
Carbon: God’s Building Blocks for Life
Formed to Be Inhabited
“The Lord, who created the heavens (He is the God who formed the earth and made it, He established it and did not create it a waste place, but formed it to be inhabited). Isaiah 45:18 (NASB)
Earth—Formed to Be Inhabited
- God’s Flawless Design of Water Makes Life Possible
- Carbon: God’s Building Blocks for Life
- Proportionally Perfect for Life: O2, CO2, N2
- God’s Gift of Metals: Crafted for Life
At-a-Glance
Only the element carbon is capable of bonding with itself to form stable, long chains, as well as branched and cyclic compounds, permitting the construction of myriad structures with different chemical and physical properties suitable for life.
Carbon is able to simultaneously bond with four very different elements thus increasing the number of reactions in which it can engage and the number of structures it can be used to construct.
Only carbon-based compounds, when oxidized by oxygen, can yield sufficient energy to meet the metabolic demands of warm-blooded animals.
Isaiah clearly states that God formed the earth to be inhabited. This would imply that every aspect of the environment has been designed for the growth and maintenance of life. This includes everything from the air we breathe, the water we drink, to even the various types of atoms and molecules from which life is made. Over the past century and a half, man has made great technological advances that have increased our knowledge and understanding of chemistry and the environment in which we live. With this increase, many have become acutely aware of just how uniquely suited the environment is for life.
Everyone is familiar with LEGO®, and that a single piece can be used to build a multitude of different structures for different purposes. Well, God has just such a piece that he uses to build the myriad different life forms. It is called carbon. Man, with the mind God has given him, also uses this piece to make many different products for different purposes. Whereas carbon may be viewed as a LEGO® piece, carbon-based compounds are the whole LEGO® set as the same pieces are used to construct the multitude of life forms from plants to insects to humans each with very different structures and purposes.

Carbon: God’s LEGO® Piece
In this article, I will recapitulate some of the many aspects of the element carbon that have clearly been shown to be perfectly matched for life. More in-depth coverage can be found in the references at the end.
The study of carbon and all its derivatives is known as organic chemistry. A subdiscipline of organic chemistry is biochemistry, which is the study of the organic chemistry of living things. Compounds that have carbon as a constituent are collectively called organic.
Carbon compounds are central to life on earth. They form the building blocks of proteins that are structural elements such as bone, contractile elements such as muscle, coverings such as skin, and enzymes that catalyze the multitude of biochemical reactions. The hereditary molecules of life, the nucleic acids DNA and RNA, are composed of carbon compounds. Together with oxygen, carbon compounds provide the energy for life.
Not all organic compounds, however, are found only in living organisms. Our wise Creator made carbon extremely versatile and gave man a mind to study carbon in order to create tens of thousands of carbon-based compounds for a wide variety of uses such as clothing, shelter, transportation, and medicines.
Carbon is the primary element of all molecules found in living organisms.
Carbon is the primary element of all molecules found in living organisms. Carbon atoms are not unique in being able to form four strong bonds with four other atoms. Silicon can also do the same. Both elements usually combine with hydrogen, oxygen, or nitrogen. Many popular sci-fi novels imagine silicon-based life forms on other planets; however, such life forms are not known on earth for good reason. Most silicon-based compounds do not have “handedness” as carbon-based compounds do. For example, amino acids that make up proteins are left-handed, and sugars found in cells are typically right-handed. This fact is extremely important as almost all organic compounds have a handedness to them. Handedness provides for proper molecular recognition between reacting molecules such as enzymes and their substrates. It allows cells to exert a high degree of control over the many metabolic reactions. Though many silicon-based analogs of carbon compounds can be made in the lab, there are no silicon analogs for many of the carbon compounds essential for life.
When carbon–based compounds are oxidized for energy, they form CO2 and water. CO2 is a gas that can be easily eliminated from the body whereas silicon would form silicon dioxide, SiO2, which is a solid. Consequently, SiO2 could not be as easily eliminated as CO2. As we’ll see in the next article in this series, CO2 can also be used to help regulate pH in the body, whereas SiO2 cannot. The regulation of pH is critical to life because most of the reactions in the cell depend upon a very narrow range of pH values.
Two or more carbon compounds can have the same atoms and the same number of each, called isomers, yet be structurally very different. (FIGURE 1). Each different arrangement of atoms represents a different compound with different chemical and physical properties.
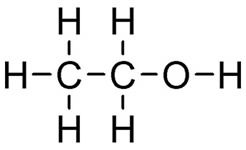
Ethanol. Image by Calvero, via Wikimedia Commons.
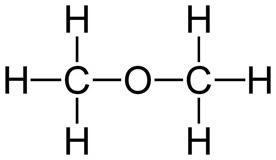
Dimethyl ether. Image by Andrew pmk~commonswiki, via Wikimedia Commons.
Figure 1. Shown are two isomers with identical elemental composition, C2H6O, ethanol and dimethyl ether. However, their structures are completely different giving rise to different chemical and physical properties.
The arrangement of covalent bonds about the carbon atom is tetrahedral; the four atoms can be oriented in four different directions. (FIGURE 2) Carbon is unique among elements in that it can form these bonds with other carbon atoms to form long chains of carbons or form cyclic and branched structures. No other element has this capability. Thus, the number of different organic compounds of carbon is in the millions. Indeed, about 95% of all known compounds are carbon-based. Indeed, about 95% of all known compounds are carbon-based.
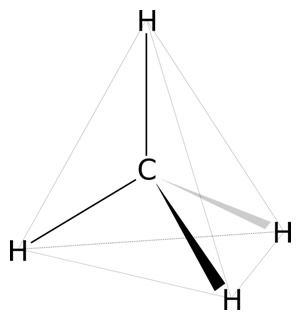
Figure 2. Shows the tetrahedral arrangement of substituents about the central carbon atom. Shown is methane, the simplest hydrocarbon. Image by Kaprak, via Wikimedia Commons.
Nitrogen, which lies next to carbon on the periodic table, does not have this ability; even short chains of nitrogen are very unstable and can be explosive. The element boron, which lies on the other side of carbon in the table, also cannot form continuous chains. Metals cannot be used to make the molecules of life since they are typically found as ions, and do not form strong, directional bonds with other metal atoms. Silicon, which sits just below carbon on the table, has some of the same properties as carbon. One of those is the ability to form chains; however, these are not nearly as stable as those of carbon. Even with some of the same properties, silicon cannot form the multitude of compounds seen with carbon. Thus silicon is less than ideal for life. Carbon compounds remain intact when dissolved in water, while metal compounds dissociate into ions. This feature is of vital importance to life. Thus carbon is a uniquely fit, low-mass element that can form four strong covalent bonds, and able to undergo numerous reactions within the temperature and pH range in which most of life is found.
All life forms require a multitude of different organic compounds which can be manipulated and transformed into each other.
Life requires more than just carbon. It also requires compounds that contain the elements oxygen, hydrogen, and nitrogen as well as many other trace elements. All life forms require a multitude of different organic compounds which can be manipulated and transformed into each other. Organic compounds composed only of carbon and hydrogen are known collectively as hydrocarbons. (FIGURE 3) Hydrocarbons are relatively stable, but may be made more reactive by the substitution of the hydrogens with various reactive groups, such as hydroxyl (–OH), carboxyl (-COOH), and amino (–NH2) groups. Only carbon is able to bind more than one reactive group at a time. (FIGURE 4) Substitution with these groups greatly increases the number of different compounds, increases the complexity of compounds, and increases the number and types of reactions carbon compounds can engage in. These groups also permit molecules to interact weakly with each other and within themselves. For example, it is essential to life that proteins and other biomolecules be held in relatively precise 3D conformations. This is accomplished through many weak interactions between various amino acid side-chains, the building blocks of proteins. Again, this is only permissible with substituted carbon compounds. These same weak interactions also permit enzymes to bind their substrate(s) and catalyze particular reactions, like a lock and key.
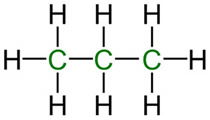
Propane. Image by NEUROtiker, via Wikimedia Commons.
Pentane. Image by Wereldburger758, via Wikimedia Commons.
Aromatic hydrocarbons. Image by Milton Beychok, via Citizendium.
Figure 3. Some common representative hydrocarbons showing both linear and cyclic compounds.
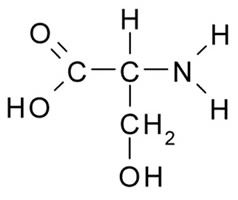
Figure 4. Shows the amino acid serine. Note the central carbon atom has a carboxyl group (-COOH), an amino group (-NH2), a H, and a –CH2OH group attached to it. The amino, the hydroxyl (-OH), and the carboxyl groups can each engage in different types of reactions. Image by Borb, via Wikimedia Commons.
Carbon, unlike other elements, can form multiple bonds with itself and other elements. This ability is crucial for those compounds involved with the absorption of light, such as retinal in the retina of the eye. Multiple bonds also contribute to the rigidity of large molecules allowing the construction of structurally stable molecules such as proteins. Metals cannot form multiple bonds with the ease that carbon can. Chains containing carbons with double bonds can be joined together, or conjugated, to form compounds that are especially designed to absorb light at various wavelengths, giving rise to the multitude of colors we see around us. (FIGURE 5) Many of these same carbon chains act as antennae molecules and pass electrons energized by the sun on to chlorophyll so the process of photosynthesis can take place.
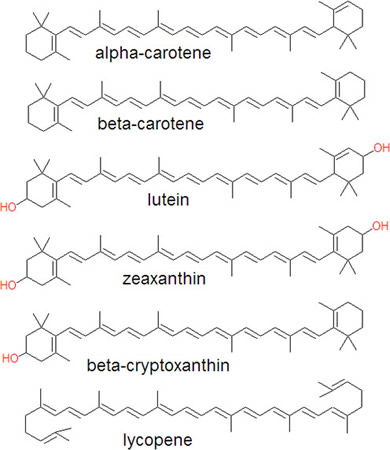
Figure 5. A variety of carotenoids showing chains of conjugated double bonds. These types of carbon-based molecules are especially designed to absorb light at various wavelengths. Image by David E. Volk, via Citizendium.
Energy Considerations
Organic compounds have mild chemical properties which are relatively stable under ambient conditions, but can be activated under relatively mild conditions to undergo reactions with other organic compounds. This property is essential for life to be able to extract energy from carbon-based molecules slowly and in a controlled manner.
The ultimate source of energy for our planet for most organisms comes from the sun.
As we all know, energy is essential for all life. The source of energy, however, varies for different organisms. With the exception of lithotrophs, such as sulfur bacteria and other bacterial forms which derive energy from inorganic compounds, the ultimate source of energy for our planet for most organisms comes from the sun. Only plants are capable of capturing and harnessing this energy directly. Most animals, instead, must obtain their energy through the consumption of plants and/or other animals. When we eat, we are taking in carbon compounds that are fairly highly reduced, and we later oxidize them to CO2 and water through a series of enzyme-controlled reactions collectively known as catabolism. In the process, we generate energy and obtain the necessary building blocks needed for the maintenance and growth of our own bodies. During catabolism we are extracting hydrogens (actually electrons) from the reduced carbon atoms and using the electrons to generate chemical energy in the form of ATP (adenosine triphosphate).
To meet the needs of the many cellular processes that make life possible, it is essential for life to be able to extract energy from carbon-based molecules. A simple, single-carbon molecule is methane, CH4; the carbon atom has a full complement of hydrogen atoms. In this state, the carbon is fully reduced (see FIGURE 2). The more reduced a carbon compound is, the greater the amount of energy that can be obtained by oxidizing it to CO2 (the most oxidized form of carbon) and water. Because only carbon has the ability to exist in chains, it makes the ideal element with which to build molecules that can store large amounts of potential energy. Indeed, carbon chains and cyclic carbon compounds with full, or nearly full, complements of hydrogen are widely used in industry as fuel molecules. In the body, the most reduced form of carbon is found in the form of fatty acids. Fatty acids (FIGURE 6) are chains of carbons that are saturated, or nearly so, with hydrogens, making them ideal for the storage of large amounts of potential energy without the concomitant storage of water as with carbohydrates such as glycogen. The body uses fatty acids stored in fat molecules as a major fuel source. In addition to serving as energy reservoirs, fats (FIGURE 7), composed of fatty acids, provide for thermal insulation, protection against potentially damaging collisions, and they give shape to the body.
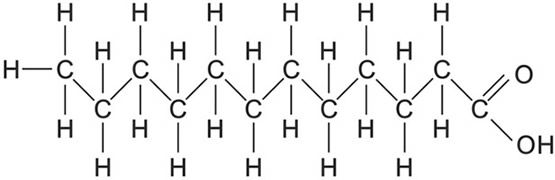
Figure 6. A saturated fatty acid showing each carbon atom with a full complement of hydrogens. Fatty acids have a carboxyl group (COOH) at one end (right). This is a reactive group used to combine fatty acids with other molecules such as glycerol. Derived from image by BruceBlaus, via Wikimedia Commons.
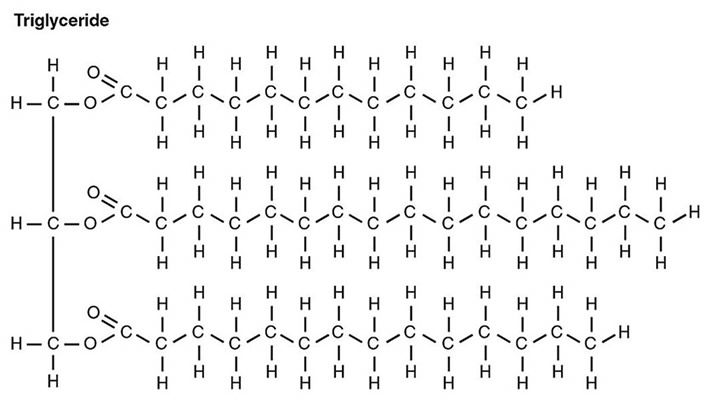
Figure 7. A neutral fat (so called because there is no charge) composed of three fatty acids attached to a glycerol backbone (left side of image). The fatty acids (horizontal chains) may be saturated with hydrogen (as shown) or unsaturated. Derived from image by CFCF, via Wikimedia Commons.
Fatty acids that are >14 carbons long are also quite hydrophobic. This property makes them ideal molecules with which to make all biological membranes. Membrane lipids, or phospholipids, (FIGURE 8), are also composed of fatty acids, and can serve as electrical insulation for neurons. Indeed, membranes also form the vital compartments of every cell. Compartments permit the regulation of various biochemical pathways. Compartmental membranes are also quite selective in what they allow into, or out of, a particular compartment. The plasma membrane also forms a selective barrier between the cell and its environment; only select ions and molecules can enter and leave the cell.
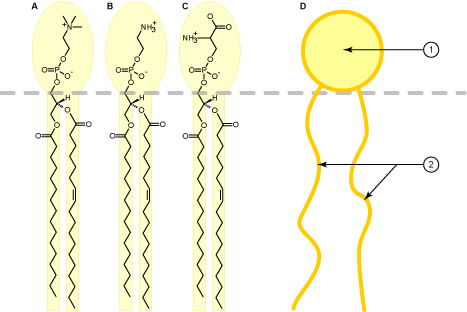
Figure 8. Typical membrane phospholipids (so called because they contain a phosphate group attached to one of the carbons in the glycerol backbone), they also contain two fatty acids, and one of several head groups. These may also differ in their fatty acid composition as well as the head group. Figure A is PC = phosphatidylcholine, B is PE = phosphatidylethanolamine, C is PS = phosphatidylserine, and D is the short-hand representation of membrane phospholipids. Image by Foobar, via Wikimedia Commons.
Conclusion
As one studies living systems and the molecules from which they are made, one sees the wisdom of the Creator who designs molecules with properties that can be used for more than one purpose. Carbon has been uniquely designed to carry out the function of being the element upon which all molecules of life are built. It truly is God’s LEGO® piece.
In the next article, we’ll take a look at the gases in our atmosphere that are so vital for life—CO2, O2, and N2. Again, we’ll see carbon in its fully oxidized form fulfilling a unique role in the maintenance of life.
Acknowledgements
I wish to thank Ms. Sabrina Hebron and Mr. Michael Kornoff for their many helpful suggestions.
References
Baggaley, Kate. “Looking for Silicon-Based Alien Life? Don’t Hold Your Breath.” Popular Science. (December 6, 2016), https://www.popsci.com/bacteria-have-bonded-carbon-and-silicon-for-first-time-what-can-they-teach-us.
Dessy, Raymond, “Could Silicon Be the Basis for Alien Life Forms, Just as Carbon Is on Earth?,” Scientific American, https://www.scientificamerican.com/article/could-silicon-be-the-basi/, accessed January 12, 2018.
Denton, Michael. Nature’s Destiny. New York, NY: The Free Press, 1998.
Dickerson, R. E. and I. Geis. Chemistry, Matter, and the Universe. Menlo Park, CA: W. A. Benjamin, 1976.
Henderson, Lawrence J. The Fitness of the Environment. New York, NY: The MacMillan Company, 1913.
Morrison, R. T., and R. N. Boyd. Organic Chemistry, 3rd edition. Boston, MA: Allyn and Bacon, 1973.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis


