The Cause of Anomalous Potassium-Argon “Ages” for Recent Andesite Flows at Mt. Ngauruhoe, New Zealand, and the Implications for Potassium-Argon “Dating”
Abstract
New Zealand’s newest and most active volcano, Mt. Ngauruhoe in the Taupo Volcanic Zone, produced andesite flows in 1949 and 1954 and avalanche deposits in 1975. Potassium-argon “dating” of five of these flows and deposits yielded K-Ar model “ages” from <0.27 Ma to 3.5±0.2 Ma. “Dates” could not be reproduced, even from splits of the same samples from the same flow, the explanation being variations in excess 40Ar* content. A survey of anomalous K-Ar “dates” indicates they are common, particularly in basalts, xenoliths, and xenocrysts such as diamonds that are regarded as coming from the upper mantle. In fact, it is now well established that there are large quantities of excess 40Ar* in the mantle, which in part represent primordial argon not produced by in situ radioactive decay of 40K and not yet outgassed. And there are mantle–crust domains between, and within which, argon circulates during global tectonic processes, magma genesis, and mixing of crustal materials. This has significant implications for the validity of K-Ar and 40Ar/39Ar “dating.”
Keywords: andesite, 1949–1975 flows, Mt. Ngauruhoe, New Zealand, potassium-argon dating, anomalous model “ages,” excess 40Ar*, excess 40Ar* in rocks and minerals, upper mantle, geochemical reservoirs, mantle-crust domains, crustal mixing, magma genesis
This paper was originally published in the Proceedings of the Fourth International Conference on Creationism, pp. 503–525 (1998), and is reproduced here with the permission of the Creation Science Fellowship of Pittsburgh (www.csfpittsburgh.org).
Introduction
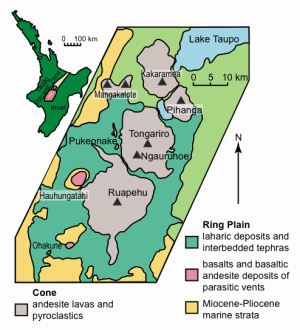
Mt. Ngauruhoe is an andesite stratovolcano of 2291 m elevation, rising above the Tongariro volcanic massif within the Tongariro Volcanic Center of the Taupo Volcanic Zone, North Island, New Zealand (fig. 1).1, 2 Though not as well publicized as its neighbor, Mt. Ruapehu (about 12 km to the south), Ngauruhoe is an imposing, almost perfect cone that rises more than 1000 m above the surrounding landscape. Eruptions from a central 400 m diameter crater have constructed the steep (33°) outer slopes of the cone.3, 4
Geologic Setting
The Taupo Volcanic Zone, a volcanic arc and marginal basin of the Taupo-Hikurangi arc-trench (subduction) system,5 is a southward extension on the Tonga-Kermadec arc into the continental crustal environment of New Zealand’s North Island. It has been interpreted as oblique subduction of the Pacific plate beneath the Australian plate. The zone extends approximately 300 km north-northeast across the North Island from Ohakune to White Island (fig. 1) and is up to 50 km wide in the central part, narrowing northward and southward. This volcanotectonic depression (Taupo-Rotorua depression6) comprises four rhyolitic centers (Rotorua, Okataina, Maroa, and Taupo), plus the calc-alkaline Tongariro Volcanic Center, part of a young (<0.25 Ma) andesite-dacite volcanic arc with no associated rhyolitic volcanism extending along the eastern side of the zone.7
The Tongariro Volcanic Center extends for 65 km south-southwest from Lake Taupo at the southern end of the Taupo Volcanic Zone (fig. 1) and consists of four large predominantly andesite volcanoes (Kakaramea, Pihanga, Tongariro, and Ruapehu; fig. 2); two smaller eroded centers at Maungakatote and Hauhungatahi; a satellite cone and associated flows at Pukeonake; and four craters at Ohakune (fig. 2).8, 9
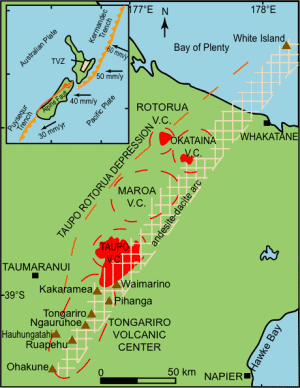
Fig. 1. The location of Mt. Ngauruhoe in the Taupo Volcanic Zone (TVZ), New Zealand, showing the main structural features. The shaded area is the andesite arc, and the inset shows the major components of the boundary between the Australian and Pacific Plates in the New Zealand region (arrows indicate relative motions). Solid triangles are basalt-andesite volcanoes.15
Most vents lie close to the axis of a large graben in which Quaternary volcanic rocks overlie a basement of Mesozoic greywacke and Tertiary sediments.10, 11 North-northeast-trending normal faults with throws up to 30 m cut the volcanoes within the graben. Nearly all vents active within the last 10 ka lie on a gentle arc which extends 25 km north-northeast from the Rangataua vent on the southern slopes of Ruapehu through Ruapehu summit and north flank vents, Tama Lakes, Ngauruhoe, Red Crater, Blue Lake, and Te Mari craters. None of the young vents lie on the mapped faults, which mostly downthrow toward the axis of the graben. The vent lineation lies above this axis, which is considered to mark a major basement fracture that allows the intrusion of andesite dikes.12, 13, 14
The Tongariro volcanics unconformably overlie late Miocene marine siltstones beneath Hauhungatahi, and a minimum age for the onset of volcanism is measured by the influx of andesite pebbles in early Pleistocene conglomerates of the Wanganui Basin to the south.16, 17 The oldest dated lavas from the Tongariro massif are hornblende andesites exposed at Tama Lakes between Ngauruhoe and Ruapehu, at 0.26±0.003 Ma; from Ruapehu, 0.23±0.006 Ma; and from Kakoramea, 0.22±0.001 Ma (potassium-argon dates).18
Tongariro itself is a large volcanic massif that consists of at least twelve composite cones, the youngest and most active of which is Ngauruhoe. A broad division has been made into older (>20 ka) and younger (<20 ka) lavas.19, 20 There is a north-northeast alignment of the younger vents of Tongariro, particularly evident between Te Mari and Ngauruhoe.
Ngauruhoe
Ngauruhoe is the newest cone of the Tongariro massif and has been active for at least 2.5 ka.21, 22, 23 It has been one of the most active volcanoes in New Zealand, with more than seventy eruptive episodes since 1839, when the first steam eruption was recorded by European settlers.24, 25, 26 Prior to European colonization the Maoris witnessed many eruptions from the mountain.27 The first lava eruption seen by European settlers occurred between April and August 1870, with two or three flows witnessed spilling down the northwestern flanks of the volcano on July 7.28, 29 Following that event there have been pyroclastic (ash) eruptions every few years,30 with major explosive activity in April–May 1948.
The next lava extrusion was in February 1949, beginning suddenly with ejection of incandescent blocks, and a series of hot block and ash flows down the northwestern slopes on February 9.31, 32 The southern sub-crater filled with lava, which by late on February 10 had flowed over the lowest part of the rim and down the northwest slopes of the cone. By February 12 the flow had ceased moving, subsequent mapping placing its volume at about 575,000 m3 (fig. 3).33, 34 Further explosive pyroclastic (ash) eruptions followed, reaching a maximum about February 19-21 . The eruptions ended on March 3.
The eruption from May 13, 1954, to March 10, 1955, began with explosive ejection of ash and blocks, although red-hot lava had been seen in the crater five months previous.35, 36 The eruption was remarkable for the estimated large volume of almost 8 million m3 of lava that then flowed from the crater from June through September 1954, and was claimed to be the largest flow of lava observed in New Zealand (that is, by the European settlers).37, 38 The lava was actually expelled from the crater in a series of seventeen distinct flows on the following dates:39, 40
- June 4, 30
- July 8, 9, 10, 11, 13, 14, 23, 28, 29, 30
- August 15 (?), 18
- September 16, 18, 26
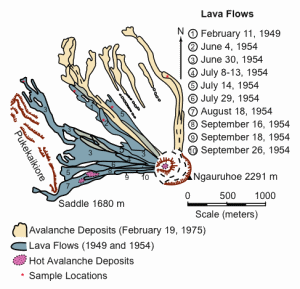
Fig. 3. Map of the northwestern slopes of Mt. Ngauruhoe showing the lava flows of 1949 and 1954 and the 1975 avalanche deposits.48, 49, 50, 51, 52 The location of samples collected for this study are marked.
Fig. 3 shows the distribution of those 1954 lava flows that are still able to be distinguished on the northwestern and western slopes of Ngauruhoe. All flows were of aa lava (as was the February 1949 flow), typified by rough, jagged, clinkery surfaces made up of blocks of congealed lava. The lava flows were relatively viscous, some being observed at close quarters slowly advancing at a rate of about 20 cm per minute.43, 44, 45 The August 18 flow was more than 18 m thick and still warm almost a year after being erupted. Intermittent explosive eruptions and spectacular lava fountaining during June and July 1954 built a spatter-and-cinder cone around the south sub-crater, modifying the western summit of the mountain. Activity decreased for two months after the last of the lava flows on September 26, but increased again during December 1954 and January 1955 with lava fountaining and many highly explosive pyroclastic (ash) eruptions. The last ash explosion was reported on March 10, 1955, but red-hot lava remained in the crater until June 1955.46, 47
After the 1954–1955 eruption, Ngauruhoe steamed semi-continuously, with numerous small eruptions of ash derived from comminuted vent debris. Incandescent ejecta were seen in January 1973, and ash erupted in December 1973 contained juvenile glassy andesite shards.53 Cannon-like, highly explosive eruptions in January and March 1974, the largest since 1954–1955, threw out large quantities of ash and incandescent blocks, one of which was reported as weighing 3000 tonnes and thrown 100 m.54, 55 Pyroclastic avalanches flowed from the base of large convecting eruption columns, down the west and north slopes of the cone, and the crater became considerably shallower.56, 57
A series of similar but more violent explosions occurred on February 19, 1975, accompanied by clearly visible atmospheric shock waves and condensation clouds.58, 59, 60, 61 Ash and blocks up to 30 m across were ejected and scattered within a radius of 3 km from the summit. The series of nine cannon-like, individual eruptions followed a 1.5 hour period of voluminous gas-streaming emission, which formed a convecting eruption plume between 11 km and 13 km high.62, 63, 64 The explosions took place at 20–60 minute intervals for more than five hours. Numerous pyroclastic avalanches were also generated by fallback from the continuous eruption column, the avalanches consisting of a turbulent mixture of ash, bombs, and larger blocks which rolled swiftly down Ngauruhoe’s sides at about 60 km per hour.65, 66 The deposits from these avalanches and the later explosions accumulated as sheets of debris in the valley at the base of the cone, but did not extend beyond 2 km from the summit. It is estimated that a minimum bulk volume of 3.4 million m3 of pyroclastic material was erupted in the seven-hour eruption sequence on that day.51 Fig. 3 shows the location of these avalanche deposits.
There have been no eruptions since February 1975. A plume of steam or gas is still often seen above the summit of the volcano, as powerful fumaroles in the bottom of the crater discharge hot gases. However, the temperature of these fumaroles in the crater floor has steadily cooled significantly since 1979, suggesting that the main vent is becoming blocked.
Sample Collection
Field work and collection of samples was undertaken in January 1996. The Ngauruhoe area was accessed from State Highway 47 via Mangateopopo Road. From the parking area at the end of the road, the Mangateopopo Valley walking trail was followed to the base of the Ngauruhoe cone, from where the darker-colored recent lava flows were clearly visible and each one easily identified on the northwestern slopes against the lighter-colored older portions of the cone (fig. 3).
Eleven 2–3 kg samples were collected: two each from the February 11, 1949, June 4, 1954, and July 14, 1954, lava flows and from the February 19, 1975, avalanche deposits; and three from the June 30, 1954 lava flows. The sample locations are marked on Fig. 3. Care was taken to ensure correct identification of each lava flow and that the samples collected were representative of each flow and any variations in textures and phenocrysts in the lavas.
Laboratory Work
All samples were sent first for sectioning one thin section from each sample for petrographic analysis. A set of representative pieces from each sample (approximately 100 g) was then dispatched to the AMDEL Laboratory in Adelaide, South Australia, for whole-rock major, minor, and trace element analyses. A second representative set (50–100 g from each sample) was sent progressively to Geochron Laboratories in Cambridge (Boston), Massachusetts, for whole-rock potassium-argon (K-Ar) dating—first a split from one sample from each flow, then a split from the second sample from each flow after the first set of results was received, and finally, the split from the third sample from the June 30, 1954, flow.
At the AMDEL Laboratory each sample was crushed and pulverized. Whole-rock analyses were undertaken by total fusion of each powdered sample and then digesting them before ICP-OES for major and minor elements, and ICP-MS for trace and rare earth elements. Fe was analyzed for amongst the major elements by ICP-OES as Fe2O3 and reported accordingly, but separate analyses for Fe as FeO were also undertaken via wet chemistry methods. The detection limit for all major element oxides was 0.01%. For minor and trace elements the detection limits varied between 0.5 and 20 ppm, and for rare earth elements between 0.5 and 1 ppm.
The potassium and argon analyses were undertaken at Geochron Laboratories under the direction of Richard Reesman, the K-Ar laboratory manager. No specific location or expected age information was supplied to the laboratory. However, the samples were described as andesites that probably contained “low argon” and therefore could be young, so as to ensure the laboratory took extra care with the analytical work.
Because the sample pieces were submitted as whole rocks, the K-Ar laboratory undertook the crushing and pulverizing preparatory work. The concentrations of K2O (weight %) were then measured by the flame photometry method, the reported values being the averages of two readings for each sample.67, 68 The 40K concentrations (ppm) were calculated from the terrestrial isotopic abundance using the measured concentrations of K2O. The concentrations in ppm of 40Ar*, the supposed “radiogenic” 40Ar, were derived using the conventional formula from isotope dilution measurements on a mass spectrometer by correcting for the presence of atmospheric argon whose isotopic composition is known.69 The reported concentrations of 40Ar* are the averages of two values for each sample. The ratios 40Ar*/Total Ar and 40Ar/36Ar are also derived from measurements on the mass spectrometer and are also the averages of two values for each sample.
Table 1. Whole-rock, major-element oxide analyses of recent lava flows at Mt. Ngauruhoe, New Zealand, as reported in the literature.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
|---|---|---|---|---|---|---|---|---|
| SiO2 | 56.63 | 57.24 | 55.90 | 56.22 | 56.2 | 55.83 | 55.73 | 56.05 |
| TiO2 | 0.81 | 0.81 | 0.76 | 0.76 | 0.8 | 0.76 | 0.77 | 0.84 |
| Al2O3 | 16.71 | 16.75 | 16.90 | 16.63 | 16.6 | 17.03 | 17.23 | 16.08 |
| Fe2O3 | 1.16 | 1.54 | 2.10 | 2.37 | 1.4 | 2.14 | 2.13 | 3.24 |
| FeO | 7.00 | 6.44 | 6.30 | 6.14 | 7.0 | 6.36 | 6.47 | 5.57 |
| MnO | 0.16 | 0.12 | 0.15 | 0.15 | 0.1 | 0.16 | 0.16 | 0.15 |
| MgO | 4.85 | 4.58 | 5.20 | 5.24 | 5.2 | 4.79 | 4.89 | 5.03 |
| CaO | 8.16 | 7.95 | 8.40 | 8.31 | 8.3 | 8.37 | 8.52 | 7.93 |
| Na2O | 2.85 | 2.74 | 2.60 | 3.14 | 3.1 | 2.93 | 2.86 | 2.82 |
| K2O | 1.16 | 1.49 | 1.00 | 1.14 | 1.2 | 1.20 | 1.08 | 1.23 |
| P2O5 | 0.16 | 0.17 | 0.10 | 0.17 | 0.2 | 0.15 | 0.15 | 0.15 |
| H2O | 0.31 | 0.24 | 0.06 | 0.19 | N/A | 0.14 | 0.10 | 0.38 |
| TOTAL | 99.95 | 100.07 | 99.47 | 100.46 | 100.1 | 99.86 | 100.09 | 99.47 |
- Ejecta from March 1928 eruption73, 74
- Lava from February 1949 eruption75, 76, 77
- Lava from June 30, 1954, flow78, 79, 80
- 1954 lava (VU 29250)81
- 1954 lava (VU 29250)82, 83
- Average of four lava flows from 1954 eruptions84, 85
- Average of five blocks and bombs from January and March 1974 eruptions86, 87
- Lapilli from February 19, 1975, eruption88
Petrography and Chemistry
Clark reported that most of the flows from Ngauruhoe are labradorite-pyroxene andesite with phenocrysts of plagioclase (labradorite), hypersthene, and rare augite in a hyalopilitic (needle-like microlites set in a glassy mesostasis) groundmass containing abundant magnetite.70 However, all lava, lapilli, and incandescent blocks that have been analyzed from eruptions this century also contain olivine; chemically they may be classed as low-silica (or basaltic) andesites (using the classification scheme of Gill).71 The published analyses in Table 1 show only trivial changes in composition between 1928 and 1975. In fact, the 1954 and 1974 andesites are so similar that Nairn, et al., suggested that a solid plug of 1954 andesite was heated to incandescence and partially remobilized on top of a rising magma column in 1974.72 This plug was disrupted and blown from the vent as ejecta ranging in texture from solid blocks, through expanded scoria to spatter bombs.
Table 2 lists the whole-rock major element analyses of the eleven samples collected in this study. Comparison of the data for each flow with the corresponding data in Table 1 indicates that in their bulk chemistries all the samples analyzed (and thus all the flows) are virtually identical to one another, the trivial differences being attributable to the statistics of analytical errors, sampling, and natural variations. Thus it is not unreasonable to conclude that these basaltic andesites are cogenetic, coming from the same magma and magma chamber, even as they have been observed to flow from the same volcano. Nevertheless, Nairn, et al., suggested that even though the 1949 and 1954 lavas were both olivine-bearing andesite, the chemical analyses (table 1) showed the 1954 lava to be slightly more basic than the 1949 lava, with slightly higher MgO, CaO, and total iron oxides, but lower SiO2 and alkalis.89 However, these trends are not duplicated with any statistical significance by the analytical results of this study (table 2). At least, they found that their analyses of the 1974 lava blocks and bombs were identical within the limits of error with the 1954 lava (table 1), which was also substantiated in this study with respect to the 1975 avalanche material and the 1954 lava (table 2).
Table 2. Whole-rock, major-element oxide analyses of five recent lava flows at Mt. Ngauruhoe, New Zealand (Analyst: AMDEL, Adelaide; April 1996.)
| 1A | 1B | 2A | 2B | 3A | 3B | 3C | 4A | 4B | 5A | 5B | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 56.7 | 56.2 | 55.3 | 55.8 | 56.3 | 55.9 | 55.6 | 56.1 | 55.6 | 56.0 | 55.4 |
| TiO2 | 0.79 | 0.85 | 0.74 | 0.77 | 0.76 | 0.75 | 0.74 | 0.75 | 0.84 | 0.79 | 0.78 |
| Al2O3 | 17.2 | 17.3 | 16.5 | 17.3 | 17.0 | 16.9 | 16.7 | 16.9 | 17.5 | 17.0 | 16.5 |
| Fe2O3* | 9.10 | 9.63 | 9.26 | 9.23 | 9.11 | 9.17 | 9.59 | 9.29 | 9.61 | 9.25 | 9.43 |
| MnO | 0.15 | 0.16 | 0.15 | 0.15 | 0.15 | 0.15 | 0.16 | 0.15 | 0.16 | 0.15 | 0.16 |
| MgO | 4.28 | 3.84 | 5.21 | 4.71 | 4.75 | 5.00 | 5.09 | 4.71 | 3.84 | 4.31 | 5.27 |
| CaO | 7.61 | 7.93 | 8.22 | 8.29 | 7.95 | 8.16 | 8.17 | 8.00 | 8.17 | 7.83 | 8.56 |
| Na2O | 3.08 | 3.19 | 2.91 | 3.03 | 3.06 | 2.98 | 2.95 | 3.02 | 3.11 | 3.08 | 2.86 |
| K2O | 1.15 | 1.01 | 1.05 | 1.00 | 1.10 | 1.08 | 1.06 | 1.12 | 1.04 | 1.10 | 1.09 |
| P2O5 | 0.13 | 0.13 | 0.12 | 0.12 | 0.13 | 0.13 | 0.13 | 0.13 | 0.13 | 0.14 | 0.14 |
| L0I | 0.42 | 0.48 | 0.51 | 0.37 | 0.38 | 0.50 | 0.53 | 0.62 | 0.42 | 0.70 | 0.41 |
| TOTAL | 100.51 | 100.72 | 99.97 | 100.77 | 100.69 | 100.72 | 100.72 | 100.79 | 100.42 | 100.35 | 100.60M |
*Total Fe as Fe2O3
- February 11, 1949, flow, samples A and B
- June 4, 1954, flow, samples A and B
- June 30, 1954, flow, samples A, B, and C
- July 14, 1954, flow, samples A and B
- February 19, 1975, flow, samples A and B
Clark and Cole recognized five lava types in the Tongariro Volcanic Center based on the modal proportions of the phenocryst minerals.90, 91 Graham modified this scheme to six types based on a combination of mineralogy and chemistry, but given their uniform bulk chemistry and petrology, these Ngauruhoe lava flows group together as plagioclase-pyroxene andesite within Graham’s “Type 1.”92 Cole, et al., have described Type 1 lavas as volumetrically dominant within the Tongariro Volcanic Center and as exhibiting coherent chemical trends with increasing silica content.93 They are relatively Fe-rich and follow a typical calc-alkaline trend on the AFM diagram.
Adapting the terminology of Gill, the Ngauruhoe lavas are described as basic andesites (53–58 wt% SiO2).94, 95 Their designation as plagioclase-pyroxene andesites is based on the predominant phenocrysts present, with plagioclase greater than or equal to pyroxene. Two modal analyses are listed in Table 3 which very closely resemble the samples collected for this study.
All samples of the five lava flows examined in this study exhibited a porphyritic texture, with phenocrysts (up to 3 mm across) consistently amounting to 35–40% by volume. The phenocryst assemblage is dominated (2:1) by plagioclase, but orthopyroxene and augite (clinopyroxene) are always major components, while olivine and magnetite are only present in trace amounts. This POAM phenocryst assemblage is a typical anhydrous mineralogy.98 The groundmass consists of microlites of plagioclase, orthopyroxene, and clinopyroxene, and is crowded with minute granules of magnetite and/or Fe-Ti oxides. Small amounts (9–10%) of brown transparent (acid-residuum) glass are also present, and the overall texture is generally pilotaxitic.
Table 3. Modal analyses of two recent lava flows at Mt. Ngauruhoe, New Zealand, as reported in the literature.
| Component | 1 | 2 |
|---|---|---|
| Plagioclase | 22.6 | 21.6 |
| Augite | 2.6 | 2.6 |
| Orthopyroxene | 6.0 | 5.8 |
| Olivine | 0.2 | 0.2 |
| Iron Oxide | — | g* |
| Xenoliths | 2.6 | 4.5 |
| Groundmass | 66.0 | 65.3 |
| TOTAL | 100.0 | 100.0 |
g* in groundmass
Steiner stressed that xenoliths are a common constituent of the 1954 Ngauruhoe lava, but also noted that Battey reported the 1949 Ngauruhoe lava was rich in xenoliths. All samples in this study contained xenoliths, including those from the 1975 avalanche material.99, 100 However, many of these aggregates are more accurately described as glomerocrysts and mafic (gabbro, websterite) nodules.101 They are 3–5 mm across, generally have hypidiomorphic-granular textures, and consist of plagioclase, orthopyroxene, and clinopyroxene in varying proportions, and very occasionally olivine. The true xenoliths are often rounded and invariably consist of fine quartzose material. Steiner also described much larger xenoliths of quartzo-feldspathic composition and relic gneissic structure.
The plagioclase phenocrysts have been reported as ranging in composition from An89 to An40 (andesine to bytownite), but in Ngauruhoe lavas are usually labradorite (An68-55). They are subhedral and commonly exhibit complex oscillatory zoning with an overall trend from calcic cores to sodic rims.102, 103, 104 Thin outer rims are usually compositionally similar to groundmass microlites. Twinning and hourglass structures are common.
Orthopyroxene predominates (>2:1) over clinopyroxene. Subhedral-euhedral orthopyroxene is typically pleochroic and sometimes zoned. Compositions range from Ca4 Mg74 Fe22 to Ca3 Mg47 Fe50, but representative bulk and partial analyses of Ngauruhoe orthopyroxenes indicate a hypersthene composition predominates, which is confirmed by optical determinations.105, 106, 107, 108, 109, 110, 111 Euhdral-subhedral clinopyroxene is typically twinned and zoned, but compositions show a restricted range of Ca43 Mg47 Fe10 to about Ca35 Mg40 Fe25, all of which is augite.112, 113
The olivine present is strongly magnesian, analyses indicating some compositional zoning from Fo88 to Fo78. The magnetite present in the groundmass is titanomagnetite, judging from the amount of TiO2 present in whole-rock analyses (tables 1 and 2), but some ilmenite is likely to occur sporadically in association with it.114, 115
K-Ar Results
All analytical results received from Geochron Laboratories are listed in table 4, grouped in chronological order according to the historic date of each flow. The 40Ar* quantity refers to the amount of radiogenic 40Ar measured in each sample. All other quantities are self-explanatory, some of them being calculated from the analytical results supplied by the laboratory.
The “age” of each sample is calculated from the analytical results using the general model-age equation:116, 117
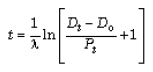 (1)
(1)
where:
| t | = the “age” |
| ? | = the decay constant of the parent isotope |
| Dt | = the number of daughter atoms in the rock presently |
| Do | = the number of daughter atoms initially in the rock |
| Pt | = the number of parent atoms presently in the rock |
To date a rock, Dt and Pt are measured, and equation (1) can then be used if an assumption about the original quantity of daughter atoms (Do) is made. Applied specifically to K-Ar dating, equation (1) thus becomes:
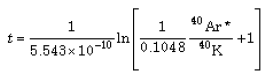 (2)
(2)
where:
| t | = the “age” in Ma (millions of years) |
| 5.543 × 10-10 | = the current estimate for the decay constant of 40K |
| 0.1048 | = the estimated fraction of 40K decays producing 40Ar |
| 40Ar*/40K | = the calculated mole ratio of radiogenic 40Ar to 40K in the sample |
It should be noted that to make equation (2) equivalent to equation (1), 40Ar* is assumed to be equal to (Dt - Do), which thus means the 40Ar* measurement has included within it an assumption concerning the initial quantity of 40Ar in the rock, namely, no radiogenic argon is supposed to have existed when the rock formed (that is, Do = 0). Thus equation (2) yields a “model age” assuming zero radiogenic argon in the rock when it formed.
The model ages listed in Table 4 range from <0.27 Ma to 3.5±0.2 Ma. However, it should be noted that the samples, one from each flow, that yielded model ages of <0.27 Ma and <0.29 Ma (that is, below the detection limits of the equipment for 40Ar*) were all processed at the K-Ar laboratory in the same batch, suggesting the possibility of a systematic problem with the analytical procedure and equipment (in particular, the gas extraction “line”). When this question was raised with the laboratory manager, Richard Reesman, he kindly rechecked his equipment and then re-ran several of the samples, producing similar results and thus ruling out a systematic laboratory “error.”
However, an independent blind check was then made, by submitting to the K-Ar laboratory duplicate splits from two samples already analyzed, to establish if results really were reproducible. The samples chosen were the A and B samples of the June 30, 1954, flow, because their first splits had produced the lowest and highest model ages, <0.27 Ma and 3.5±0.2 Ma, respectively. The results of these additional analyses are shown in Table 4 as A#2 and B#2, and yielded model ages of 1.3±0.3 Ma and 0.8±0.2 Ma, respectively. Clearly, reproducibility was not obtained, but this is not surprising given the analytical uncertainties at such low to negligible levels of 40Ar*, which are at the detection limits of the laboratory’s equipment (R. Reesman, pers. commun., February 19, 1997, and January 26, 1998).
Discussion
In spite of the wide variations in model “ages” obtained between and within these recent lava flows, and of the difficulties obtaining analytical reproducibility, it is apparent that the cause of the anomalous K-Ar model “ages” is excess argon in the lavas, that is, non-zero concentrations of radiogenic argon (40Ar*). This of course is contrary to the assumption of zero radiogenic argon in equation (2) for calculating the model “ages.” When analyzed the oldest of the lavas was less than 50 years old, so there has been insufficient time since cooling for measurable quantities of 40Ar* to have accumulated within the lavas due to the slow radioactive decay of 40K. Thus the measurable 40Ar* can’t be from in situ radioactive decay since cooling, and therefore must have been present in the molten lavas when extruded from Mt. Ngauruhoe.
No Radiogenic Argon Assumption Violated by Many Anomalous “Ages”
The assumption of no radiogenic argon (40Ar*) when the rocks formed is usually stated as self-evident. For example, Geyh and Schleicher state:
What is special about the K-Ar method is that the daughter nuclide is a noble gas, which is not normally incorporated into minerals and is not bound in the mineral in which it is found.118 (p. 56)
Similarly, Dalrymple and Lanphere state:
a silicate melt will not usually retain the 40Ar that is produced, and thus the potassium-argon clock is not “set” until the mineral solidifies and cools sufficiently to allow the 40Ar to accumulate in the mineral lattice.119 (p. 46)
Dalrymple has recently put the argument more strongly:
The K-Ar method is the only decay scheme that can be used with little or no concern for the initial presence of the daughter isotope. This is because 40Ar is an inert gas that does not combine chemically with any other element and so escapes easily from rocks when they are heated. Thus, while a rock is molten the 40Ar formed by decay of 40K escapes from the liquid.120 (p. 91)
Table 4. K-Ar analytical results and model ages for five recent lava flows at Mt. Ngauruhoe, New Zealand (Analyst: Geochron Laboratories, Boston; July 1996, December 1996, September 1997, and January 1998.) Constants used: 40K/K = 1.193 × 10-4g/g; Fraction of 40K decays to 40Ar* = 0.1048; Decay constant of 40K = 5.543 × 10-10yr-1; Atmospheric 40Ar/36Ar = 295.5
| Flow Date | Sample | K2O (wt%) | 40K (ppm) | 40Ar* (ppm)× 10-4 | 40Ar* (%) | 40Ar* Total 40Ar | Total 40Ar (ppm) | 40Ar 36Ar | 36Ar (ppm) × 10-5 | 40Ar* 40K | Model Age (Ma) | Uncertainty (Ma) (1s) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| February 11, 1949 | A B | 1.066 1.057 | 1.272 1.261 | <0.2 0.75 | — 3.75 | — 0.0375 | — 0.0020 | 296 310 | — 0.645 | <0.000016 0.000059 | <0.27 1.0 | — 0.2 |
| June 4, 1954 | A B | 1.034 1.087 | 1.234 1.297 | <0.2 1.10 | — 3.00 | — 0.030 | — 0.00367 | 288 308 | — 1.192 | <0.000016 0.000085 | <0.27 1.5 | — 0.1 |
| June 30, 1954 | A#1 A#2 | 1.074 0.977 | 1.281 1.166 | <0.2 0.87 | — 2.75 | — 0.0275 | — 0.00316 | 285 306 | — 1.033 | <0.000016 0.000075 | <0.27 1.3 | — 0.3 |
| B#1 B#2 | 0.944 0.995 | 1.126 1.186 | 2.28 0.56 | 4.05 1.70 | 0.405 0.017 | 0.00691 0.00329 | 311 304 | 2.222 1.082 | 0.000202 0.000047 | 3.5 0.8 | 0.2 0.2 |
|
| C | 1.097 | 1.308 | 0.93 | 3.75 | 0.0375 | 0.00248 | 311 | 0.798 | 0.000071 | 1.2 | 0.2 |
|
| July 14, 1954 | A B | 1.033 1.004 | 1.232 1.198 | 0.69 <0.2 | 3.40 — | 0.034 — | 0.00203 — | 301 283 | 0.675 — | 0.000056 <0.000017 | 1.0 <0.29 | 0.2 — |
| February 19, 1975 | A B | 1.128 1.048 | 1.346 1.250 | 0.79 <0.2 | 2.55 — | 0.0255 — | 0.00310 — | 307 291 | 1.010 — | 0.000059 <0.000016 | 1.0 <0.27 | 0.2 — |
However, these dogmatic statements by Dalrymple are inconsistent with even his own work on historic lava flows, some of which he found had non-zero concentrations of 40Ar* in violation of this key assumption of the K-Ar dating method.121 He does go on to admit, “Some cases of initial 40Ar remaining in rocks have been documented but they are uncommon,” but then refers to his study of 26 historic, subaerial lava flows.122, 123 Five (almost 20%) of those flows contained “excess argon,” but Dalrymple still then says “that ‘excess’ argon is rare in these rocks!” The flows and their “ages” were:124
| Hualalai basalt, Hawaii (AD 1800–1801) | 1.6±0.16 Ma |
| 1.41±0.08 Ma | |
| Mt. Etna basalt, Sicily (122 BC) | 0.25±0.08 Ma |
| Mt. Etna basalt, Sicily (AD 1792) | 0.35±0.14 Ma |
| Mt. Lassen plagioclase, California (AD 1915) | 0.11±0.03 Ma |
| Sunset Crater basalt, Arizona (AD 1064–1065) | 0.27±0.09 Ma |
| 0.25±0.15 Ma |
Far from being rare, there are numerous examples reported in the literature of excess 40Ar* in recent or young volcanic rocks producing excessively old whole-rock K-Ar “ages”:
| Akka Water Fall flow, Hawaii (Pleistocene) | 32.3±7.2 Ma125 |
| Kilauea Iki basalt, Hawaii (AD 1959) | 8.5±6.8 Ma126 |
| Mt. Stromboli, Italy, volcanic bomb (September 23, 1963) | 2.4±2 Ma127 |
| Mt. Etna basalt, Sicily (May 1964) | 0.7±0.01 Ma128 |
| Medicine Lake Highlands obsidian, Glass Mountains, California (<500 years old) | 12.6±4.5 Ma129 |
| Hualalai basalt, Hawaii (AD 1800–1801) | 22.8±16.5 Ma130 |
| Rangitoto basalt, Auckland, New Zealand (<800 years old) | 0.15±0.47 Ma131 |
| Alkali basalt plug, Benue, Nigeria (<30 Ma) | 95 Ma132 |
| Olivine basalt, Nathan Hills, Victoria Land, Antarctica (<0.3 Ma) | 18.0±0.7 Ma133 |
| Anorthoclase in volcanic bomb, Mt. Erebus, Antarctica (1984) | 0.64±0.03 Ma134 |
| Kilauea basalt, Hawaii (<200 years old) | 21±8 Ma135 |
| Kilauea basalt, Hawaii (<1000 years old) | 42.9±4.2 Ma136 |
| 30.3±3.3 Ma137 | |
| East Pacific Rise basalt (<1 Ma) | 690±7 Ma138 |
| Seamount basalt, near East Pacific Rise (<2.5 Ma) | 580±10 Ma139 |
| 700±150 Ma140 | |
| East Pacific Rise basalt (<0.6 Ma) | 24.2±1.0 Ma141 |
Other studies have also reported measurements of excess 40Ar* in lavas. Fisher investigated submarine basalt from a Pacific seamount and found “the largest amounts of excess 4He and 40Ar ever recorded” (at that time).142 McDougall not only found “extraneous radiogenic argon present in three of the groups of basalt flows” on the young volcanic island of Réunion in the Indian Ocean, but “extraneous argon” was also “detected in alkali feldspar and amphibole in hyperbyssal drusy syenites that are exposed in the eroded core of Piton des Neiges volcano.”143 Significant quantities of excess 40Ar* have also been recorded in submarine basalts, basaltic glasses and olivine phenocrysts from the currently active Hawaiian volcanoes, Loihi Seamount and Kilauea, as well as on the flanks of Mauna Loa and Hualalai volcanoes, also part of the main island of Hawaii, and in samples from the Mid-Atlantic Ridge, East Pacific Rise, Red Sea, Galapagos Islands, McDonald Seamount and Manus Basin.144, 145, 146, 147 Patterson, Honda, and McDougall claimed that some of the initial Loihi analytical results were due to atmospheric contamination of the magma either during intrusion or eruption, but subsequent work has confirmed that the excess 40Ar* is not from atmospheric contamination at all.148, 149, 150
Excess 40Ar* Occluded in Minerals
Austin has investigated the 1986 dacite lava flow from the post–October 26, 1980, lava dome within the Mount St. Helens crater and has established that the 10-year-old dacite yields a whole-rock K-Ar model “age” of 0.35±0.05 Ma due to excess 40Ar* in the rock.151 He then produced concentrates of the constituent minerals, which yielded anomalous K-Ar model “ages” of 0.34±0.06 Ma (plagioclase), 0.9±0.2 Ma (hornblende), 1.7±0.3 Ma (pyroxene), and 2.8±0.6 Ma (pyroxene ultra-concentrate). While these mineral concentrates were not ultra-pure, given the fine-grained glass in the groundmass and some Fe-Ti oxides, it is nonetheless evident that the excess 40Ar* responsible for the anomalous K-Ar “ages” is retained within the different constituent minerals in different amounts. Furthermore, the whole-rock “age” is very similar to the “age” of the plagioclase concentrate because plagioclase is the dominant constituent of the dacite.
That the excess 40Ar* can be occluded in the minerals within lava flows, rather than between the mineral grains, has been established by others also. Laughlin, et al., found that the olivine, pyroxene, and plagioclase in Quaternary basalts of the Zuni-Bandera volcanic field of New Mexico contained very significant quantities of excess 40Ar*, as did the olivine and clinopyroxene phenocrysts in Quaternary flows from New Zealand volcanoes.152, 153 Similarly, Poths, Healey, and Laughlin separated olivine and clinopyroxene phenocrysts from young basalts from New Mexico and Nevada and then measured “ubiquitous excess argon” in them.154 Damon, Laughlin, and Precious have reported several instances of phenocrysts with K/Ar “ages” 17 million years greater than that of the whole rocks, and one K/Ar “date” on olivine phenocrysts of greater than 110 Ma in a recent (<13,000 year old) basalt.155 Damon, et al., thus suggested that large phenocrysts in volcanic rocks contain the excess 40Ar* because their size prevents them from completely degassing before the flows cool, but Dalrymple concluded that there does not appear to be any correlation of excess 40Ar* with large phenocrysts or with any other petrological or petrographic parameter.156
Most investigators have come to the obvious conclusion that the excess 40Ar* had to have been present in the molten lavas when extruded, which then did not completely degas as they cooled, the excess 40Ar* becoming “trapped” in the constituent minerals, and in some instances, the rock fabrics themselves. Laboratory experiments have tested the solubility of argon in synthetic basalt melts and their constituent minerals near 1300°C at one atmosphere pressure in a gas stream containing argon.157, 158 When quenched, synthetic olivine in the resultant material was found to contain 0.34 ppm 40Ar*. Broadhurst, et al., commented that “The solubility of Ar in the minerals is surprisingly high,” and concluded that the argon is held primarily in lattice vacancy defects within the minerals.159
In a different experiment, Karpinskaya, Ostrovskiy, and Shanin heated muscovite to 740–860°C under high argon pressures (2800–5000 atmospheres) for periods of 3 to 10.5 hours.160 The muscovite absorbed significant quantities of argon, producing K/Ar “ages” of up to 5 billion years, and the absorbed argon appeared like ordinary radiogenic argon (40Ar*). Karpinskaya subsequently synthesized muscovite from a colloidal gel under similar argon pressures and temperatures, the resultant muscovite retaining up to 0.5 wt% argon at 640°C and a vapor pressure of 4000 atmospheres.161 This is approximately 2,500 times as much argon as is found in natural muscovite. These experiments show that under certain conditions argon can be incorporated into minerals and rocks that are supposed to exclude argon when they crystallize.
Applications to the Mt. Ngauruhoe Andesite Flows
Therefore, the analytical results from the very recent (1949–1975) andesite flows at Mt. Ngauruhoe, New Zealand, that yield anomalous K-Ar model “ages” because of excess 40Ar* are neither unique nor an artifact of poor analytical equipment or technique. This realization that the presence of the excess 40Ar* in these rocks is both real and measurable, and has not been derived from radioactive decay of 40K in situ, leads to the obvious questions as to whether there is any pattern in the occurrences of excess 40Ar*, and from whence came this excess 40Ar*?
It is clear that the excess 40Ar* was in the lavas when they flowed from the Mt. Ngauruhoe volcano and were trapped in the andesite as it cooled. That there were gases in the lavas is readily evident from the copious “frozen” bubble holes now in the rock, implying that much of the gas content escaped as the lavas flowed and cooled. When choosing samples, care was taken to select pieces from each flow that were different from one another (for example, copious “frozen” gas bubble holes compared with virtually no such holes). It is hardly surprising, therefore, that the 40Ar* measurements on four of the five flows were consistent with such differences the samples from each flow which had very few or virtually no “frozen” gas bubble holes yielded excess 40Ar* and thus anomalous K-Ar model “ages,” whereas the other samples from each of these flows that contained copious “frozen” gas bubble holes failed to yield detectable 40Ar* (<0.27 Ma and <0.29 Ma in Table 4).
The exception was the June 30, 1954, flow—not only was this expected relationship between excess 40Ar* and lack of “frozen” gas bubble holes not duplicated, but analyses on duplicate splits off the same samples yielded widely divergent results (<0.27 Ma versus 1.3±0.3 Ma and 3.5±0.2 Ma versus 0.8±0.2 Ma; see Table 4). Thus the presence (or absence) of excess 40Ar* must also depend on which portion of a rock sample is being analyzed, which in turn implies dependence on the mineral constituents present, including the glass in the groundmass. As already noted, Austin found widely different amounts of excess 40Ar* in the mineral separates concentrated from Mount St. Helens 1986 dacite, while numerous other studies have located excess 40Ar* in phenocrysts.162, 163, 164, 165, 166
Cooling Rates, Pressures, and Potassium Alteration
Another factor is the rate of cooling of lavas. Dalrymple and Moore found that the 1 cm thick glassy rim of a pillow in a Kilauea submarine basalt had greater than forty times more excess 40Ar* than the basalt interior just 10 cm below.167 The glassy pillow rim is, of course, produced by rapid quenching of the hot basalt lava immediately as it contacts the cold ocean water, so the excess 40Ar* in the lava is rapidly trapped and retained. Dymond obtained similar results on four deep-sea basalt pillows from near the axis of the East Pacific Rise.168 Dalrymple and Moore also found that the excess 40Ar* contents of the glassy rims of basalt pillows increased systematically with water depth, leading them to conclude that the amount of excess 40Ar* is a direct function of both the hydrostatic pressure and the rate of cooling.169 In a parallel study, Noble and Naughton reported K-Ar “ages” from zero to 22 Ma with increasing sample depth for submarine basalts probably less than 200 years old, also from the active Kilauea volcano.170
Seidemann has reported yet another intriguing relationship.171 He analyzed deep-sea basalt samples obtained from DSDP drillholes in the floor of the Pacific Ocean basin and found that K-Ar “ages” increased with increasing K contents of the basalts, a relationship he noted also appeared in similar data published by DSDP staff (Seidemann172, fig. 1). In basalt pillows the K content increases from the margin to a maximum at an intermediate distance into the pillows, whereas holocrystalline basalts show a decrease of K inward from the margin.173 Seidemann concluded, as had others before him, that submarine weathering adds K to the basalts, as does alteration at the time of formation, whereas the glassy pillow margins are largely impervious to seawater. The net result, however, is unreliable K-Ar “dates,” because the measured 40Ar* was probably not derived by radioactive decay of the measured 40K contents. Seidemann also determined that sediment cover is not a significant barrier to the diffusion of K into basalt.
It is possible that some of these factors are relevant to the pattern of excess 40Ar* measured in the samples from the recent Mt. Ngauruhoe andesite flows. For example, the surfaces of the flows would have cooled more rapidly to crusts on top of the still molten flow interiors, which would certainly have been the case with the August 18, 1954, flow that was reported as being 18 m thick. Furthermore, the overburden pressure within the deep interiors of such thick flows would likewise inhibit degassing of the lava as it cooled. However, so long as the crusts on the tops of the flows remained unbroken and intact they would have sealed in the molten lava and its contained gases, including excess 40Ar*. But this sealing was probably short-lived, because today the flows mostly outcrop as piles of pieces of andesite that look like rubble (typical aa lavas). The continued flow of the lavas down the sides of the volcano would have broken up the crusts as soon as they congealed, as would contraction with cooling, thus enabling the molten interiors to degas as they cooled. So the speed of cooling was likely the most relevant factor, and this would have varied laterally and vertically within the flows, even at localised scales of a few centimeters.
Any effects of weathering on the K contents of these flows can be discounted. On the one hand these are subaerial flows that do not appear to have been subjected to leaching or addition of K, or to any K-rich alteration for that matter, while on the other they have very uniform K contents (see tables 1, 2, and 4). This is not unexpected, given the fact they flowed from the same magma source/chamber close together timewise. The K-Ar data in table 4 do not reveal any discernible relationship between K contents and K-Ar model “ages” of these flows, unlike the negative correlation found in the Middle Proterozoic Cardenas Basalt upper member flows in Grand Canyon, Arizona.174
Perhaps the key issues, though, are where this excess 40Ar* has come from, and whether it has been derived from radioactive decay of 40K. One possibility is that the excess 40Ar* can be accounted for by radioactive decay during long-term residence of magmas in chambers before eruption. Esser, McIntosh, Heizler, and Kyle discounted this option for the Mt. Erebus anorthoclase phenocrysts.175 Dalrymple found that whereas the Mt. Lassen (1915) plagioclase phenocrysts yielded excess 40Ar* and an anomalous K-Ar model “age,” a plagioclase from the 1964 eruption of Surtsey only had argon whose isotopic composition matched that of air.176 Because phenocrysts usually crystallize from lavas after eruption, they may arbitrarily trap excess 40Ar* during lava cooling, 40Ar* that will thus not be from in situ 40K radioactive decay.
Negative K-Ar Model “Ages” and Atmospheric Argon
Another relevant consideration bearing on these issues is the observation noted by Dalrymple that some modern lava samples actually yield negative K-Ar model “ages,” apparently due to excess 36Ar177. Air has an 40Ar/36Ar ratio of 295.5, but some of Dalrymple’s samples had ratios less than 295.5 (and hence negative “ages”). Some of the Mt. Ngauruhoe samples in this study also yielded 40Ar/36Ar ratios less than 295.5 (see table 4). According to the straightforward interpretation of the K-Ar dating methodology, this should be impossible. Dalrymple was not willing to attribute these anomalous ratios to experimental error, and neither was Richard Reesman of Geochron Laboratories.
Dalrymple suggested three possible explanations that might account for the excess 36Ar178:
- incorporation of “primitive argon,”
- production of 36Ar by the radioactive decay of 36Cl, or
- fractionation of atmospheric argon by diffusion.
He rejected the possibility of significant 36Ar formation in situ from nuclear reactions (option 2) because the Cl content of basalts and the production rate of 36Cl by cosmic-ray neutrons both are too low to account for any significant amount of 36Ar. Instead, Dalrymple seemed to favor option 3, that when atmospheric argon diffused back into lavas as they cooled, 36Ar diffused in preferentially. However, he also recognized the weakness of this argument—it is difficult to explain why some lavas are enriched in 36Ar while others are not (as at Mt. Ngauruhoe also). To be consistent, if fractionation of atmospheric argon occurred during diffusion, then this would mean that even supposedly “zero age” lavas actually have an apparent age, and that most lavas do not degas upon eruption. In fact, depending on how strong the fractionation of 36Ar was during diffusion, it could even be that all lavas do not completely degas.
This only leaves Dalrymple’s option 1, that the lavas with the anomalously high 36Ar come from areas of the mantle (and perhaps also the crust) which have primordial argon that has not been diluted with radiogenic 40Ar and have not completely degassed. However, this means that there is no reason to assume that lavas whose argon matches that in the atmosphere have degassed either, because they may have simply started with argon which matches atmospheric argon. Nevertheless, Dalrymple is convinced that “much of the volatile juvenile content may still be present in volcanic rocks quenched on the ocean floor.”179 Indeed, Dalrymple has specifically defined excess 40Ar* as 40Ar that is not attributed to atmospheric argon or in situ radioactive decay of 40K.180 Krummenacher is more cautious, attributing anomalous 40Ar/36Ar ratios and excess 40Ar* to the “mass fractionation effect on argon of atmospheric isotopic composition” trapped in the lavas, as well as to the presence of “magmatic” argon different in isotopic composition.181
The Role of Xenoliths
Is the excess 40Ar* simply “magmatic” argon, that is, argon that collects in the magma and then is inherited by the lavas from it? Funkhouser and Naughton found that the excess 40Ar* in the 1800–1801 Hualalai flow, Hawaii, resided in fluid and gaseous inclusions in olivine, plagioclase, and pyroxene in ultramafic xenoliths in the basalt.182 The quantities of excess 40Ar* were sufficient to yield K-Ar model “ages” from 2.6 Ma to 2960 Ma. However, Dalrymple subsequently only used the presence of the ultramafic xenoliths and their excess 40Ar* contained in inclusions to explain partly the excess 40Ar* and anomalous K-Ar model “ages” he obtained from the same 1800–1801 Hualalai flow, suggesting instead that the large single inclusions are not directly responsible for the excess argon in the flows and that the 40Ar* is distributed more uniformly throughout the rocks.183 Nevertheless, those K-Ar and Ar-Ar geochronologists who are concerned about the excess 40Ar* in their samples undermining their “dating” are careful to check for xenoliths and xenocrysts. Esser, et al., did so and discounted xenocrystic contamination.184
Xenoliths are present in the Ngauruhoe andesite flows (table 3), but they are minor and less significant as the location of the excess 40Ar* residing in these flows than the plagioclase and pyroxene phenocrysts and the much larger glomerocrysts of plagioclase, pyroxene, or plagioclase and pyroxene that predominate. The latter are probably the early-formed phenocrysts that accumulated together in the magma within its chamber prior to eruption of the lava flows. Nevertheless, any excess 40Ar* they might contain had to have been supplied to the magma from its source. The xenoliths that are in the andesite flows have been described by Steiner as gneissic and are therefore of crustal origin, presumably from the basement rocks through which the magma passed on its way to eruption.185
Noble Gases from the Mantle
With the advent of the necessary technology, the isotopic concentrations and ratios of noble gases (including argon) in rock and mineral samples are now obtainable. Honda, et al., have reported such analyses on submarine basalt glass samples from Loihi Seamount and Kilauea, Hawaii, and concluded that helium and neon isotopic ratios in particular, being uniquely different from atmospheric isotopic ratios, are indicative of the mantle source area of the plume responsible for the Hawaiian volcanism rather than from atmospheric contamination of the magma.186, 187 The 40Ar/36Ar ratios are consistent with excess 40Ar* having also come with the magma from the mantle. A subsequent study, in which a larger suite of basalt glass samples, and also samples of olivine phenocrysts, from the same and additional Hawaiian volcanoes were analyzed, concluded that the isotopic systematics indicate that the helium and neon have been derived from the mantle and have not been preferentially affected by secondary processes.188 Consequently, the excess 40Ar* also in these samples would have been also carried from the upper mantle source area of these basalts by the magma plume responsible for the volcanism. Moreira, Kunz, and Allègre have suggested, based on new experimental data from single vesicles in mid-ocean ridge basalt samples dredged from the North Atlantic, that the excess 40Ar* in the upper mantle may be almost double previous estimates (that is, almost 150 times more than the atmospheric content relative to 36Ar), and represents a primordial mantle component not yet outgassed.189, 190 Burnard, Graham, and Turner obtained similar results on the same samples, but maintained that because some of the 36Ar is probably surface-adsorbed atmospheric argon, the upper mantle content of excess 40Ar* could be even ten times higher.191
Similar results have been obtained from ultramafic mantle xenoliths in basalts from the Kerguelen Archipelago in the southern Indian Ocean, and the considerable excess 40Ar* measured concluded to be a part of the mantle source signature of this hotspot volcanism.192 However, it has not only been the suboceanic mantle that has thus been sampled for its excess 40Ar* via such magma plumes. Matsumoto, Honda, McDougall, and O’Reilly have reported high 40Ar/36Ar ratios in spinel-lherzolites from five eruption centers in the youthful (<7 Ma) Newer Volcanics of southeastern Australia.193 These anhydrous lherzolites have compositions representative of the upper lithospheric mantle, and the significant excess 40Ar* in them indicates the presence of a subcontinental mantle reservoir with a very high 40Ar/36Ar ratio, and thus substantial excess 40Ar*, similar to that found in mid-ocean ridge and plume/hotspot basalts. Another example is the Cardenas Basalt and associated diabase (Middle Proterozoic) of eastern Grand Canyon, regarded as part of the pervasive mafic mid-continental magmatism of the southwestern United States and thus also sourced from the subcontinental mantle. Austin and Snelling have found that the 40Ar/36Ar–40K/36Ar isochrons for fourteen and six samples of these rocks, respectively, yield initial 40Ar/36Ar ratios of 787±118 and 453±42, indicative of some initial excess 40Ar*.194
Sampling the Mantle with Diamonds and Their Inclusions
Another means of “sampling” the mantle is the study of diamonds and their micro-inclusions. It is now firmly established that diamonds are thermodynamically stable in the pressure-temperature regime in the mantle at depths greater than 150 km, and their origin is believed to extend back to the Archean and the early crust of the Earth.195, 196 Diamonds are formed in a number of processes associated with two rock types, ecologite and peridotite, xenoliths of which are also brought up into the upper crust with diamonds from the upper mantle below continental Precambrian shields (cratons) by kimberlite and lamproite “pipe” eruptions.197, 198, 199 Even though the host kimberlite or lamproite may be relatively young (even in conventional terms), many diamonds date back to the Archean and thus the early history of the Earth.200, 201 To account for all this evidence, it is postulated that the formation of most diamonds was closely associated with subduction of the Archean oceanic crust into the mantle, the required carbon (which was originally thought to be primordial carbon already in the mantle) now believed to derive from sedimentary marine carbonates and biogenic carbon from bacteria/ algae in the sediments subducted with the oceanic crust.202, 203, 204, 205, 206
The noble gas contents of diamonds are consistent with their ancient and mantle origin, high helium isotopic ratios (290 times the atmospheric ratio) being regarded as primordial and rivaling those measured for the sun today.207, 208 Of significance here is the postulation that He, Ar, K, Pb, Th, and U are added to the convecting upper mantle circulation, and the proportions and isotopic compositions are strongly determined by entrainment from the lower mantle (below 670 km).209, 210 This is reflected in those Ar isotopic measurements that have been made on diamonds and their micro-inclusions.
Rather than focus on attempting to date only diamond micro-inclusions as others had done, Zashu, Ozima, and Nitoh carefully selected ten Zaire diamonds and examined them for purity before undertaking K-Ar dating analyses of the diamonds themselves.211 However, at the outset they noted that there had been almost no direct radiometric dating of diamonds except for conventional K-Ar dating, and the results had been questionable due to the possible presence of excess 40Ar*. To avoid this problem, they used the K-Ar isochron dating method. Their experimental data showed good linear correlations, but these isochrons yielded an age of 6.0±0.3 Ga, which of course was unacceptable because these diamonds would be older than the earth itself. Mistakes in the experimental procedure were easily discounted, so they were forced to conclude that excess 40Ar* was responsible, and that it needed to be in a fluid state to ensure the homogenization necessary to give such a constant 40Ar/K ratio. Alternately, they speculated that the diamonds might differ in K isotopic composition from common potassium, but this was discounted in a follow-up study in which it was found that 40K was present in these diamonds in normal abundance.212 Because 40Ar/39Ar analyses yielded the same unacceptable “age,” it was concluded that the excess 40Ar* was not generated in situ, but was an inherited or “trapped” component from the mantle reservoir when and where the diamonds formed.
These Zaire diamonds are not the only ones which have yielded excess 40Ar*. Phillips, Onstott, and Harris used a laser-probe to 40Ar/39Ar date eclogitic clinopyroxene inclusions in diamonds from the Premier kimberlite, South Africa, and found moderate 40Ar/36Ar ratios indicative of much less excess 40Ar* than in the Zaire diamonds.213 The “age” of these eclogitic diamonds was thus determined to be 1.198±0.014 Ga, much younger than the 3.3 Ga peridotitic diamonds at Kimberley and Finsch, also in South Africa, so Phillips, et al., interpreted the moderate excess 40Ar* as characteristic of mantle conditions prevailing at the time and in the region of Premier eclogitic diamond formation.214
Zashu, et al., postulated that the excess 40Ar* in the Zaire diamonds needed to be in a fluid state.215 Though Navon, Hulcheon, Rossman, and Wasserburg did not analyze for argon when they investigated fluids in micro-inclusions in diamonds from Zaire and Botswana, they found a high content of volatiles and incompatible elements in the uniform average composition of the micro-inclusions, with the amounts of water and CO2 (in carbonates) almost an order of magnitude higher than the volatile contents of kimberlites and lamproites (host rocks to diamonds).216 At 13 wt%, the chlorine levels were also much higher than those of kimberlites (<0.1%), although the bulk composition of the micro-inclusions, including the high K2O content (up to 29.7 wt%), resembled that of such potassic magmas. They concluded that these micro-inclusions represent the volatile-rich (~40% volatiles) fluid or melt from the upper mantle in which the diamonds grew, and that because of the high volatile content in this hydrous mantle fluid, high levels of rare gases may also be expected and explain the high 40Ar/K ratios (the excess 40Ar*) and anomalous “ages.”
As a result of continued investigation of the Zaire cubic diamonds, which produced 40Ar/39Ar age spectra yielding a ~5.7 Ga isochron, Ozima, Zashu, Takigami, and Turner discovered that just as there was an excellent correlation between their potassium contents and 40Ar/36Ar ratios, there is also a correlation between their chlorine contents and 40Ar.217 They concluded from their data
that the 40Ar is an excess component which has no age significance, and that the 40Ar and its associated potassium are contained in sub-micrometer inclusions of mantle-derived fluid.
Turner, Burgess, and Bannon also used the 40Ar/39Ar technique through correlations with K, Cl, and 36Ar to unscramble the mixtures of radiogenic and parentless (excess) Ar components in fluid inclusions in “coated” Zaire diamonds and in olivine from an East African mantle xenolith.218 Their results proved conclusively that 40Ar is present in a widespread chlorine-rich component, which implies the existence of H2O/CO2-rich phases with 40Ar/Cl ratios that are “remarkably uniform over large distances,” with enrichments of these two incompatible elements by almost four orders of magnitude relative to bulk upper-mantle values. Clearly, excess 40Ar* is abundant in the mantle and can be easily transported up into the crust.
Crustal Excess 40Ar*
Is there only evidence for excess 40Ar* in the mantle, gleaned from rocks (basalts and ultramafic xenoliths) and minerals (olivine, pyroxene, plagioclase, and diamonds) that were formed in, or ascended from, the mantle? Patterson, et al., envisage noble gases from the mantle (and the atmosphere) migrating and circulating through the crust, so there should be evidence of excess 40Ar* in crustal rocks and minerals.219 In fact, noble gases in CO2-rich natural gas wells support such migration and circulation—that is, the isotopic signatures clearly indicate a mantle origin for the noble gases, including amounts of excess 40Ar* in some CO2-rich natural gas wells exceeding those in the mantle-derived mid-ocean ridge basalts.220, 221, 222, 223, 224 Staudacher also notes that the quantities of excess 40Ar* in the continental crust can be as much as five times that found in such mantle-derived midocean ridge basalts, strongly suggesting that excess 40Ar* in crustal rocks and their constituent minerals could well be the norm rather than the exception, thus making all K-Ar (and Ar-Ar) dating questionable.225
It has now been established that some diamonds can form in the crust during high-grade metamorphism and via shock metamorphism during meteorite or asteroid impact.226, 227, 228 The pressures and temperatures of high-grade metamorphism had been regarded as insufficient to produce diamonds, but the key ingredient was found to be volatile N2-CO2-rich fluids. Noble gas data on these diamonds are not yet available, due to their size and rarity, but such data have been definitive in establishing the crustal origin of carbonado diamonds.229 Nevertheless, they still contain excess 40Ar*.
Dalrymple, referring to metamorphism and anatexis of rocks in the crust, commented, “If the rock is heated or melted at some later time, then some or all the 40Ar may escape and the K-Ar clock is partially or totally reset.”230 In other words, 40Ar* escapes to migrate in the crust where it may then be incorporated in other minerals as excess 40Ar*, just as 40Ar* degassing from the mantle does. Thus, for example, excess 40Ar* has been recorded in many minerals (some of which contain no 40K) in crustal rocks, such as quartz, plagioclase, pyroxene, hornblende, biotite, olivine, beryl, cordierite, tourmaline, albite, and spodumene in pegmatites, metamorphic rocks, and lavas.231, 232 And it is not just K-Ar dating analyses that detect excess 40Ar*, as Lanphere and Dalrymple used the 40Ar/39Ar method to confirm the presence of excess 40Ar* in feldspars and pyroxenes.233 Indeed, in a recent study, 128 Ar isotopic analyses were obtained from ten profiles across biotite grains in amphibolite-granulite facies metamorphic rocks, and apparent 40Ar/39Ar “ages” within individual grains ranged from 161 to 514 Ma.234 The investigators concluded that these observations cannot be solely due to radiogenic build-up of 40Ar*, but must be the result of incorporation by diffusion of excess 40Ar* from an external source, namely, 40Ar* from the mantle and other crustal rocks and minerals. Indeed, Harrison and McDougall were able to calculate a well-defined law for 40Ar diffusion from hornblende in a gabbro due to heating.235 They also found that the excess 40Ar* which had developed locally in the intergranular regions of the host gabbro reached partial pressures in some places of at least 10-2 atm.
This crustal migration of 40Ar* is known to cause grave problems in attempted regional geochronology studies. In the Middle Proterozoic Musgrave Block of northern South Australia, Webb found a wide scatter of K-Ar mineral ages ranging from 343 Ma to 4493 Ma due to inherited (or excess) 40Ar*, so that no meaningful interpretation could be drawn from the rocks (granulite, gneiss, pseudotrachylite, migmatite, granite, and diabase).236 Of the diabase dikes which gave anomalous ages, he concluded that
The basic magmas probably formed in or passed through zones containing a high partial pressure of 40Ar*, permitting inclusion of some of the gas in the crystallizing minerals.
Likewise, when Baski and Wilson attempted to argon date Proterozoic granulite-facies rocks in the Fraser Range (Western Australia) and Strangways Range (central Australia), they found that garnet, sapphirine, and quartz in those rocks contained excess 40Ar* that rendered their argon dating useless because of “ages” higher than expected.237 They also concluded that the excess 40Ar* was probably incorporated at the time of formation of the minerals, and their calculations suggested a partial pressure of ~0.1 atm Ar in the Proterozoic lower crust of Australia, which extends over half the continent.
In a detailed 40Ar/39Ar dating study of high-grade metamorphic rocks in the Broken Hill region of New South Wales (Australia), Harrison and McDougall found evidence of widely distributed excess 40Ar*.238 The minerals most affected were plagioclase and hornblende, with step heating 40Ar/39Ar “age” spectra yielding results of up to 9.588 Ga. Such unacceptable “ages” were produced by excess 40Ar* release, usually at temperatures of 350–650°C and/or 930–1380°C, suggesting the excess 40Ar* is held in sites within the respective mineral lattices with different heating requirements for its release. There are three principal trapping sites for Ar in solids: structural holes, edge dislocations, and lattice vacancies. (Argon is also known to be held sometimes in some minerals in fluid inclusions.) Clearly, this study shows that at crustal temperatures, which are less than 930°C, some excess 40Ar* will always be retained in those trapping sites in minerals where it is obviously “held” more tightly, thus rendering K-Ar and 40Ar/39Ar dating questionable. Harrison and McDougall were only able to produce a viable interpretation of the data because they made assumptions about the expected age of the rocks and of a presumed subsequent heating event (based on Pb-Pb and Rb-Sr dating), the latter being the time when they conjecture that accumulated 40Ar* was released from minerals causing a significant regional Ar partial pressure of ~3x10-4 atm to develop.239
Mantle-Crust Domains and Excess 40Ar*
Harte and Hawkesworth have identified domains within the mantle and crust and described the interaction between them, all of which is relevant to the migration and circulation of argon (and thus excess 40Ar*) from the lower mantle to the crust and to lavas extruded on the earth’s surface.240 The six domains are physically distinct units which show wide differences in average physical and chemical properties, as well as apparent age, structure, and tectonic behavior. They are the lower mantle (below 670 km), upper mantle, continental mantle lithosphere, oceanic mantle lithosphere, continental crust, and oceanic crust, and each is a distinct geochemical reservoir. Each domain may provide material for magmatic rocks, and particular geochemical features of magmas may be associated with particular domains. Thus the convecting upper mantle which comes to the surface at mid-ocean ridges may be identified as the source of most geochemical features of midocean ridge basalts, including their excess 40Ar* content. Similarly, the convecting lower mantle is regarded as the primordial or bulk earth geochemical reservoir, which may also contribute excess 40Ar* to mid-ocean ridge basalts, but is more important for its contribution to ocean island basalts (for example, Hawaii) and other plume-related basalts (continental alkali basalts and continental flood basalts). However, considerable complexity may be added to the deeper mantle geochemical structure as a result of localized accumulation of subducted oceanic lithosphere.
Porcelli and Wasserburg have proposed a steady-state upper mantle model for mass transfer of rare gases, including argon.241 The rare gases in the upper mantle are derived from mixing of rare gases from the lower mantle, subducted rare gases, and radiogenic nuclides produced in situ. Porcelli and Wasserburg claim that all of the 40Ar in the closed-system lower mantle has been produced by 40K decay in the lower mantle, but this claim is based on the assumption of a 4.5 Ga earth. In any case, they contradict themselves, because they also state, “The lower mantle is assumed to have evolved isotopically approximately as a closed system with the in situ decay of 129I, 244Pu, 238U, 232Th, and 40K adding to the complement of initial rare gases.”242 In other words, they admit that some of the 40Ar must be primordial and not derived from radioactive 40K. They then go on to claim that in the upper mantle, 40K decay further increases the radiogenic 40Ar from the lower mantle by a factor of ~3, but again this presupposes a 4.5 Ga earth and doesn’t allow for primordial 40Ar that could well also be in the upper mantle if it is admitted to be in the lower mantle.
In the case of the continental and oceanic lithospheric domains, the lack of convective stirring means that different geological processes and events may implant in each domain a variety of geochemically distinct materials that will remain isolated from one another. Therefore, these domains do not have a single set of geochemical characteristics; thus identification of geochemically defined “sources” with particular physically defined crust-mantle domains is complex, and the geochemical definition of particular reservoirs cannot be regarded as simply definition of major physical entities. Nevertheless, excess 40Ar* will be added to these domains by the passage of basaltic magma plumes from the upper mantle to the earth’s surface.
Furthermore, the processes of oceanic lithosphere formation from the convecting upper mantle in association with mid-ocean ridge activity mean that its isotopic characteristics everywhere will be largely similar to those of the convecting upper mantle and mid-ocean ridge basalts, including the addition of excess 40Ar*. The corollary to this is that the oceanic crust is formed as part of these same processes. However, the oceanic crust generally has a thin veneer of sediments over it, and thick wedges of sediments adjacent to the domains of continental crust, whereas sections of oceanic crust are hydrothermally altered. The compositions of these components of the oceanic crust may, therefore, include a considerable contribution from continental detritus and ocean water, so that this oceanic crustal material may give rise to a distinct geochemical reservoir, the fate of which during subduction back into the upper mantle becomes critically important if it contributes to island arc volcanics, plume-related intra-place magmas, and mantle-derived xenoliths.
The complexity of continental crustal material is well known through direct observation, and the mantle lithosphere attached to it may be expected to show a similar complexity. Nevertheless, it is evident that excess 40Ar* also resides in the continental mantle lithosphere, as indicated by xenoliths.243 Likewise, there is evidence of excess 40Ar* in crustal magmatic rocks (for example, gabbros, pegmatites, migrating through metamorphic terrains, and in natural gas in sedimentary reservoirs.244, 245, 246, 247, 248, 249
Mt. Ngauruhoe in its Tectonic Framework
The presence, therefore, of excess 40Ar* in the recent andesite flows at Mt. Ngauruhoe is to be expected. The Taupo Volcanic Zone is a volcanic arc and marginal basin of the Taupo-Hikurangi arc-trench system (see fig. 1 again), which is a southward extension of the Tonga-Kermadec arc into the continental crustal environment of New Zealand’s North Island.250 Geophysical investigations indicate that the Pacific Plate is being obliquely subducted beneath the Australian Plate on which most of New Zealand’s North Island sits, and that the volcanoes of the Taupo Volcanic Zone, including Ngauruhoe in the Tongariro Volcanic Center, are about 80 km directly above the subducting Pacific Plate, a zone of earthquakes revealing where the movement is taking place.251 Friction along the plane of contact is believed to cause melting to produce pockets of magma, which then feed via conduits to the volcanoes above. Thus the recent andesite flows at Mt. Ngauruhoe are calcalkaline island arc volcanics.
The tectonic and geochemical framework of the Ngauruhoe andesite flows within the mantle-crust domains of Harte and Hawkesworth is that of subducting oceanic crust (derived from the convecting upper mantle), carrying with it the wedge of continental sedimentary detritus which has accumulated at the continental margin and in the adjacent trench to the east of the coastline.252 Attached beneath the subducting oceanic crust is its associated oceanic mantle lithosphere, and together they are being thrust downwards into the upper mantle. Above the subducting plate are the continental crust and continental mantle lithosphere of the overriding plate, the continental crust being at the contact plane at shallow depths near the trench, and then the attached continental mantle lithosphere beneath at a depth of about 35 km.253 Thus the geochemical reservoir from which the Ngauruhoe andesite magma has been drawn is potentially a mixture of melted oceanic crust, continental sedimentary detritus and continental crust, and possibly continental mantle lithosphere, or even upper mantle.
Genesis of the Mt. Ngauruhoe Andesite Magma and its Excess 40Ar*
One of the easier investigations of the petrogenesis of these volcanic rocks of the Taupo Volcanic Zone was that of Stipp and of Ewart and Stipp.254, 255 They analyzed samples that had been systematically collected, including not only the lavas and the pyroclastics, but also the Permian to Jurassic interbedded greywackes, siltstones, and shales (the potential crustal source rocks), which are spatially related to, and underlie, the volcanics. Of primary interest were Sr, Rb, and K contents, and 87Sr/86Sr and 87Rb/86Sr ratios. Three possibilities for the origin of the calc-alkaline andesite magma were under investigation: fractional crystallization of a basalt magma under oxidizing conditions; some form of hybridization between basaltic and acidic magmas, possibly followed by fractional crystallization; and derivation of a primary andesite magma from the upper mantle. Ewart and Stipp regarded their Sr isotopic data as more consistent with the production of the andesites by partial assimilation of sedimentary material by basaltic magma (derived from the upper mantle), the adjacent greywackes, siltstones, and shales being the most likely sedimentary material, and the unassimilated gneissic xenoliths probably representing the basement rocks to those sediments.256 However, they admitted that the data did not exclude the possibility of a primary andesitic magma derived directly from the upper mantle, provided that some assimilation of crustal material modified it prior to eruption.
Subsequent investigations by Cole favored the alternate petrogenetic model of a primary andesitic magma.257 He suggested that the subducting oceanic crust assimilated the greywacke-siltstone-shale and overlying sediments east of the Taupo Volcanic Zone to produce amphibolite, which subsequently broke down to produce phlogopite eclogite below 90 km. This in turn partially melted at 150–200 km, and the resultant magma fractionated in the upper mantle or lower crust to produce andesite. However, based on rare-earth element geochemistry, Cole, Cashman, and Rankin modified that petrogenetic model, suggesting that while the andesite magma genesis was probably associated in the upper mantle with the downgoing slab and some crustal contamination occurred, the andesite does not appear to have had an eclogite parent.258 This would then suggest that the melting associated with the subducting slab to generate the andesite magma occurred at a depth of less than 90 km.
Graham and Hackett agreed with this conclusion, demonstrating from geophysical evidence that the top of the subducting slab is at a depth of about 80 km below Ngauruhoe, and that the crust there is probably less than 20 km thick.259 Thus the upper mantle wedge between would consist only of plagioclase-peridotite and spinelperidotite. At 80 km depth the hydrated amphibolite assemblage of the upper portion of the subducting slab of oceanic crust and oceanic mantle lithosphere would have started to dehydrate, thus liberating water and possibly other volatile constituents into the overlying upper mantle wedge, significantly lowering its melting point. Graham and Hackett then showed that the geochemical evidence requires the andesite magma for the Ngauruhoe lava flows to have been generated from an original low-alumina basalt magma produced in the upper mantle wedge by anatexis of the asthenosphere (uppermost mantle) and/or subcontinental mantle lithosphere probably catalyzed by hydrous, metasomatic fluids from the subducting slab.
Some specific geochemical enrichment then appears to have occurred as a result of this mantle metasomatism and continental crustal contamination during ascent and storage of the magma. Graham and Hackett used least squares geochemical modeling to show how the andesite magma could be generated from such a parent basalt magma by a process of combined assimilation of crustal material (addition of 6% assimilant) and fractional crystallization (30% removal of crystals).260 Furthermore, the presence of xenoliths in the Ngauruhoe andesite flows, particularly the vitrified meta-greywacke and gneissic xenoliths, indicate conclusively that the assimilant was most likely a partial melt of gneiss, originally the adjacent greywacke-siltstone-shale sediments.261, 262
These processes responsible for the generation of the andesite magma did not diminish the excess 40Ar* content of the resultant flows. Though the amount of excess 40Ar* is not high when compared with that found in mid-ocean ridge basalts, it is nonetheless significant that the excess 40Ar* was still present in the lavas upon eruption and cooling. The evidence indicates that the parent basaltic magma was generated in the upper mantle where the excess 40Ar* in the geochemical reservoir is now known to be upwards of 150 times more than the atmospheric content, relative to 36Ar. The subsequent crustal contamination and fractional crystallization to form the andesite magma during ascent, and the degassing of the magma during eruption and lava flow and cooling, did not remove all the excess 40Ar*, a small portion of which was left to be trapped in the congealed lava and its constituent minerals.
This model for the generation of the andesite magma in the post-Flood world is, of course, based on the plate tectonics model for global tectonics through earth history. Even though the postulated plate movements today are extremely slow, and thus extrapolated back over millions of years by uniformitarians, a catastrophic model for plate tectonics in the context of the Flood is entirely compatible with both Scripture and the scientific data.263 Plate movements are regarded as occurring catastrophically during the Flood and then rapidly slowing down to today’s rates in the post-Flood era.
Conclusions
The fact that there is even some excess 40Ar* in these recent andesite flows, and that it appears to have ultimately come from the upper mantle geochemical reservoir—where it is regarded as leftover primordial argon not yet fully expelled by the process of outgassing that is supposed to have occurred since the initial formation of the earth—has very significant implications.
First, this is clearly consistent with a young earth, where the very short time scale since the creation of the earth has been insufficient for all the primordial argon to be released yet from the earth’s deep interior. Furthermore, it would also seem that even the year-long global catastrophic Flood, when large-scale convection and turdecer occurred in the mantle, was insufficient to expel all the deep earth’s primordial argon.264
Second, this primordial argon is, in part, “excess” 40Ar not generated by radioactive decay of 40K, which has then been circulated up into crustal rocks where it may continue migrating and building up to partial pressure status regionally. Because the evidence clearly points to this being the case, when samples of crustal rocks are analyzed for K-Ar “dating” the investigators can never really be sure that whatever 40Ar* is in the samples is from in situ radioactive decay of 40K since the formation of the rocks, or whether some or all of it is from the “excess 40Ar*” geochemical reservoirs in the lower and upper mantles. This could even be the case when the K-Ar analyses yield “dates” compatible with other radioisotopic “dating” systems and/or with fossil “dating” based on evolutionary assumptions. And there would be no way of knowing because the 40Ar* from radioactive decay of 40K cannot be distinguished analytically from primordial 40Ar that is not from radioactive decay, except, of course, by external assumptions about the ages of the samples.
Therefore, these considerations call into question all K-Ar “dating,” whether “model ages” or “isochron ages,” and all 40Ar/39Ar “dating,” as well as “fossil dating” that has been calibrated against K-Ar “dates.” Although seemingly insignificant in themselves, the anomalous K-Ar “model ages” for these recent andesite flows at Mt. Ngauruhoe, New Zealand, lead to deeper questions. Why is there excess40Ar* in these rocks? From where did it come? Answers to these questions in turn point to significant implications that totally undermine such radioactive “dating” and that are instead compatible with a young earth.
Future Research
Further research is very definitely warranted. The most pressing need is to attempt to quantify how much primordial 40Ar there is today in the upper mantle, as well as how much has circulated into crustal rocks, how much is in natural gas reservoirs, and how much might have escaped into the atmosphere during 6,000–7,000 years (including at accelerated rates during the Flood). It might then be possible to quantify how much primordial 40Ar there was in the mantle at the time of the earth’s creation. From these calculations and associated modeling exercises there might develop quantifiable evidence for the earth’s youth.
Additionally, further research is needed to quantify how much “excess 40Ar*” is in all the crustal rocks and minerals that have been, and are, subject to K-Ar and 40Ar/39Ar “dating.” This would include what are regarded as mantle xenoliths and xenocrysts (for example, diamonds). It is helpful to show on the one hand that such “dating” is questionable, but on the other hand there are still many “dates” that are concordant, that is, they agree with other uniformitarian dating systems and schemes. So ultimately we need to explain this agreement when other “dates” are discordant and anomalous. There may, in fact, be some pattern or systematic way in which “excess 40Ar” has been trapped in rocks and occluded in minerals at different levels (depths and relative ages) in the geological record. If so, then K-Ar and 40Ar/39Ar “dating” would irrevocably be discredited.
Footnotes
- Graham, I. J. and W. R. Hackett, 1987. Petrology of calc-alkaline lavas from Ruapehu Volcano and related vents, Taupo Volcanic Zone, New Zealand. Journal of Petrology 28(3):531–567.
- Williams, K., 1994. Volcanoes of the South Wind: A field guide to the volcanoes and landscape of the Tongariro National Park. Turangi, New Zealand: Tongariro Natural History Society.
- Hackett, W. R. and B. F. Houghton, 1987. Active composite volcanoes of Taupo Volcanic Zone. Central North Island volcanism. New Zealand geological survey record 21, pp. 61–114.
- Williams, Ref. 2.
- Cole, J. W. and K. B. Lewis, 1981. Evolution of the Taupo-Hikurangi subduction system. Tectonophysics 72:121.
- Grindley, G. W., 1960. Geological map of New Zealand 1:250,000, Sheet 8, Taupo. New Zealand Geological Survey.
- Graham and Hackett, Ref. 1.
- Cole, J. W., I. J. Graham, W. R. Hackett, and B. F. Houghton, 1986. Volcanology and petrology of the Quaternary composite volcanoes of Tongariro Volcanic Centre, Taupo Volcanic Zone, Late Cenozoic volcanism in New Zealand. In Smith, I. E. M. (ed.), Royal Society of New Zealand bulletin, vol. 23, pp. 224–250.
- Hackett and Houghton, Ref. 3.
- Gregg, D. R., 1960. The geology of Tongariro subdivision. New Zealand Geological Survey Bulletin, n.s. 40.
- Nairn, I. A. and C. P. Wood, 1987. Active volcanoes of Taupo volcanic zone, active volcanoes and geothermal systems, Taupo Volcanic Zone. New Zealand Geological Survey Record 22:5–84.
- Gregg, Ref. 10.
- Grindley, G.W., 1965. Tongariro National Park, stratigraphy and structure. New Zealand Department of Scientific and Industrial Research Information Series 50:79–86.
- Nairn and Wood, Ref. 11.
- Graham and Hackett, Ref. 1.
- Grindley, Ref. 13.
- Cole, et al., Ref. 8.
- Stipp, J. J., 1968. The Geochronology and Petrogenesis of the Cenozoic Volcanics of the North Island, New Zealand. Unpublished PhD thesis, Australian National University, Canberra.
- Cole, et al., Ref. 8.
- Topping, W. W., 1973. Tephrostratigraphy and chronology of Late Quaternary eruptives from the Tongariro volcanic centre, New Zealand. New Zealand Journal of Geology and Geophysics 16:397–423.
- Grindley, Ref. 13.
- Nairn and Wood, Ref. 11.
- Williams, Ref. 2.
- Gregg, Ref. 10.
- Nairn and Wood, Ref. 11.
- Williams, Ref. 2.
- Gregg, Ref. 10.
- Gregg, Ref. 10.
- Nairn and Wood, Ref. 11.
- Nairn and Wood, Ref. 11.
- Gregg, Ref. 10.
- Nairn and Wood, Ref. 11.
- Battey, M. H., 1949. The recent eruption of Ngauruhoe. Records of the Auckland Institute and Museum 3:389–395.
- Gregg, Ref. 10.
- Gregg, Ref. 10.
- Nairn and Wood, Ref. 11.
- Gregg, Ref. 10.
- Williams, Ref. 2.
- Gregg, D. R., 1956. Eruption of Ngauruhoe 1954–55. New Zealand Journal of Science and Technology B37:675–688.
- Gregg, Ref.10.
- Cole, et al., Ref. 8.
- Hackett and Houghton, Ref. 9.
- Gregg, Ref. 39.
- Gregg, Ref. 10.
- Williams, Ref. 2.
- Gregg, Ref. 39.
- Gregg, Ref. 10.
- Battey, Ref. 33.
- Gregg, Ref. 39.
- Gregg, Ref. 10.
- Nairn, I. A. and S. Self, 1978. Explosive eruptions and pyroclastic avalanches from Ngauruhoe in February 1975. Journal of Volcanology and Geothermal Research 3:39–60.
- Nairn and Wood, Ref. 11.
- Nairn and Wood, Ref. 11.
- Nairn and Wood, Ref. 11.
- Williams, Ref. 2.
- Nairn and Wood, Ref. 11.
- Nairn, I. A., C. A. Y. Hewson, J. H. Latter, and C. P. Wood, 1976. Pyroclastic eruptions of Ngauruhoe Volcano, central North Island, New Zealand, 1974 January and March. In, Johnson, R. W. (ed.), Volcanism in Australasia, pp. 385–405. Amsterdam: Elsevier.
- Nairn, I. A., 1976. Atmospheric shock waves and condensation clouds from Ngauruhoe explosive eruptions. Nature 259: 190–192.
- Nairn and Self, Ref. 51.
- Nairn and Wood, Ref. 11.
- Williams, Ref. 2.
- Nairn and Self, Ref. 51.
- Nairn and Wood, Ref. 11.
- Williams, Ref. 2.
- Nairn and Wood, Ref. 11.
- Williams, Ref. 2.
- Nairn and Self, Ref. 51.
- Dalrymple, G. B. and M. A. Lanphere, 1969. Potassium-argon dating: Principles, techniques and applications to geochronology. San Francisco: W. H. Freeman.
- Dalrymple and Lanphere, Ref. 68.
- Clark, R. H., 1960. Petrology of the volcanic rocks of Tongariro subdivision. In, Gregg, D. R. (ed.), The geology of the Tongariro subdivision, pp. 107–123. New Zealand Geological Survey Bulletin n.s.40.
- Gill, J. B., 1981. Orogenic andesites and plate tectonics. Berling: Springer-Verlag.
- Nairn, et al., Ref. 57.
- Clark, Ref. 70.
- Hackett and Houghton, Ref. 9.
- Steiner, A., 1958. Petrogenetic implications of the 1954 Ngauruhoe lava and its xenoliths. New Zealand Journal of Geology and Geophysics 1:325–363.
- Clark, Ref. 70.
- Hackett and Houghton, Ref. 9.
- Steiner, Ref. 75.
- Clark, Ref. 70.
- Ewart, A. and J. J. Stipp, 1968. Petrogenesis of the volcanic rocks of the central North Island, New Zealand, as indicated by a study of Sr87/Sr86 ratios, and Sr, Rb, K, U and Th abundances. Geochimica et Cosmochimica Acta 32:699–736.
- Graham and Hackett, Ref. 1.
- Cole, et al., Ref. 8.
- Nairn and Wood, Ref. 11.
- Nairn, et al., Ref. 57.
- Hackett and Houghton, Ref. 9.
- Nairn, et al., Ref. 57.
- Hackett and Houghton, Ref. 9.
- Hackett and Houghton, Ref. 9.
- Nairn, et al., Ref. 57.
- Clark, Ref. 70.
- Cole, J. W., 1978. Andesites of the Tongariro Volcanic Centre, North Island, New Zealand. Journal of Volcanology and Geothermal Research 3:121–153.
- Graham, I. J., 1985. Petrochemical and Sr isotopic studies of lavas and xenoliths from Tongariro Volcanic Centre implications for crustal contamination in calc-alkaline magmas. Unpublished Ph.D. Thesis, Victoria University of Wellington.
- Cole, et al., Ref. 8.
- Gill, Ref. 71.
- Cole, et al., Ref. 8.
- Cole, et al., Ref. 8.
- Cole, Ref. 91.
- Cole, et al., Ref. 8.
- Steiner, Ref. 75.
- Battey, Ref. 33.
- Graham, I. J., J. W. Cole, R. M. Briggs, J. A. Gamble, and I. E. M. Smith, 1995. Petrology and petrogenesis of volcanic rocks from the Taupo Volcanic Zone: A review. Journal of Volcanology and Geothermal Research 68:59–87.
- Cole, Ref. 91.
- Cole, J. W., K. V. Cashman, and P. C. Rankin, 1983. Rare-earth element geochemistry and the origin of andesites and basalts of the Taupo Volcanic Zone, New Zealand. Chemical Geology 38:255–274.
- Cole, et al., Ref. 8.
- Cole, et al., Ref. 103.
- Cole, et al., Ref. 8.
- Ewart, A., 1971. Notes on the chemistry of ferromagnesian phenocrysts from selected volcanic rocks, Central Volcanic Region. New Zealand Journal of Geology and Geophysics 14(2):323–340.
- Cole, Ref. 91.
- Graham and Hackett, Ref. 1.
- Clark, Ref. 70.
- Cole, Ref. 91.
- Cole, et al., Ref. 8.
- Graham, et al., Ref. 101.
- Cole, et al., Ref. 8.
- Graham, et al., Ref. 101.
- Faure, G., 1986. Principles of isotope geology, 2nd ed. New York: John Wiley and Sons.
- Dickin, A. P., 1995. Radiogenic isotope geology. Cambridge: Cambridge University Press.
- Geyh, M. A. and H. Schleicher, 1990. Absolute dating methods. Berlin: Springer-Verlag.
- Dalrymple and Lanphere, Ref. 68.
- Dalrymple, G. B., 1991. The age of the earth. Stanford: Stanford University Press.
- Dalrymple, G. B., 1969. 40Ar/36Ar analyses of historic lava flows. Earth and Planetary Science Letters 6:47–55.
- Dalrymple, Ref. 120.
- Dalrymple, Ref. 121.
- Dalrymple, Ref. 121.
- Krummenacher, D., 1970. Isotopic composition of argon in modern surface volcanic rocks. Earth and Planetary Science Letters 8:109–117.
- Krummenacher, Ref. 125.
- Krummenacher, Ref. 125.
- Krummenacher, Ref. 125.
- Krummenacher, Ref. 125.
- Krummenacher, Ref. 125.
- McDougall, I., H. A. Polach, and J. J. Stipp, 1969. Excess radiogenic argon in young subaerial basalts from the Auckland volcanic field, New Zealand. Geochimica et Cosmochimica Acta 33:1485–1520.
- Fisher, D. E., 1971. Excess rare gases in a subaerial basalt from Nigeria. Nature 232:60–61.
- Armstrong, R. L., 1978. K-Ar dating: Late Cenozoic McMurdo volcanic group and dry valley glacial history, Victoria Land, Antarctica. New Zealand Journal of Geology and Geophysics 21(6):685–698.
- Esser, R. P., W. C. McIntosh, M. T. Heizler, and P. R. Kyle, 1997. Excess argon in melt inclusions in zero-age anorthoclase feldspar from Mt Erebus, Antarctica, as revealed by the 40Ar/39Ar method. Geochimica et Cosmochimica Acta 61: 3789–3801.
- Noble, C. S. and J. J. Naughton, 1968. Deep-ocean basalts: Inert gas content and uncertainties in age dating. Science 162: 265–267.
- Dalrymple, G. B. and J. G. Moore, 1968. Argon 40: Excess in submarine pillow basalts from Kilaueau Volcano, Hawaii. Science 161:1132–1135.
- Dalrymple and Moore, Ref. 136.
- Funkhouser, J. G., D. E. Fisher, and E. Bonatti, 1968a. Excess argon in deep-sea rocks. Earth and Planetary Science Letters 5:95–100.
- Funkhouser, J. G., I. L. Barnes, and J. J. Naughton, 1966. Problems in the dating of volcanic rocks by the potassium-argon method. Bulletin of Volcanology 29:709–717.
- Fisher, D. E., 1972. U/He ages as indicators of excess argon in deep sea basalts. Earth and Planetary Science Letters 14: 255–258.
- Dymond, J., 1970. Excess argon in submarine basalt pillows. Geological Society of America Bulletin 81:1229–1232.
- Fisher, D. E., 1970. Heavy rare gases in a Pacific seamount. Earth and Planetary Science Letters 9:331–335.
- McDougall, I., 1971. The geochronology and evolution of the young volcanic island of Réunion, Indian Ocean. Geochimica et Cosmochimica Acta 35:261–288.
- Honda, M., I. McDougall, D. B. Patterson, A. Doulgeris, and D. A. Clague, 1993. Noble gases in submarine pillow basalt glasses from Loihi and Kilauea, Hawaii: A solar component in the earth. Geochimica et Cosmochimica Acta 57:859–874.
- Valbracht, P. J., H. Staudigel, M. Honda, I. McDougall, and G. R. Davies, 1996a. Isotopic tracing of volcanic source regions from Hawaii: Decoupling of gaseous from lithophile magma components. Earth and Planetary Science Letters 144:185–198.
- Marty, B. and F. Humbert, 1997. Nitrogen and argon isotopes in oceanic basalts. Earth and Planetary Science Letters 152:101–112.
- Staudacher, T., P. Sarda, S. H. Richardson, C. J. Allègre, I. Sagna, and L. V. Dimitrieu, 1989. Noble gases in basalt glasses from a mid-Atlantic ridge topographic high at 14N: Geodynamic consequences. Earth and Planetary Science Letters 96:119–133.
- Patterson, D. B., M. Honda, and I. McDougall, 1990. Atmospheric contamination: A possible source for heavy noble gases in basalts from Loihi Seamount, Hawaii. Geophysical Research Letters 17:705–708.
- Honda et al., Ref.144.
- Valbrachet, et al., Ref.145.
- Austin, S. A., 1996. Excess argon within mineral concentrates from the new dacite lava dome at Mount St. Helens volcano. Creation Ex Nihilo Technical Journal 10(3):335–343.
- Laughlin, A. E., J. Poths, H. A. Healey, S. Reneau, and G. Wolde Gabriel, 1994. Dating of Quaternary basalts using the cosmogenic 3He and 14C methods with implications for excess 40Ar. Geology 22:135–138.
- Patterson, D. B., M. Honda, and I. McDougall, 1994. Noble gases in mafic phenocrysts and xenoliths from New Zealand. Geochimica et Cosmochimica Acta 58:4411–4427.
- Poths, J., H. Healey, and A. W. Laughlin, 1993. Ubiquitous excess argon in very young basalts. Geological Society of America Abstracts with Programs 25:A462.
- Damon, P. E., A. W. Laughlin, and J. K. Precious, 1967. Problem of excess argon-40 in volcanic rocks. Radioactive dating methods and low-level counting, pp. 463–481. Vienna: International Atomic Energy Agency.
- Dalrymple, Ref. 121.
- Broadhurst, C. L., M. J. Drake, B.E. Hagee, and T. J. Benatowicz, 1990. Solubility and partitioning of Ar in anorthite, diopside, fosterite, spinel, and synthetic basaltic liquids. Geochimica et Cosmochimica Acta 54:299–309.
- Broadhurst, C. L., M. J. Drake, B. E. Hagee, and T. J. Benatowicz, 1992. Solubility and partitioning of Ne, Ar, Kr and Xe in minerals and synthetic basaltic melts. Geochimica et Cosmochimica Acta 56:709–723.
- Broadhurst, et al., Ref. 157.
- Karpinskaya, T. B., I. A. Ostrovskiy, and L. L. Shanin, 1961. Synthetic introduction of argon into mica at high pressures and temperatures. Isv Akad Nauk S.S.S.R. Geology Series 8:87–89.
- Karpinskaya, T. B., 1967. Synthesis of argon muscovite. International Geology Review 9:1493–1495.
- Austin, Ref. 151.
- Poths, et al., Ref. 154.
- Laughlin, et al., Ref. 152.
- Patterson, et al., Ref. 153.
- Valbrachet, et al., Ref. 145.
- Dalrymple and Moore, Ref. 136.
- Dymond, Ref. 141.
- Dalrymple and Moore, Ref. 136.
- Noble and Naughton, Ref. 135.
- Seidemann, D. E., 1977. Effects of submarine alteration on K-Ar dating of deep-sea igneous rocks. Geological Society of America Bulletin 88:1660–1666.
- Seidemann, Ref. 171.
- Hart, S. R., 1969. K, Rb, Cs Contents and K/Rb, K/Cs ratios of fresh and altered submarine basalts. Earth and Planetary Science Letters 6:295–303.
- Austin, S.A. and A. A. Snelling, 1998. Discordant potassium-argon model and isochron “ages” for Cardenas Basalt (Middle Proterozoic) and associated diabase of Eastern Grand Canyon, Arizona. In, Walsh, R. E. (ed.), Proceedings of the fourth international conference on creationism, pp. 35–51. Pittsburgh, Pennsylvania: Creation Science Fellowship.
- Esser, et al., Ref. 134.
- Dalrymple, Ref. 121.
- Dalrymple, Ref. 121.
- Dalrymple, Ref. 121.
- Dalrymple and Moore, Ref. 136.
- Dalrymple and Lanphere, Ref. 68.
- Krummenacher, Ref. 125.
- Funkhouser, J. G. and J. J. Naughton, 1968. Radiogenic helium and argon in ultramafic inclusions from Hawaii. Journal of Geophysical Research 73:4601–4607.
- Dalrymple, Ref. 121.
- Esser, et al., Ref. 134.
- Steiner, Ref. 75.
- Honda et al., Ref. 144.
- Patterson, Honda, and McDougall, Ref. 148.
- Valbrachet, et al., Ref.145.
- Moreira, M., J. Kunz, and C. Allègre, 1998. Rare gas systematics in popping rock: Isotopic and elemental compositions in the upper mantle. Science 279:1178–1181.
- Staudacher, et al., Ref. 147.
- Burnard, P., D. Graham, and G. Turner, 1997. Vesicle-specific noble gas analyses of “popping rock”: Implications for primordial noble gases in earth. Science 276:568–571.
- Valbracht, P. J., M. Honda, T. Matsumoto, N. Mattielli, I. McDougall, R. Ragettli, and D. Weis, 1996b. Helium, neon and argon isotope systematics in Kerguelen ultramafic xenoliths: Implications for mantle source signatures. Earth and Planetary Science Letters 138:29–38.
- Matsumoto, T., M. Honda, I. McDougall, and S. Y. O’Reilly, 1997. Noble gases in anhydrous lherzolites from the newer volcanics, southeastern Australia: A MORB-like reservoir in the subcontinental mantle. Research School of Earth Sciences annual report 1997, pp. 156–157. Canberra: Australian National University.
- Austin and Snelling, Ref. 174.
- Kesson, S. E. and A. E. Ringwood, 1989. Slab-mantle interactions 2. The formation of diamonds. Chemical Geology 78:97–118.
- Kirkley, M. B., J. J. Gurney, and A. A. Levinson, 1992. Age, origin and emplacement of diamonds: A review of scientific advances in the last decade. Canadian Institute of Mining Bulletin 84:48–57.
- Gurney, J. J., 1986. Diamonds. Kimberlites and related rocks, Vol. 2: Their mantle/crust setting, diamonds and diamond exploration. Proceedings of the fourth international kimberlite conference, Geological Society of Australia special publication no. 14, pp. 935–965.
- Gurney, J. J., 1990. The diamondiferous roots of our wandering continent. South African Journal of Geology 93:423–437.
- Kirkley, Gurney, and Levinson, Ref. 196.
- Richardson, S. H., J. J. Gurney, A. J., Erlank, and J. W. Harris, 1984. Origin of diamonds in old enriched mantle. Nature 310:198–202.
- Kirkley, Gurney, and Levinson, Ref. 196.
- Kesson and Ringwood, Ref. 195.
- Kirkley, Gurney, and Levinson, Ref. 196.
- Eldridge, C. S., W. Compston, I. S. Williams, J. W. Harris, and J. W. Bristow, 1991. Isotope evidence for the involvement of recycled sediments in diamond formation. Nature 353:649–653.
- Kirkley, M. B., J. J. Gurney, M. L. Otter, S. J. Hill, and L. R. Daniels, 1991. The application of C isotope measurements to the identification of the sources of C in diamonds: A review Applied Geochemistry 6:477–494.
- Kirkley, Gurney, and Levinson, Ref. 196.
- Ozima, M. and S. Zashu, 1983. Primitive helium in diamonds. Science 219:1067–1068.
- Gurney, Ref. 197.
- Harte, B. and C. J. Hawkesworth, 1986. Mantle domains and mantle xenoliths. Kimberlites and related rocks, Vol. 2: Their mantle/crust setting, diamonds and diamond exploration. Proceedings of the fourth international kimberlite conference, Geological Society of Australia special publication no. 14, pp. 649–686.
- Gurney, Ref. 198.
- Zashu, S., M. Ozima, and O. Nitoh, 1986. K-Ar isochron dating of Zaire cubic diamonds. Nature 323:710–712.
- Podosek, F. A., J. Pier, O. Nitoh, S. Zashu, and M. Ozima, 1988. Normal potassium, inherited argon in Zaire cubic diamonds. Nature 334:607–609.
- Phillips, D., T. C. Onstott, and J. W. Harris, 1989. 40Ar/39Ar laser-probe dating of diamond inclusions from the Premier Kimberlite. Nature 340:460–462.
- Kirkley, Gurney, and Levinson, Ref. 196.
- Zashu, Ozima, and Nitoh, Ref. 211.
- Navon, O., I. D. Hulcheon, G. R. Rossman, and G. J. Wasserburg, 1988. Mantle-derived fluids in diamond micro-inclusions. Nature 335:784–789.
- Ozima, M., S. Zashu, Y. Takigami, and G. Turner, 1989. Origin of the anomalous 40Ar-39Ar age of Zaire cubic diamonds: Excess 40Ar in pristine mantle fluids. Nature 337:226–229.
- Turner, G., R. Burgess, and M. Bannon, 1990. Volatile-rich mantle fluids inferred from inclusions in diamond and mantle xenoliths. Nature 344:653–655.
- Patterson, D. B., M. Honda, and I. McDougall, 1992. The noble gas cycle through subduction systems. Research School of Earth Sciences, annual report 1992 , pp. 104–106. Canberra: Australian National University.
- Staudacher, T., 1987. Upper mantle origin of Harding County well gases. Nature 325:605–607.
- Staudacher, et al., Ref. 147.
- Ballentine, C. J., 1997. Resolving the mantle He/Ne and crustal 21Ne/22Ne in well gases. Earth and Planetary Science Letters 152:233–249.
- Burnard, Graham, and Turner, Ref. 191.
- Moreira, Kunz, and Allègre, Ref. 189.
- Staudacher, et al., Ref. 147.
- Sobolev, N. V. and V. S. Shatsky, 1990. Diamond inclusions in garnets from metamorphic rocks: A new environment for diamond formation. Nature 343:742–746.
- Dobrzhinetskaya, L. F., E. A. Eide, R. B. Larsen, B. A. Sturt, R. G. Trønnes, D. C. Smith, W. R. Taylor, and T. V. Posukhova, 1995. Microdiamond in high-grade metamorphic rocks in the Western Gneiss Region, Norway. Geology 23:597–600.
- Hough, R. M., I. Gilmour, C. T. Pillinger, F. Langenhorst, and A. Montanari, 1997. Diamonds from the iridium-rich K-T boundary layer at Arroyo El Mimbral, Tamaulipas, Mexico. Geology 25:1019–1022.
- Ozima, M., S. Zashu, K. Tomura, and Y. Matsuhisa, 1991. Constraints from noble-gas contents on the origin of Carbonado diamonds. Nature 351:472–474.
- Dalrymple, Ref. 120.
- Funkhouser, Barnes and Naughton, Ref. 139.
- Laughlin, A. W. 1969. Excess radiogenic argon in pegmatite minerals. Journal of Geophysical Research 74:6684–6690.
- Lanphere, M. A. and G. B. Dalrymple, 1976. Identification of excess 40Ar by the 40Ar/39Ar age spectrum technique. Earth and Planetary Science Letters 12:359–372.
- Pickles, C. S., S. P. Kelley, S. M. Reddy, and J. Wheeler, 1997. Determination of high spatial resolution argon isotope variations in metamorphic biotites. Geochimica et Cosmochimica Acta 61:3809–3833.
- Harrison, T. M. and I. McDougall, 1980. Investigations of an intrusive contact, northwest Nelson, New Zealand II. Diffusion of radiogenic and excess 40Ar in hornblende revealed by 40Ar/39Ar age spectrum analysis. Geochimica et Cosmochimica Acta 44:2005–2020.
- Webb, A. W., 1985. Geochronology of the Musgrave Block. Mineral Resources Review, South Australia 155:23–27.
- Baksi, A. K. and A. F. Wilson, 1980. An attempt at argon dating of two granulite-facies terranes. Chemical Geology 30:109–120.
- Harrison, T. M. and I. McDougall, 1981. Excess 40Ar in metamorphic rocks from Broken Hill, New South Wales: Implications for 40Ar/39Ar age spectra and the thermal history of the region. Earth and Planetary Science Letters 55:123–149.
- Harrison and McDougall, Ref. 238.
- Harte and Hawkesworth, Ref. 209.
- Porcelli, D. and G. J. Wasserburg, 1995. Transfer of helium, neon, argon, and xenon through a steady-state upper mantle. Geochimica et Cosmochimica Acta 59:4921–4937.
- Porcelli and Wasserburg, Ref. 241, p. 4924.
- Matsumoto, et al., Ref. 193.
- Harrison and McDougall, Ref. 235.
- Laughlin, Ref. 232.
- Baksi and Wilson, Ref. 237.
- Harrison and McDougall, Ref. 238.
- Webb, Ref. 236.
- Staudacher, Ref. 220.
- Cole and Lewis, Ref. 5.
- Williams, Ref. 2.
- Harte and Hawkesworth, Ref. 209.
- Graham and Hackett, Ref. 1.
- Stipp, Ref. 18.
- Ewart and Stipp, Ref. 80.
- Ewart and Stipp, Ref. 80.
- Cole, Ref. 91.
- Cole, Cashman and Rankin, Ref. 103.
- Graham and Hackett, Ref. 1.
- Graham and Hackett, Ref. 1.
- Graham and Hackett, Ref. 1.
- Graham, et al., Ref. 101.
- Austin, S. A., J. R. Baumgardner, D. R. Humphreys, A. A. Snelling, L. Vardiman, and K. P. Wise, 1994. Catastrophic plate tectonics: A global Flood model of earth history. In, Walsh, R. E. (ed.), Proceedings of the third international conference on creationism, pp. 609–621. Pittsburgh, Pennsylvania: Creation Science Fellowship.
- Austin, et al., Ref. 263.

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis