
Diamonds—Evidence of Explosive Geological Processes
Evidence of Explosive Geological Processes
Originally published in Creation 16, no 1 (December 1993): 42-45.
Diamonds have been highly prized throughout history, being regarded as symbols of wealth and power.
A diamond was amongst the 12 gemstones on the high priest’s breastplate representing the 12 tribes of Israel before God (Exodus 28:18, 39:11). Diamonds were looked upon by God as things of beauty, purity and value. Indeed, in reference to the ‘anointed cherub’, Ezekiel describes his covering in Eden, ‘the garden of God’, and ‘upon the Holy mountain of God’, as ‘every precious stone’ including the diamond (Ezekiel 28:13–14).
The prized value has a lot to do with the appearance of cut gem-quality diamonds and their hardness. Even Jeremiah speaks of writing ‘with a pen of iron, and with the point of a diamond’ (Jeremiah 17:1). On the scale of hardness of minerals and natural materials diamonds are rated as the hardest—hence their industrial uses. Ironically however, diamonds are merely one physical form of the element carbon, another being graphite, which is of course quite soft, black and relatively unattractive. To many it is an enigma that an element like carbon could occur in such starkly contrasting physical forms, but such are the properties with which God has endowed this element.
An Aboriginal ‘myth’
Like many other native peoples around the world, the Australian Aborigines have stories about events in their history, including what had to be geological events. Their stories include one of a flood, the breakup of the continents, and a morning star.1 Of course, these traditions are usually dismissed as either simply religious beliefs, mythology, or the primitive explanation of local natural phenomena. Yet it is apparent that these stories, stripped of their grotesque elements and embellishments, are simply technically unsophisticated eyewitness accounts of real events. Thus it is significant that the Aborigines in the East Kimberleys of far northern Western Australia have a story associated with the Argyle diamond deposit which is in their tribal area. They say that the place where the diamond deposit is found is where the Barramundi (a large fish found in northern Australian estuaries) jumped out of the ground during the ‘Dreamtime’ (the time in which the earth received its present form, and cycles of life and nature were initiated).
This story of course has generally been accepted as fanciful native mythology, with no basis in fact, because the diamond deposit is supposed to be millions of years old. But could it be that these Aboriginal people actually saw this diamond deposit form in the recent past? Since diamond deposits appear to have formed by what can best be described as explosive volcanism, it would certainly seem that their description fits such an event.
Diamond deposits
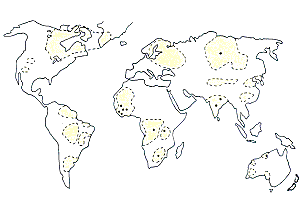
Diamond deposits are found in only a few isolated and restricted locations around the world where particular rock types occur. Figure 1 shows the location of the economic primary deposits, which are located in what are known geologically as ‘cratonic areas’, that appear to be the old foundational basement rocks on which the continents have been built.2 Historically, diamonds have been found and mined in southern Africa, from where sprang the DeBeers empire that grew to essentially control the world diamond market. However, today the largest deposits are found in Siberia, but the world’s largest diamond mine is at Argyle in northern Western Australia, the deposit whose formation the Aborigines appear to have witnessed. This mine currently produces 25 million carats of diamonds a year, about 30% of the world’s production.3
Diamond deposits are termed primary when found in the host rocks that have brought them from deep in the earth’s interior to the surface. The two host rock types in which significant quantities of diamond occur are called kimberlite (named after the best-known and earliest-mined diamond deposits at Kimberley in South Africa) and lamproite. Secondary diamond deposits are formed from these primary host rocks due to weathering and transportation by surface erosion processes. Because diamonds are so hard they survive weathering and erosion to end up in either gravels or alluvium, and even on beaches, as along the west coast of Namibia, southern Africa.
Host rock transport
Careful scientific investigations have revealed that diamonds have come from deep in the earth to the surface extremely rapidly. Identification of a rock as kimberlite or lamproite does not guarantee that it will contain economic quantities of diamonds. There are two reasons for this.4 It is now accepted by most geologists specializing in the study of diamond deposits that the diamonds themselves are crystals that are foreign to these rocks, that is, they were not part of the original rock material. Additionally, during the process of bringing the diamonds from inside the earth to the surface, in these rocks the diamonds may revert to graphite and/or be chemically dispersed and so be eliminated. Therefore, a given barren kimberlite or lamproite may never have contained diamonds, due to its failure to incorporate diamonds in it before its passage to the earth’s surface, or any diamonds originally present may have been completely destroyed during their passage upwards. Current research has thus shown that kimberlites and lamproites are merely vehicles which transport diamonds from deep inside the earth to the surface.
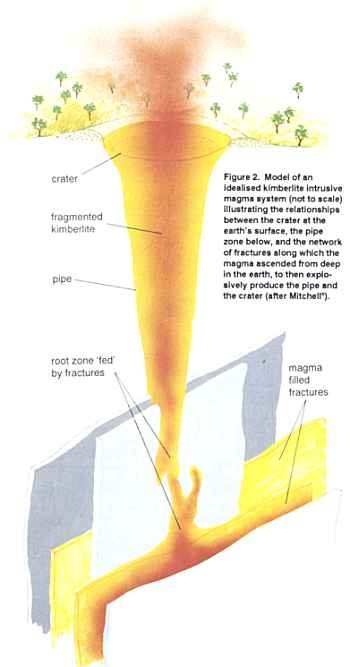
Kimberlites and lamproites could broadly be classified as volcanic rocks. They appear to have been produced from molten (and melted) rocks derived from the mantle below the earth’s outer crust.5 However, they have unusual compositions that set them apart from other volcanic rocks. The generalized three-dimensional shape of kimberlite and lamproite rock masses is depicted in Figure 2.6 This shape is usually called a pipe, but the uppermost part towards the land surface can be splayed out to make the shape more like a champagne glass. At depth the pipe narrows down and connects with a system of deep fractures. It is along these fractures that the molten rock forced its way upwards.
Diamond formation
Kimberlites and lamproites often contain fragments of other rocks types that have been ‘broken off and picked up’ during the passage of the molten rock from the earth’s interior to the surface. Amongst these foreign rock fragments are pieces of the probable source rocks that melted to form the molten masses of kimberlites and lamproites. Laboratory studies of the minerals in these rock fragments suggest that they formed at temperatures of 900–1400°C (approximately 1630–2500°F) and pressures of 50–80 kilobars (approximately 370–600 tons force per square inch). Such conditions evidently exist in the earth’s upper mantle at a depth of 150–250km (approximately 90–155 miles),7 and are also the conditions under which diamond is known to be stable. At shallower depths, where temperatures and pressures are lower, diamonds are not stable and any carbon present will occur as graphite. This implies that diamonds had to form at depths of more than 150km (about 90 miles) below the earth’s surface in the upper mantle.
Current ideas about diamond formation differ with respect to the postulated source of the carbon.8 Some researchers believe that diamonds form from carbon in methane or other hydrocarbon gases that ascend through the upper mantle from deeper inside the earth. Other scientists suggest that the carbon has come from the earth’s surface (and may even be ultimately of biological origin), presumably having been deeply buried and ‘pushed’ to these depths as a result of upheavals in the earth’s crust (explainable as due to the Flood).
In either case, it appears that the formation of a primary diamond deposit depends upon there having developed diamond–bearing horizons at depths greater than 150km in the ‘root zones’ of the continental foundation areas. Below in the upper mantle proper, localized melting of the rocks produced molten ‘blobs’ of kimberlite and lamproite compositions, which in their molten state were then ‘lighter’ (less dense and relatively buoyant) than the surrounding mantle rocks. As a consequence these molten ‘blobs’ (magmas) began to rise along fractures upwards towards the earth’s crust. If these molten ‘blobs’ passed through diamond–bearing horizons in the continental ‘root zones’, then diamond crystals may have become incorporated into the magmas, which then transported them into the crust and up to the earth’s surface.
A rapid ascent
However, once these ‘blobs’ of molten magma containing diamonds reached depths of less than 150km (about 90 miles) below the earth’s surface they were then in the zone where diamonds are no longer the stable form of carbon under the ambient temperature and pressure conditions. Consequently, if the magma ‘blobs’ moved too slowly up through the crust to the surface, and took too long to cool and harden there, then the contained diamonds would have been converted to graphite. These magmas would also have contained water and carbon dioxide gas, the water particularly enhancing the oxygen chemical reactivity of the ascending magma and potentially assisting the rapid oxidation and combustion of contained diamonds.
Laboratory experiments, coupled with other mineralogical and textural features in kimberlites and lamproites, indicate an ascent rate for these molten ‘blobs’ of diamond–bearing magmas of between 10 and 30km (6–19 miles) per hour.9 In other words, if these magma ‘blobs’ began their journey surfacewards at depths of 250km (about 155 miles), picking up diamonds on the way up to 150km (about 90 miles) depth, then they would have needed to reach the earth’s surface in only 8–25 hours!
Many people, of course, find it hard to conceive of a geological process like that occurring so rapidly, particularly as we have been constantly indoctrinated with the slow and gradual, uniformitarian philosophy. So how is it that these molten ‘blobs’ of magma made their way through solid rock from depths of around 250km (about 155 miles) or less to the earth’s surface in a day or less?
The magmas had within them components that produced a driving force for their rapid ascent. First there was the carbon dioxide gas content which would have built up an explosive gas drive.10 Then there was the water content, which at the magma’s temperatures was present as superheated steam, and would have been responsible for hydraulic fracturing and wedging.11 Both the carbon dioxide and water in the magmas were confined under pressure, much like soda in a corked bottle, ready to explosively exploit any weaknesses in the rocks above them.
It is hardly surprising then that most kimberlite and lamproite pipes occur at the intersections of major deep fracture systems. These fractures extend to within 2km (1.25 miles) of the surface, where they connect with the root zones of the pipes (see Figure 2 again). But why the change of mode at that depth? As the magmas rise along the fractures towards the earth’s surface the temperatures of the surrounding rocks and the confining pressures progressively fall. Because the magmas have travelled rapidly upwards they have had little time to cool.
At the shallow depth of 2km (1.25 miles) the confining pressures on the magmas were greatly diminished, which allowed the superheated steam in the magmas to in effect boil. Concurrently, the magmas came in contact with circulating groundwater, which had also been brought rapidly to boiling point by the heat of the rising magmas. The net result was explosive releases of energy, which are estimated as equivalent to two or three times the energy released per kilogram of magma in the Mount St Helens blast of May 18, 1980.12 These decompression reactions which suddenly produced rapid expansion of the confined gases and steam explosively, like a cork being popped out of a soda bottle, or like a bomb detonating, ‘punched’ holes all the way up to the surface. This is how the chimney-like pipes were formed.
The explosive eruptions were very localized and probably very short-lived, but resulted in ‘shattering’ of the then rapidly cooling magmas. Thus fragmented, they ended up filling the pipes as broken masses of coarse volcanic ash (or tuffs) whose grains and fragments were welded together to form hard masses (because of the heat still being released).13 The explosiveness and force involved is evident from the fragments of rocks, through which the pipes have been cut, that have also been included in the pipes. The diamond crystals themselves are thus carried rapidly from their place of formation deep in the earth into these pipes, where we find them today along with the shattered remains of the magmas that brought them up to the earth’s surface.
Conclusions
This evidence for the rapid formation of diamond deposits confirms that there are extremely rapid and catastrophic geological processes which evolutionary geologists have been forced to concede do occur. Furthermore, the eyewitness testimony from the Australian Aborigines, distorted by verbal transmission and the ‘mists of time’, undoubtedly points to their having seen the explosive eruption that produced the Argyle diamond deposit, which places its formation therefore in the very recent post-Flood period. To these undoubtedly awe-struck Aboriginal observers such an eruption could well, in their technically unsophisticated understanding, look like a giant fish jumping out of water.
Today’s diamond deposits at today’s land surface are in kimberlite and lamproite pipes that were intruded through strata most of which were undoubtedly deposited by the Flood. This means that the rapid ascent of molten kimberlites and lamproites and the explosive volcanism that resulted in the pipes must have occurred late in the Flood, soon after it, or sometime later (after Babel; in the case of the Argyle event seen by the Australian Aborigines. And of course the 8–25 hours it took the magmas to ascend and explosively form the pipes and craters is totally consistent with the biblical time framework in which the Flood occurred some 4500–5000 years ago.
References
- Hissink, L., 1993. Euhemerism and aboriginal myths. Letter to the Editor, The Australian Geologist, No. 86, pp. 6–7.
- Gurney, 1.1., 1989. Diamonds. In: Kimberlites and Related Rocks, J. Ross, A.L. Jaques, J. Ferguson, D.H. Green, S.Y. O’Reilly, R.V. Danchin and A.J.A. Janse (eds), Geological Society of Australia Inc., Special Publication No. 14 and Blackwell Scientific Publications (Australia), Melbourne, vol. 2, pp. 935–965.
- Fussell, A., 1988. The diamond club. Panorama, August, pp. 13–16.
- Mitchell, R.H., 1991. Kimberlites and lamproites: primary sources of diamond. Geoscience Canada, vol. 18(1), pp. 1–16.
- Eggler, D.H ., 1989. Kimberlites: how do they form? In. Kimberlites and Related Rocks, J. Ross, A.L. Jaques. J.Ferguson, D.H. Green, S.Y. O’Reilly, R.V. Danchin and A.J.A. Janse (eds), Geological Society of Australia Inc., Special Publication No. 14 and Blackwell Scientific Publications (Australia), Melbourne, vol. I. pp. 489-504.
- Mitchell, Ref. 4, p. 5.
- Mitchell, Ref. 4, p. 3.
- Mitchell, Ref. 4, p. 3.
- Eggler, Ref. 5, pp. 489, 497–499.
- Kennedy, G.C. and Nordlie, B.E., 1968. The genesis of diamond deposits. Economic Geology. vol. 63(5) pp. 495–503.
- Eggler, Ref. 5, pp. 499–500.
- Burnham, C.W., 1985. Energy release in subvolcanic environments: implications for breccia formation Economic Geology, vol. 80(6), pp. 1515–1522.
- Clement, C.R. and Reid, A.N., 1989. The origin of kimberlite pipes: an interpretation based on a synthesis of geological features displayed by southern Africa: occurrences. In: Kimberlites and Related Rocks, J Ross, A.L. Jaques, J. Ferguson, D.H. Green, S.Y O’Reilly, R.V. Danchin and A.J.A. Janse (eds) Geological Society of Australia Inc., Special Publication: No. 14 and Blackwell Scientific Publications (Australia) Melbourne, vol. 2, pp. 1007–1011.

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis
