
Chapter 12
Radioactive Dating of Rocks: Questions Answered
Most people today think that geologists have proven the earth and its rocks to be billions of years old by their use of the radioactive dating methods. Ages of many millions of years for rocks and fossils are glibly presented as fact in many textbooks, the popular media, and museums.
For decades, the biologists have boldly proclaimed that, whereas we cannot observe today one type of creature evolving into a totally different type of creature, “Time is the hero of the plot. . . . Given so much time, the ‘impossible’ becomes possible, the possible probable, and the probable virtually certain. One has only to wait: time itself performs the miracles.”1
Yet few people seem to know how these radiometric dating methods work. No one even bothers to ask what assumptions drive the conclusions. So let’s take a closer look at these methods and see how reliable they really are.
Atoms—Basics We Observe Today
Each chemical element, such as carbon and oxygen, consists of atoms unique to it. Each atom is understood to be made up of three basic parts. The nucleus contains protons (tiny particles each with a single positive electric charge) and neutrons (particles without any electric charge). Orbiting around the nucleus are electrons (tiny particles each with a single electric charge).
The atoms in each chemical element may vary slightly in the numbers of neutrons within their nuclei. These slightly different atoms of the same chemical element are called isotopes of that element. However, while the number of neutrons varies, every atom of any chemical element always has the same number of protons and electrons. So, for example, every carbon atom contains six protons and six electrons, but the number of neutrons in each nucleus can be six, seven, or even eight. Therefore, carbon has three isotopes, which are specified as carbon-12, carbon-13 and carbon-14 (figure 1).
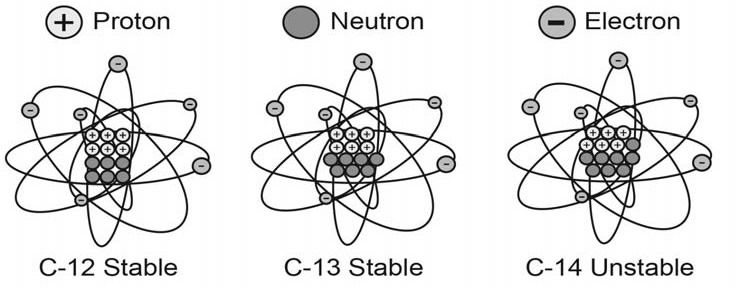
Figure 1. Comparison of stable and unstable atoms of the element carbon. They have six protons in their nuclei and six electrons orbiting their nuclei, which gives carbon its chemical properties. It is the number of neutrons in their nuclei that varies, but too many neutrons make the nuclei unstable, as in carbon-14.
Radioactive Decay
Some isotopes of some elements are radioactive; that is, they are unstable because their nuclei are too large. To achieve stability, these atoms must make adjustments, particularly in their nuclei. In some cases, the isotopes eject particles, primarily neutrons and protons. (These are the moving particles which constitute the radioactivity measured by Geiger counters and the like.) The end result is stable atoms, but of a different chemical element (not carbon) because these changes have resulted in the atoms having different numbers of protons and electrons.
This process of changing the isotope of one element (designated as the parent) into the isotope of another element (referred to as the daughter) is called radioactive decay. Thus, the parent isotopes that decay are called radioisotopes. Actually, it isn’t really a decay process in the normal sense of the word, like the decay of fruit. The daughter atoms are not lesser in quality than the parent atoms from which they were produced. Both are complete atoms in every sense of the word. Rather, it is a transmutation process of changing one element into another.
Geologists regularly use five parent isotopes as the basis for the radioactive methods to date rocks: uranium-238, uranium-235, potassium-40, rubidium-87, and samarium-147. These parent radioisotopes change into daughter lead-206, lead-207, argon-40, strontium-87, and neodymium-143 isotopes, respectively. Thus, geologists refer to uranium-lead (two versions), potassium-argon, rubidium-strontium, or samarium-neodymium dates for rocks. Note that the carbon-14 (or radiocarbon) method is not used to date rocks, because most rocks do not contain carbon.
Unlike radiocarbon (14C), the other radioactive elements used to date rocks—uranium (238U), potassium (40K), rubidium (87Rb), and samarium (147Sm)—are not being formed today within the earth, as far as we know. Thus it appears that God probably created those elements when He made the original earth.
Chemical Analyses of Rocks Today
Geologists must first choose a suitable rock unit for dating. They must find rocks that contain these parent radioisotopes, even if they are only present in minute amounts. Most often, this is a rock body, or unit, which has formed from the cooling of molten rock material (called magma). Examples are granites (formed by cooling under the ground) and basalts (formed by cooling of lava flows at the earth’s surface).
The next step is to measure the amounts of the parent and daughter isotopes in a sample of the rock unit. This is done by chemical analyses in specially equipped laboratories with sophisticated instruments capable of very good accuracy and precision. So, in general, few people quarrel with the resulting chemical analyses.
However, it is the interpretation of these chemical analyses of the parent and daughter isotopes that raises potential problems with these radioactive dating methods. To understand how geologists “read” the age of a rock from these chemical analyses using the radioactive “clock,” let’s use the analogy of an hourglass “clock” (figure 2).

Figure 2. See chapter 11, “Carbon Dating: Questions Answered”
In an hourglass, grains of fine sand fall at a steady rate from the top glass bowl to the bottom. At time zero, the hourglass is turned upside-down so that all the sand starts in the top bowl. After one hour, all the sand has fallen into the bottom glass bowl. So, after only half an hour, half the sand should be in the top bowl and the other half should be in the bottom glass bowl.
Suppose now that a person, who did not observe when the hourglass was turned upside-down (i.e., time zero), wants to “read” this “clock.” He walks into the room and notices that half the sand is in the top bowl, and half the sand is in the bottom bowl. Because he knows the rate at which the sand falls (a full bowl of sand all falls in one hour), and assuming all the sand started in the top bowl, he is able to calculate that the “clock” evidently started half an hour ago.
Reading the Radioactive “Clock”
The application of this analogy to reading the radioactive “clock” should be readily apparent. The sand grains in the top glass bowl (figure 2) represent atoms of the parent radioisotope (uranium-238, potassium-40, etc.). The falling of the sand grains equates to radioactive decay, while the sand grains at the bottom represent the daughter isotope (lead-206, argon-40, etc.).
When a geologist today collects a rock sample to be dated, he has it analyzed for the parent and daughter isotopes it contains—for example, potassium-40 and argon-40. He then assumes all the daughter argon-40 atoms have been produced by radioactive decay of parent potassium-40 atoms in the rock since the rock formed. So if he knows the rate at which potassium-40 decays radioactively to argon-40 (i.e., the rate at which the sand grains fall), then he can calculate how long it has taken for the argon-40 (measured in the rock today) to form. Since the rock supposedly started with no argon-40 in it when it formed, then this calculated time span back to no argon-40 must be the date when the rock formed (i.e., the rock’s age).
The radioactive methods for dating rocks are thus simple to understand. If one knows the rate of radioactive decay of a parent radioisotope in a rock (the sand falling rate in the analogous hourglass “clock” of figure 2), and how much daughter isotope is in the rock today (the quantity of sand at the bottom), then the age of the rock is the time it has taken for the daughter isotope to accumulate in the rock by radioactive decay of the parent radioisotope since the rock formed (since the hourglass “clock” was turned upside-down).
But what if the assumptions are wrong? For example, what if radioactive material was added to the rock (to the top bowl) or if the decay rates have changed since the rock formed?
The Necessary Assumptions
It is not readily apparent from the analogy with an hourglass that the reliability of the radioactive “clocks” is subject to three unprovable assumptions. After all, the reliability of an hourglass can be tested, for example, by turning the hourglass upside-down to start the clock, and by then watching the sand grains fall and timing it with a trustworthy clock. In contrast, no geologist was present when the rock unit to be dated was formed, to see and measure its initial contents. Nor has any geologist been present to measure how fast the radioactive “clocks” have been running in that rock unit through the millions of years that supposedly passed after the rock formed. Thus, there are three critical (but unprovable) assumptions every geologist must make when he dates a rock using the radioactive “clocks”:
- The conditions at time zero when the rock formed are, or can be, known.
- The radioactive “clocks” in the rock have to be closed to any disturbances or outside interferences (from weathering or ground waters, for instance); that is, all the atoms of the daughter isotopes must have been derived by radioactive decay of atoms of the parent radioisotopes.
- The radioactive decay rates of the parent radioisotopes must have remained constant through all the supposed millions of years since the rock formed, at the same slow rates we have measured today.
So let’s now examine these in more detail.
Assumption 1: Conditions at Time Zero
No geologists were present when most rocks formed, so they cannot test whether the original rocks already contained daughter isotopes alongside their parent radioisotopes. In the case of argon-40, for example, it is simply assumed that none was in the rocks, such as volcanic lavas, when they erupted, flowed, and cooled. For the other radioactive “clocks,” it is assumed that by analyzing multiple samples of a rock body, or unit, today it is possible to determine how much of the daughter isotopes (lead, strontium, and neodymium) were present when the rock formed (via the so-called isochron technique, which is still based on unproven assumptions 2 and 3).
Yet many lava flows that have occurred in the present have been tested soon after they erupted, and they invariably contained much more argon-40 than expected.2 For example, when a sample of the lava in the Mount St. Helens crater (that had been observed to form and cool in 1986) was analyzed in 1996, it contained so much argon-40 that it had a calculated “age” of 350,000 years!3 Similarly, lava flows on the sides of Mt. Ngauruhoe, New Zealand, known to be less than 50 years old, yielded “ages” of up to 3.5 million years.4 So it is logical to conclude that if recent lava flows of known age yield incorrect old potassium-argon ages due to the extra argon-40 that they inherited from the erupting volcanoes, then ancient lava flows of unknown ages could likewise have inherited extra argon-40 and yield excessively old ages.
There are similar problems with the other radioactive “clocks.” For example, consider the dating of Grand Canyon basalts (rocks formed by lavas cooling on the earth’s surface). In the western Grand Canyon area are former volcanoes on the North Rim that erupted after the canyon itself was formed, sending lavas cascading over the walls and down into the canyon. Obviously, these eruptions took place recently, after all the layers now exposed in the walls of the canyon were deposited. These basalts yield ages of up to 1 million years based on the amounts of potassium and argon isotopes in these rocks. But when the same rocks are dated using the rubidium and strontium isotopes, an age of 1,143 million years is obtained. This is the same as the rubidium-strontium age obtained for ancient basalt layers deep below the walls of the eastern Grand Canyon.5 How could both the recent and ancient lavas—one at the top and one at the bottom of the canyon, respectively—be the same age based on the same parent and daughter isotopes? One solution is that both the recent and earlier lava flows inherited the same rubidium-strontium chemistry—not age—from their same source, deep in the earth’s upper mantle. This source already had both rubidium and strontium. To make matters even worse for the claimed reliability of these radiometric dating methods, these same young basalts that flowed from the top of the canyon yield a samarium-neodymium age of about 916 million years,6 and a uranium-lead age of about 2.6 billion years!7
Assumption 2: No Contamination by Disturbances or Interferences
The problems with contamination, as with inheritances, are already well documented in the textbooks on radioactive dating of rocks.8 Unlike the hourglass, where its two bowls are sealed, the radioactive “clocks” in rocks are open to contamination by gain or loss of parent or daughter isotopes because of waters flowing in the ground from rainfall infiltration and from molten rocks beneath volcanoes. Similarly, as molten lava rises through a conduit from deep inside the earth to be erupted through a volcano, pieces of the conduit wallrocks and their isotopes can mix into the lava and contaminate it. Because of such contamination, the less-than-50-year-old lava flows at Mt. Ngauruhoe, New Zealand, yield a rubidium-strontium “age” of 133 million years, a samarium-neodymium “age” of 197 million years, and a uranium-lead “age” of 3,908 million years!9
Assumption 3: Constant Decay Rates
Physicists have carefully measured the radioactive decay rates of parent radioisotopes in laboratories over the last 100 or so years and have found them to be essentially constant (within the measurement error margins). Furthermore, they have not been able to significantly change these decay rates by heat, pressure, or electrical and magnetic fields. So geologists have assumed these radioactive decay rates have been constant for billions of years. However, this is an enormous extrapolation of seven orders of magnitude back through immense spans of unobserved time without any concrete proof that such an extrapolation is credible. Nevertheless, geologists insist the radioactive decay rates have always been constant, because it makes these radioactive “clocks” work!
New evidence, however, has recently been discovered that can only be explained by the radioactive decay rates not having been constant in the past.10 For example, the radioactive decay of uranium in tiny crystals in a New Mexico granite yields a uranium-lead “age” of 1.5 billion years. Yet the same uranium decay also produced abundant helium, but only 6,000 years’ worth of that helium was found to have leaked out of those tiny crystals. This helium leakage is definitely more accurate as a dating method, because it is based on well-known physical laws. So this means that the uranium must have decayed very rapidly over the same 6,000 years that the helium was leaking. The rate of uranium decay must have been at least 250,000 times faster than today’s measured rate, because the decay products (lead and helium) equivalent to 1.5 billion years of slow decay have in fact accumulated in only 6,000 years!
Thus, the necessary assumptions on which the radioactive “clocks” for the dating of rocks are based are unprovable. No geologists were there to test these clocks in the past, but they have been demonstrated, even by secular geologists, to be plagued with problems. Rocks may have inherited parent and daughter isotopes from their sources, or they may have been contaminated when they moved through other rocks to their current locations. Or inflowing water may have mixed isotopes into the rocks. In addition, the radioactive decay rates have not been constant. So if these clocks are based on faulty assumptions and yield unreliable results, then scientists should not trust or promote the claimed radioactive “ages” of countless millions of years, especially since they contradict the true history of the universe as recorded in God’s Word.
So we have seen that even though the general principles of using radioisotopes to date rocks, and the chemical analyses involved, seem sound, anomalous and conflicting results are frequently obtained, as documented in the secular literature. Thus the claimed “ages” of many millions of years are totally unreliable.
Does this mean we should throw out the radioactive “clocks”? Surprisingly, they are useful! The general principles of using radioisotopes to date rocks are sound; it’s just that the assumptions have been wrong and led to exaggerated dates. While the clocks cannot yield absolute dates for rocks, they can provide relative ages that allow us to compare any two rock units and know which one formed first. They also allow us to compare rock units in different areas of the world to find which ones formed at the same time. Furthermore, if physicists examine why the same rocks yield different dates, they may discover new clues about the unusual behavior of radioactive elements during the past.
Different Dates for the Same Rocks
Usually geologists do not use all four main radioactive clocks to date a rock unit. This is considered an unnecessary waste of time and money. After all, if these clocks really do work, then they should all yield the same age for a given rock unit. Sometimes though, using different parent radioisotopes to date different samples (or minerals) from the same rock unit does yield different ages, hinting that something is amiss.11
Recent research has utilized all four common radioactive clocks to date the same samples from the same rock units.12 Among these were four rock units far down in the Grand Canyon rock sequence (figure 3), chosen because they are well known and characterized. These were as follows:
- Cardenas Basalt (lava flows deep in the east canyon sequence)
- Bass Rapids diabase sill (where basalt magma squeezed between layers and cooled)
- Brahma amphibolites (basalt lava flows deep in the canyon sequence that later metamorphosed)
- Elves Chasm Granodiorite (a granite regarded as the oldest canyon rock unit)
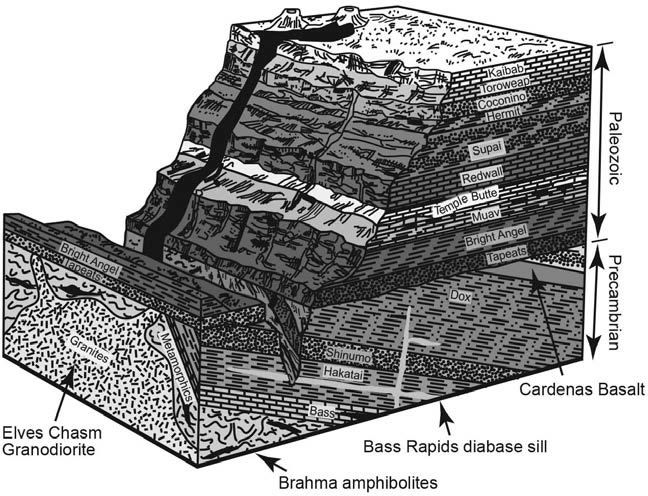
Figure 3. A geologic diagram to schematically show the rock layers exposed in the walls and inner gorge of the Grand Canyon and their relationships to one another. The deeper rocks were formed first, and the rock layers higher in the walls were deposited on top of them. The named rock units mentioned in the text are indicated.
Table 1 lists the dates obtained. Figure 4 graphically illustrates the ranges in the supposed ages of these rock units, obtained by utilizing all four radioactive clocks.
| Rock Unit | Ages (million years) | |||
|---|---|---|---|---|
| Potassium-argon | Rubidium-strontium | Uranium-lead | Samarium-neodymium | |
| Cardenas Basalt | 516 (±30) | 1111 (±81) | — | 1588 (±170) |
| Bass Rapids diabase sill | 842 (±164) | 1060 (±24) | 1250 (±130) | 1379 (±140) |
| Brahma amphibolites | — | 1240 (±84) | 1883 (±53) | 1655 (±40) |
| Elves Chasm Granodiorite | — | 1512 (±140) | 1933 (±220) | 1664 (±200) |
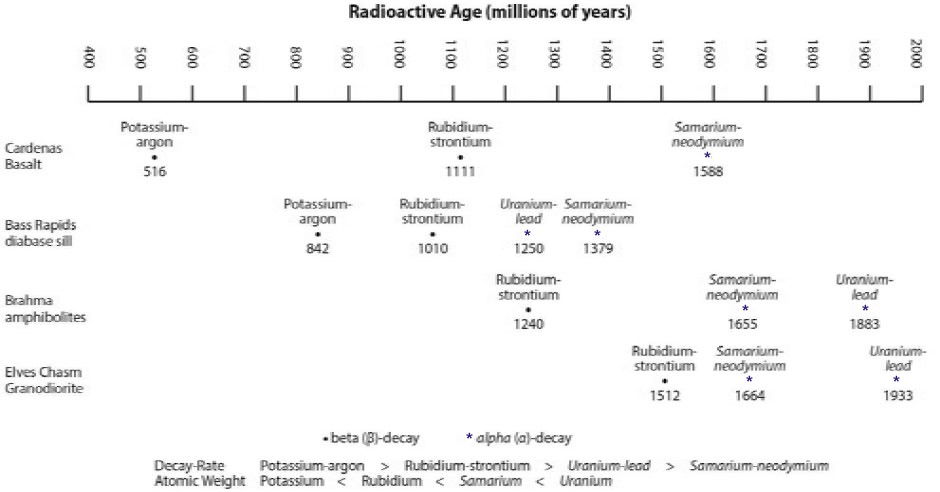
Figure 4. The comparative spread of ages for these four Grand Canyon rock units determined by the different radioactive methods on the same samples from these rock units. No two methods agree, and the ranges of ages enormous, well beyond the analytical errors inherent in all laboratory measurements. Indicated on the diagram are the two types of radioactive decay. The systematic patterns of ages obtained follow according to decay type, decay rate, and the atomic weights, suggesting an underlying physical cause for the acceleration of radioactive decay in the past.
It is immediately apparent that the ages for each rock unit do not agree. Indeed, in the Cardenas Basalt, for example, the rubidium-strontium age is more than double the potassium-argon age, and the samarium-neodymium age is three times the potassium-argon age.
Nevertheless, the ages follow three obvious patterns. Two techniques (potassium-argon and rubidium-strontium) always yield younger ages than two other techniques (uranium-lead and samarium-neodymium). Furthermore, the potassium-argon ages are always younger than the rubidium-strontium ages. And often the samarium-neodymium ages are younger than the uranium-lead ages.
What then do these patterns mean? All the radioactive clocks in each rock unit should have started “ticking” at the same time, the instant that each rock unit was formed. So how do we explain that they have each recorded different ages? The answer is simple but profound. Each of the radioactive elements must have decayed at different, faster rates in the past! In the case of the Cardenas Basalt, while the potassium-argon clock ticked through 516 million years, the rubidium-strontium clock must have ticked through 1,111 million years, and the samarium-neodymium clock through 1,588 million years. So if these clocks ticked at such different rates in the past, not only are they inaccurate, but these rocks may not be millions of years old!
But how could radioactive decay rates have been different in the past? We don’t fully understand yet. However, the observed age patterns provide clues. Potassium and rubidium decay radioactively by the process known as beta (β) decay, whereas uranium and neodymium decay via alpha (α) decay (figure 4). The former always give younger ages. We see another pattern within beta decay. Potassium today decays faster than rubidium and always gives younger ages. Both of these patterns suggest something happened in the past inside the nuclei of these parent atoms to accelerate their decay. The decay rate varied based on the stability or instability of the parent atoms. Research is continuing.
Relative Ages
Look again at figure 3, which is a geologic diagram depicting the rock layers in the walls of the Grand Canyon, along with the rock units deep in the inner gorge along the Colorado River. This diagram shows that the radiometric dating methods accurately confirm the top rock layer is younger than the layers beneath it. That’s logical, because the sediment making up that layer was deposited on top of, and therefore after, the layers below. So reading this diagram tells us basic information about the time that rock layers and rock units were formed relative to other layers.
Based on the radioactive clocks, we can conclude that these four rock units deep in the gorge (table 1) are all older in a relative sense than the horizontal sedimentary layers in the canyon walls. Conventionally the lowermost or oldest of these horizontal sedimentary layers is labeled early to middle Cambrian,13 and thus regarded as about 510 to 520 million years old.14 All the rocks below it are then labeled Precambrian and regarded as older than 542 million years. So, accordingly, all four dated rock units (table 1) are Precambrian (figure 3). And apart from the potassium argon age for the Cardenas Basalt, all the radioactive clocks have correctly shown that these four rock units were formed earlier than the Cambrian, so they are pre-Cambrian. (But the passage of time between these Precambrian rock units and the horizontal sedimentary layers above them was a maximum of about 1,650 years—the time between creation and the Flood—not millions of years.)
Similarly, in the relative sense, the Brahma amphibolites and Elves Chasm Granodiorite are older (by hours or days) than the Cardenas Basalt and Bass Rapids diabase sill (figure 3). Once again, the radioactive clocks have correctly shown that those two rock units are older than the rock units above them.
Why then should we expect the radioactive clocks to yield relative ages that follow a logical pattern? (Actually, younger sedimentary layers yield a similar general pattern15—figure 5.) The answer is again simple but profound! The radioactive clocks in the rock units at the bottom of the Grand Canyon, formed during the Creation Week, have been ticking for longer than the radioactive clocks in the younger sedimentary layers higher up in the sequence that were formed later during the Flood.
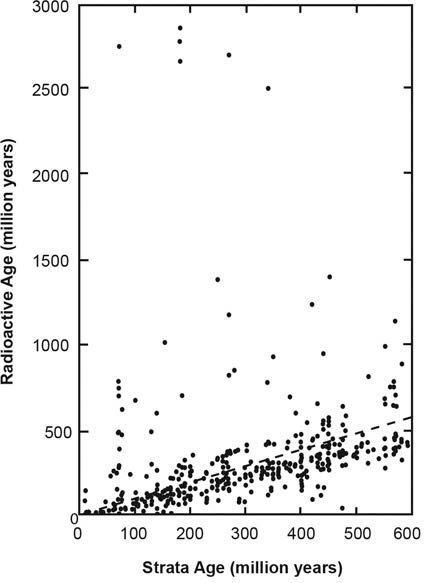
Figure 5. In general, the radioactive ages of rock layers match the ages for the strata according to their relative positions in the rock record. In this diagram the radioactive ages for rock units (vertical axis) are plotted against the strata ages determined by relative positions (horizontal axis). It can be clearly seen that often there is agreement, with the radioactive ages being in the right order according to the relative positions of the rock layers in the sequence. This is exactly what would be produced by an underlying systematic physical cause for accelerated radioactive decay.
Therefore, although it is a mistake to accept radioactive dates of millions of years, the clocks can still be useful to us, in principle, to date the relative sequence of rock formation during earth history. The different clocks have ticked at different, faster rates in the past, so the standard old ages are certainly not accurate, correct, or absolute. However, because the radioactive clocks in rocks that formed early in earth history have been ticking longer, they should generally yield older radioactive ages than rock layers formed later. So it is possible that relative radioactive ages of rocks, in addition to mineral contents and other rock features, could be used to compare and correlate similar rocks in other areas, to find which ones formed at the same time during the events detailed in Genesis, God’s eyewitness account of earth history.
How Do We Know the Bible Is True? Volume 2
Join a team of biblical scholars and Christian apologists present answers to twenty more relevant debates. Be prepared to give a reason for your faith in God!
Read OnlineOriginally published as “Radioactive Dating of Rocks?”
Footnotes
- George Wald, “The Origin of Life,” Scientific American 191, no.2 (1954): p. 48.
- Dr. Andrew A. Snelling, “Geochemical Processes in the Mantle and Crust,” in Larry Vardiman, Dr. Andrew A. Snelling, and Eugene F. Chaffin, editors, Radioisotopes and the Age of the Earth: A Young-Earth Creationist Research Initiative (El Cajon, CA: Institute for Creation Research, and St. Joseph, MO: Creation Research Society, 2000), p. 123–304.
- Steven A. Austin, “Excess Argon within Mineral Concentrates from the New Dacite Lava Dome at Mount St. Helens Volcano,” Creation Ex Nihilo Technical Journal 10, no.3 (1996): p. 335–343.
- Dr. Andrew A. Snelling, “The Cause of Anomalous Potassium-argon ‘Ages’ for Recent Andesite Flows at Mt. Ngauruhoe, New Zealand, and the Implications for Potassium-argon ‘Dating,’ ” in Robert E Walsh, editor, Proceedings of the Fourth International Conference on Creationism (Pittsburgh, PA: Creation Science Fellowship, 1998), p. 503–525.
- Dr. Andrew A. Snelling, “Isochron Discordances and the Role of Inheritances and Mixing of Radioisotopes in the Mantle and Crust,” in Larry Vardiman, Dr. Andrew A. Snelling, and Eugene F. Chaffin, editors, Radioisotopes and the Age of the Earth: Results of a Young-Earth Creationist Research Initiative (El Cajon, CA: Institute for Creation Research, and Chino Valley, AZ: Creation Research Society, 2005), p. 393–524; Don B. DeYoung, “Radioisotope Dating Case Studies,” in Thousands . . . Not Billions: Challenging an Icon of Evolution, Questioning the Age of the Earth (Green Forest, AR: Master Books, 2005), p. 123–139.
- Ibid.
- Steven A. Austin, editor, Grand Canyon: Monument to Catastrophe (Santee, CA: Institute for Creation Research, 1994), p. 123–126.
- Gunter Faure and Teresa M. Mensing, Isotopes: Principles and Applications, third edition (Hoboken, NJ: John Wiley & Sons, 2005); Alan P Dickin, Radiogenic Isotope Geology, second edition (Cambridge, UK: Cambridge University Press, 2005).
- Dr. Andrew A. Snelling, “The Relevance of Rb-Sr, Sm-Nd and Pb-Pb Isotope Systematics to Elucidation of the Genesis and History of Recent Andesite Flows at Mt. Ngauruhoe, New Zealand, and the Implications for Radioisotope Dating,” in Robert L. Ivey, Jr., editor, Proceedings of the Fifth International Conference on Creationism (Pittsburgh, PA: Creation Science Fellowship, 2003), p. 285–303; DeYoung, “Radioisotope Dating Case Studies,” p. 123–139.
- Vardiman, Snelling, and Chaffin, Radioisotopes and the Age of the Earth; DeYoung, Thousands . . . Not Billions.
- Thomas Oberthür, Donald W. Davis, Thomas G Blenkinsop, and Axel Höhndorf, “Precise U-Pb Mineral Ages, Rb-Sr and Sm-Nd Systematics of the Great Dyke, Zimbabwe—Constraints on Late Archean Events in the Zimbabwe Craton and Limpopo Belt,” Precambrian Research 113 (2002): p. 293–305; Stephen B. Mukasa, Alan H. Wilson, and Richard W. Carlson, “A Multielement Geochronologic Study of the Great Dyke, Zimbabwe: Significance of the Robust and Reset Ages,” Earth and Planetary Science Letters 164 (1998): p. 353–369; Jian-xin Zhao, and Malcolm T. McCulloch, “Sm-Nd Mineral Isochron Ages of Late Proterozoic Dyke Swarms in Australia: Evidence for Two Distinctive Events of Mafic Magmatism and Crustal Extension,” Chemical Geology 109 (1993): p. 341–354.
- Snelling, “Isochron Discordances and the Role of Inheritances and Mixing of Radioisotopes in the Mantle and Crust,” p. 393–524; DeYoung, “Radioisotope Dating Case Studies,” p. 123–139.
- Larry T. Middleton and David K. Elliott, “Tonto Group,” in Stanley S. Beus and Michael Morales, editors, Grand Canyon Geology, second edition (New York: Oxford University Press, 2003), p. 90–106.
- Felix M. Gradstein, James G. Ogg, and Alan G. Smith, editors, A Geologic Time Scale 2004 (Cambridge, UK: Cambridge University Press, 2004).
- D. Russell Humphreys, “Accelerated Nuclear Decay: A Viable Hypothesis?” in Larry Vardiman, Dr. Andrew A. Snelling, and Eugene F. Chaffin, editors, Radioisotopes and the Age of the Earth: A Young-Earth Creationist Research Initiative (El Cajon, California: Institute for Creation Research, and St. Joseph, Missouri: Creation Research Society, 2000), p. 333–379.
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- Available Monday–Friday | 9 AM–5 PM ET
- © 2026 Answers in Genesis


