here are the basics: breaking down hydroquinone produces hydrogen, which then combines with oxygen from the hydrogen peroxide to produce a runaway steam explosion.
Andy concludes, “In every respect, the bombardier beetle is irreducibly complex because the system will not work unless you have the chemistry right, the catalysts right, the inlet valve right, and the outlet valve right. Not to mention that the reaction chamber must be there to begin with or else the beetle will blow itself to bits.”
After watching bombardier beetles in action for nearly 20 years, Andy knows they are one of the most obvious examples of irreducible complexity in all of nature.
In fact, the bombardier beetle’s blaster is so sophisticated in the way it senses and responds to danger, producing chemicals on demand, that scientists still don’t fully understand how all the parts work. For instance, they would love to learn how this beetle produces hydrogen peroxide. If they could figure it out, it could lead to low-cost manufacturing of this essential chemical found in medicine cabinets, hair dyes, and military rockets.
Far from hindering him, Andy’s belief in creation and the Bible has helped him solve problems that nobody else was thinking of—because he asked the right questions.
“As you look at biology and look at nature through Bible-believing eyes, you see things that biologists who are governed by evolutionary thinking often do not see. My belief in the creation perspective opened a whole new area of research.”
An Unlikely Weapon
A toad searching for an afternoon snack spies a beetle sitting on a leaf. But before he can flick out his long, sticky tongue, his face is covered in a cloud of scalding, noxious chemical spray. He has just come face to face with a bombardier beetle.
There’s nothing like it in nature, and any sensible person knows that a tiny beetle less than an inch in size could never produce a controlled explosion by accident. It shouts an intelligent Creator.
All the parts had to be working from the start, not a product of step-by-step construction. Like a mousetrap won’t work unless all the pieces are working together, the same goes for this supreme example of irreducible complexity. In fact, the bombardier beetle’s blaster is so complex that scientists still don’t fully understand how all the parts work.

1: Chemical Production
Bombardier beetles are unique in their ability to superheat a liquid and expel it in an intense, pulsating jet. It starts with two chemicals, hydrogen peroxide and hydroquinone, produced in the secretory lobe and stored in the reservoir. Chemists still have not figured out how the insect produces the hydrogen peroxide, which is very unstable.
2: Movable Turret
The beetle can aim its turret in any direction, sending out repeated jets of steam through nozzles up to a stunning 65 feet (20 m) per second. Scientists still don’t know fully how the turret works.
3: Reaction Chamber
The two main chemicals do not react unless two other chemicals, known as catalysts, are present in the reaction chamber. How the beetle produces and stores these catalysts is still a mystery. The inner surface of the chamber is designed to withstand boiling temperatures produced by the reaction (221°F or 105°C).
The flow and direction of chemicals must be controlled by a valve system, in two stages. When the beetle is ready to fire, the inlet valve first opens, allowing the reactants to enter the chamber. Once the chamber is full and the chemicals react, the pressure pinches the inlet valve shut. At the same time, the growing heat and pressure forces the outlet valve open. After the ejection of each explosion of the hot pressurized fluid, the pressure drops and the valve closes.
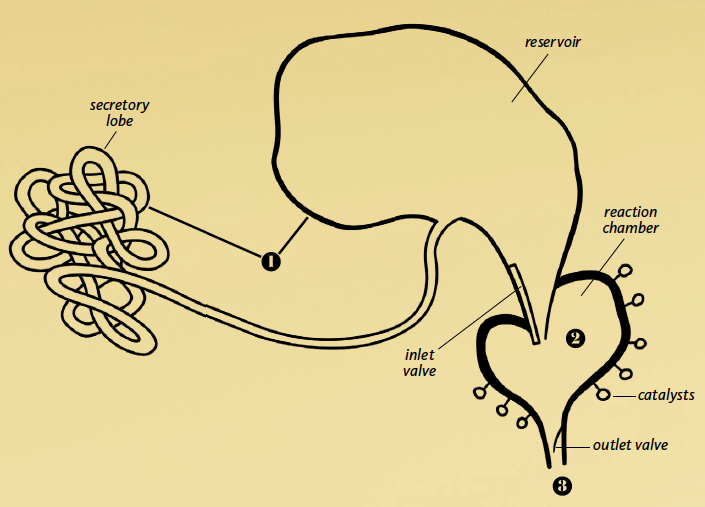

Actual Size
(under 1 inch)
368–735 Explosions per Second
The spray isn’t continuous. Instead, the beetle fires several bursts in rapid succession, which keeps the reaction chamber from overheating.
221°F
Average temperature of the chemicals when released. Because the chemicals are under pressure, the temperature is higher than the boiling point of water (212°F).
An Explosive Cocktail
Two common chemicals produce the beetle’s boiling spray. But they need an extra push to explode.
Hydrogen Peroxide
This chemical (H2O2) is commonly kept in medicine cabinets to clean wounds. In the beetle’s reaction chamber, a catalyst (called catalase) breaks the hydrogen peroxide down into water (H2O), free oxygen (O), and heat.
Hydroquinone
This bleaching agent (C6H6O2) is common in skin products to lighten skin. In the beetle’s reaction chamber, a catalyst (called peroxidases) releases hydrogen (H). The hydrogen then combines to initiate a runaway steam explosion with the free oxygen (above).
Answers Magazine
January–February 2020
Unlikely Weapon: The bombardier beetle provides explosive evidence of God’s intricate design.
Browse IssueRecommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis