A Unique Blend
Special Feature: There’s No Place Like Home
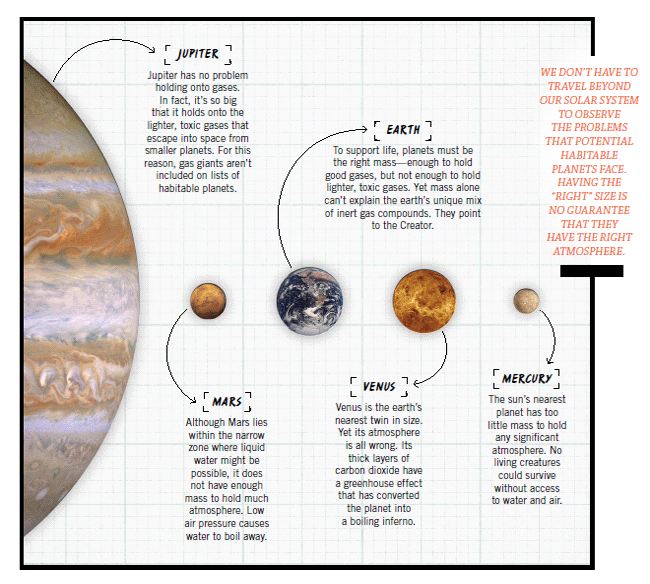
As long as a planet is the right size, it could retain an atmosphere with all the right ingredients needed for life. Just one problem. Only one known planet does. Not just the right elements are needed but the right combination of elements. Earth is unique.
In a ground test of the Apollo 1 spacecraft in 1967, three astronauts perished in a horrible fire that broke out and rapidly spread through the capsule. This disaster required major changes in the design of future Apollo capsules, including reducing the oxygen content from 100% to the 21% present in our atmosphere.
You might think that a pure oxygen atmosphere would be better than just 21% since that is the only gas our bodies utilize when we breathe. However, a pure or nearly pure oxygen atmosphere would be disastrous. Not only would fires quickly rage out of control but experiments have shown that the cells of living organisms would be damaged if exposed to excessive oxygen for a long time.
Bulking up with Nitrogen
We tend to take for granted the healthfulness of the air around us as long as we get the oxygen we need. We don’t pay much attention to the other elements. Yet the bulk of the atmosphere consists of nitrogen (78%), and the remaining 1% is a mixture of trace materials. What is all that nitrogen doing in the atmosphere?
Nitrogen is in a diatomic form, meaning that each molecule of nitrogen is made of two tightly bound atoms of nitrogen. We write this chemically as N2. Diatomic nitrogen is chemically inert, which means that it rarely interacts with anything. So the majority of the earth’s atmosphere doesn’t do anything except provide bulk and take up space. This is ideal, because it provides the proper amount of air pressure to support life without interfering with basic life processes. For example, just the simple exchange of gases in breathing requires a certain amount of air pressure. If all that inert nitrogen weren’t present, breathing would be difficult.
Avoiding the Greenhouse Effect
The oxygen is diatomic as well (O2). Together, more than 99% of the earth’s atmosphere is diatomic, and that’s also a good thing! Other types of gases contribute to the greenhouse effect. Too much of them would turn our planet into an unbearable hothouse.
No other known planets have atmospheres that are diatomic. The most common gases on the other planets are polyatomic (molecules containing three or more atoms per molecule). Examples of polyatomic gases are carbon dioxide (CO2), water vapor (H2O), ozone (O3), and methane (CH4). We call these “greenhouse gases” because they have the same effect as windowpanes in a greenhouse.
As sunlight passes through the panes, the interior heats up, but the glass prevents the heat from escaping. Polyatomic gases have the same effect on a planet. Sunlight heats up the surface, but polyatomic gases trap the heat in the lower atmosphere so that it can’t escape. This explains why the surface of Venus is very hot; its thick atmosphere consists almost entirely of CO2.
photos: NASA
Avoiding Radical Temperature Swings
The earth’s atmosphere is very different from any other planet’s. The small amount of polyatomic gas produces just enough greenhouse effect to keep the temperatures from getting too cold at night, as heat escapes. It also blocks much of the infrared radiation coming from the sun, preventing the earth from heating up too quickly during the day. If the earth didn’t have any polyatomic gases, then the temperature would fluctuate well over 100° F (55.5° C) every day.
Scientists understand this danger very well, so they have begun looking for extrasolar planets that might have diatomic atmospheres. They know that no other atmosphere will work. No luck, so far. It seems we live on a unique planet, specially created to be inhabited.
Special Feature: There’s No Place Like Home
- Just Right for Life
- Not Just Another Star
- A Perfect Partner
- A Unique Blend
Related Videos
Our Created Solar System Excerpt
Answers Magazine
January – March 2014
Placed safely in our solar system’s “goldilocks zone” and engineered with the perfect balance of atmosphere, chemicals, and water, our earth was miraculously formed to be inhabited (Isaiah 45:18). This issue examines the earth’s unique suitability for life. We’ll also investigate what seminaries are actually teaching our pastors, the possibility that viruses could be beneficial, and more.
Browse IssueRecommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis