Research conducted by Answers in Genesis staff scientists or sponsored by Answers in Genesis is funded solely by supporters’ donations.
Abstract
Meteorites date the earth with a 4.55 ± 0.07 Ga Pb-Pb isochron called the geochron. They appear to consistently yield 4.55–4.57 Ga radioisotope ages, adding to the uniformitarians’ confidence in the radioisotope dating methods. Among the achondrites, meteorites not containing chondrules, are the primitive achondrites, angrites, aubrites, mesosiderites, and irons. Many radioisotope dating studies in the last six decades have used the K-Ar, Ar-Ar, Rb-Sr, Sm-Nd, U-Th-Pb, Lu-Hf, Re-Os, Mn-Cr, Hf-W, Al-Mg, I-Xe, and Pu-Xe methods to yield an abundance of isochron and model ages for these achondrites from whole-rock samples, and mineral and other fractions. Such age data for 10 of these achondrites were tabulated and plotted on frequency versus age histogram diagrams. They strongly cluster in many of these achondrites at 4.55–4.57 Ga, dominated by Pb-Pb, U-Pb, and Pb-Pb calibrated isochron and model ages, testimony to the Pb-Pb technique’s supremacy as the uniformitarians’ ultimate dating tool, which they consider very reliable. These ages are sometimes confirmed by Ar-Ar, Rb-Sr, Lu-Hf, Re-Os, and Sm-Nd isochron and/or model ages, but agreement could be due to calibration with the Pb-Pb system. There is also scattering of many K-Ar, Ar-Ar, Rb-Sr, Re-Os, Sm-Nd, and a few U-Pb, Pb-Pb, Th-Pb ages, in most cases likely due to thermal disturbances resulting from impact cratering of the parent asteroids. No pattern was found in these meteorites’ isochron ages similar to the systematic patterns of isochron ages found in Precambrian rock units during the RATE project, so there is no evidence of past accelerated radioisotope decay having occurred in these achondrites, and therefore on their parent asteroids. This is not as expected, yet it is the same for all meteorites so far studied. Thus it is argued that accelerated radioisotope decay must have only occurred on the earth, and only the 500–600 million years’ worth we have physical evidence for during the Flood. Otherwise, due to their 4.55–4.57 Ga “ages” these achondrites and their parent asteroids are regarded as originally representing “primordial material” that God made on Day One of the Creation Week, from which He made the non-earth portion of the solar system on Day Four, which is compatible with the Hebrew text of Genesis. Thus today’s measured radioisotope compositions of these achondrites could reflect a geochemical signature of that “primordial material,” which included atoms of all elemental isotopes. So if most of the measured daughter isotopes were already in these achondrites when they were formed on their parent asteroids, then their 4.55–4.57 Ga “ages” obtained by Pb-Pb, U-Pb, and Pb-Pb-calibrated isochron and model age dating are likely not their true real-time ages, which according to the biblical paradigm is only about 6000 real-time years. It is anticipated that further investigation of radioisotope ages data for groups of meteorites, and martian and lunar meteorites, for lunar rocks, and for rocks from every level in the earth’s geologic record, should enable these interim ideas to be confirmed or modified.
Keywords: meteorites, classification, primitive achondrites, Acapulco, Caddo County IAB iron, angrites, Angra dos Reis, D’Orbigny, aubrites, Norton County, Shallowater, mesosiderites, Estherville, IIE irons, Colomera, Kodaïkanal, Weekeroo Station, radioisotope dating, K-Ar, Ar-Ar, Rb-Sr, Sm-Nd, U-Th-Pb, Re-Os, Lu-Hf, Mn-Cr, Hf-W, Al-Mg, I-Xe, Pu-Xe, isochron ages, model ages, discordant radioisotope ages, scattering of radioisotope ages, accelerated radioactive decay, thermal disturbance, resetting, “primordial material,” geochemical signature, mixing, inheritance, Day Three Upheaval, the Flood
Introduction
In 1956 Claire Patterson at the California Institute of Technology in Pasadena reported a Pb-Pb isochron age of 4.55±0.07Ga for three stony and two iron meteorites, which since then has been declared the age of the earth (Patterson 1956). Adding weight to that claim is the fact that many meteorites appear to consistently date to around the same “age” (Dalrymple 1991, 2004), thus bolstering the evolutionary community’s confidence that they have successfully dated the age of the earth and the solar system at around 4.56Ga. These apparent successes have also strengthened their case for the supposed reliability of the increasingly sophisticated radioisotope dating methods.
Creationists have commented little on the radioisotope dating of meteorites, apart from acknowledging the use of Patterson’s geochron to establish the age of the earth, and that many meteorites give a similar old age. Morris (2007) did focus on the Allende carbonaceous chondrite as an example of a well-studied meteorite analyzed by many radioisotope dating methods, but he only discussed the radioisotope dating results from one, older (Tatsumoto, Unruh, and Desborough 1976) paper.
In order to rectify this lack of engagement by the creationist community with the meteorite radioisotope dating data, Snelling (2014a) obtained as much radioisotope dating data as possible for the Allende CV3 carbonaceous chondrite meteorite (due to its claimed status as the most studied meteorite), displayed the data, and attempted to analyze them. He found that both isochron and model ages for the total rock, separated components, or combinations of these strongly clustered around a Pb-Pb age of 4.56–4.57Ga, the earliest (Tatsumoto, Unruh, and Desborough 1976) and the latest (Amelin et al. 2010) determined Pb-Pb isochron ages at 4.553±0.004Ga and 4.56718±0.0002Ga respectively being essentially the same. Apart from scatter of the U-Pb, Th-Pb, Rb-Sr, and Ar-Ar ages, no systematic pattern was found in the Allende isochron and model ages similar to the systematic pattern of isochron ages found in Precambrian rock units during the RATE project that was interpreted as produced by an episode of past accelerated radioisotope decay (Snelling 2005c; Vardiman, Snelling, and Chaffin 2005).
Snelling (2014b) grouped together all the radioisotope ages obtained for ten ordinary (H, L, and LL) and five enstatite (E) chondrites and similarly displayed the data. They generally clustered, strongly in the Richardton (H5), St. Marguerite (H4), Bardwell (L5), Bjurbole (L4), and St. Séverin (LL6) ordinary chondrite meteorites, at 4.55–4.57 Ga, dominated by Pb-Pb and U-Pb isochron and model ages, but confirmed by Ar-Ar, Rb-Sr, Re-Os, and Sm-Nd isochron ages. There was also scatter of the U-Pb, Th-Pb, Rb-Sr, and Ar-Ar model ages, in some cases possibly due to thermal disturbance. Again, no pattern was found in these meteorites’ isochron ages indicative of past accelerated radioisotope decay.
Snelling (2014c) subsequently compiled all the radioisotope ages for 12 eucrite (basaltic) achondrites. The data for many of these meteorites again strongly clustered at 4.55–4.57Ga, dominated by Pb-Pb and U-Pb isochron and model ages but confirmed by Rb-Sr, Lu-Hf, and Sm-Nd isochron ages. There was also scatter of the U-Pb, Pb-Pb, Th-Pb, Rb-Sr, K-Ar, and Ar-Ar model ages, in most cases likely due to thermal disturbances resulting from metamorphism or impact cratering of the parent asteroid, identified as 4-Vesta. Again, no pattern was found in these meteorites’ isochron ages similar to the systematic patterns of isochron ages found in Precambrian rock units during the RATE project, so there was no evidence of past accelerated radioisotope decay having occurred in these eucrites, and therefore on their parent asteroid.
Snelling (2014a,b,c) then sought to discuss the possible significance of this clustering in terms of various potential creationist models for the history of radioisotopes and their decay. He favored the idea that asteroids and the meteorites derived from them are residual “primordial material” from the formation of the solar system, which is compatible with the Hebrew text of Genesis that could suggest God made “primordial material” on Day One of the Creation Week, from which He made the non-earth portion of the solar system on Day Four. Thus he argued that today’s measured radioisotope compositions of all these chondrites and eucrites may reflect a geochemical signature of that “primordial material,” which included atoms of all elemental isotopes. So if some of the daughter isotopes were already in these chondrites and eucrites when they were formed, then the 4.55–4.57Ga “ages” for them obtained by Pb-Pb and U-Pb isochron and model age dating are likely not their true real-time ages, which according to the biblical paradigm is only about 6000 real-time years. However, Snelling (2014a,b,c) suggested that drawing final conclusions from the radioisotope dating data for just these 16 chondrite and 12 eucrite meteorites was still premature, and recommended further studies of more meteorites from still other classification groups. This present contribution is therefore designed to further document the radioisotope dating data for more meteorites, the primitive achondrites and the other achondrites, the latter encompassing the angrites, aubrites, mesosiderites (stony-irons), and irons, so as to continue the discussion of the potential significance of these data.
The Classification of Primitive and Other Achondrite Meteorites
The most recent classification scheme for the meteorites is that of Weisberg, McCoy, and Krot (2006), which is reproduced in Fig. 1. Based on their bulk compositions and textures, Krot et al. (2005) divided meteorites into two major categories, chondrites (meteorites containing chondrules) and achondrites (meteorites not containing chondrules or non-chondritic meteorites). They further subdivided the achondrites into primitive achondrites and igneously differentiated achondrites. However, Weisberg, McCoy, and Krot (2006) simply subdivided all meteorites into three categories—chondrites, primitive achondrites, and achondrites (fig. 1).
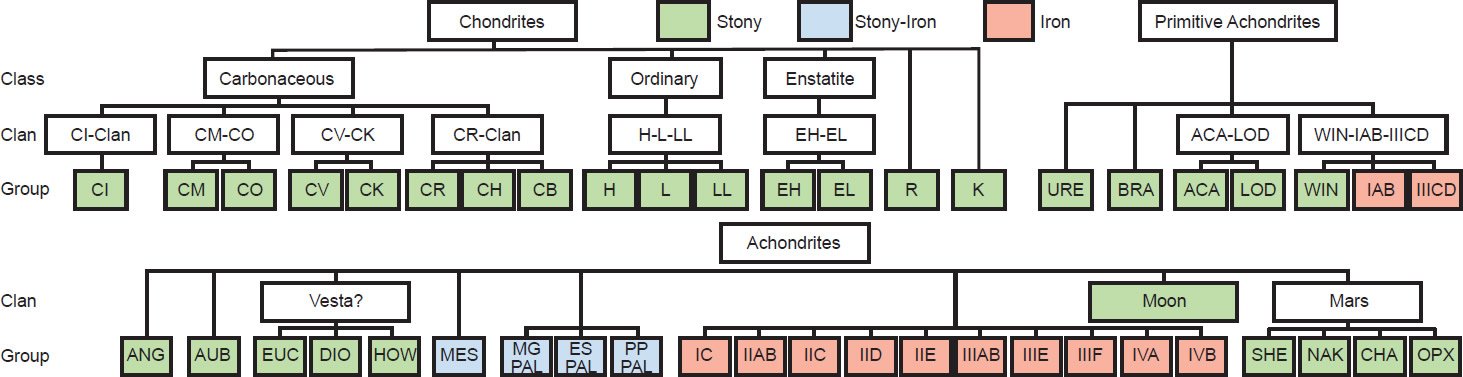
Fig. 1. The classification system for meteorites (after Weisberg, McCoy, and Krot 2006).
The non-chondritic meteorites or achondrites contain virtually none of the components found in chondrites. It is conventionally claimed that they were derived from chondritic materials by planetary melting, and that fractionation caused their bulk compositions to deviate to various degrees from chondritic materials (Krot et al. 2005). The degrees of melting that these rocks experienced are highly variable, and thus, these meteorites have been divided into the two major categories—primitive and differentiated. However, there is no clear cut boundary between these categories.
The differentiated non-chondritic meteorites, or achondrites (fig. 1), are conventionally regarded as having been derived from parent bodies that experienced large-scale partial melting, isotopic homogenization (ureilites are the only exception), and subsequent differentiation. Based on abundance of Fe-Ni metal, these meteorites are commonly divided into three types—achondrites, stony-irons, and irons. Each of these types contains several meteorite groups and ungrouped members (fig. 1). According to uniformitarians, several groups of achondrites and iron meteorites are likely to be genetically related and were possibly derived from single asteroids or planetary bodies.
The Primitive Achondrites
Several groups of stony (silicate-rich) meteorites that have essentially bulk chondritic compositions but achondritic textures apparently formed by low degrees of melting have been collectively referred to as “primitive achondrites” (Krot et al. 2005; Mittlefehldt 2005). Such partial melting would have not occurred during passage through the earth’s atmosphere, because there is only sufficient time during passage for the outer surfaces of meteorites to be heated momentarily. Thus, these have only been partially melted on their parent asteroids and therefore not completely differentiated, so they are regarded by uniformitarians as chondritic meteorites that began their conversion to achondrites but the process was aborted before completion (Norton 2002). They are therefore “intermediate forms” between the chondrites and achondrites. These include the acapulcoites and lodranites in the acapulcoites-lodranite clan, the winonaites and silicate-inclusion-bearing IAB and IIICD irons in the winonaites-IAB-IIICD silicate inclusions clan, and the brachinites and ureilites (fig. 1), although the latter two groups are sometimes included in the differentiated achondrites.
Acapulco
This type specimen for the acapulcoites fell at El Quemado near Acapulco, Mexico, in August 1976 (Norton 2002; Palme et al. 1981). A single, partially crust-covered massive “stone” weighing 1.9kg (4.2lb) was recovered from a crater 30cm (11.8in) in diameter (Palme et al. 1981; Zipfel et al. 1995). At the time it was a confusing meteorite since it has a bulk composition comparable to average H chondrites but a highly crystallized achondritic texture and unique mineral compositions intermediate between those of E and H chondrites, with some similarities to those of silicate inclusions in some iron meteorites. A second meteorite, which had fallen in 1868 a few miles east of Lodran in Punjab, Pakistan, was found to be very similar in all characteristics, so these two anomalous stony meteorites remained unique until others of their kind began to be recovered in Antarctica. Today, about 20 of these meteorites are known, most from Antarctica with a few from the Sahara, plus the Monument Draw meteorite found in Texas in 1985 (Norton 2002; Zipfel et al. 1995).
Acapulco is a fine-grained equigranular rock that consists of a tight assemblage of orthopyroxene, olivine, and plagioclase,associated with comparatively large amounts of metallic Fe-Ni alloys, and minor amounts of diopside (clinopyroxene), troilite [FeS], chromite [FeCr2O4], and the phosphates apatite and whitlockite (Palme et al. 1981). The olivine with a fayalite content of 10–12.5% (Fa10–12.5) and the orthopyroxene with a ferrosilite content of 11–13% (Fs11–13) are the two main minerals and occur in equal amounts. They are thus both Mg-rich, but their Fe/(Fe+Mg) ratio is sufficiently high (oxidized Fe) to place Acapulco’s mineralogy between the H and E chondrites. The plagioclase has an oligoclase composition, averaging An14Ab82Or4, while the clinopyroxene is emerald-green chromian diopside (En54Fs5Wo41). The phosphates occur either in irregular patches or as small grains, the latter often being associated with the sulfide or metal phases. Their modal abundance of 1.6wt% is higher than that of ordinary (O) equilibrated chondrites (0.6wt%). Also in contrast to the O chondrites whitlockite [Ca9(MgFe)(PO4)6PO3OH] constitutes only 10% of the phosphates, the remaining 90% being chlorapatite [Ca10(PO4)6(OH,Cl)2].
The high amount of the Fe-Ni metal alloys kamacite and taenite (~20 wt%) and their heterogeneous distribution in this meteorite convinced some early investigators that this was some kind of stony-iron meteorite (Norton 2002). The degree of textural equilibrium observed in Acapulco is unusually high compared with type 6 and 7 O chondrites (see figs 5 and 6 in Snelling 2014b), indicating thermal metamorphism at a high temperature and/or of longer duration, or an igneous origin (Zipfel et al. 1995). While the peak metamorphic temperature is unknown, it was sufficiently high to melt the Fe-Ni alloys and troilite. Oxygen isotopic compositions cluster between the terrestrial and carbonaceous chondrite fractionation line, showing its lack of homogeneity, so differentiation and igneous processes were apparently insufficient to homogenize it (Norton 2002).
Caddo County IAB Iron
The ~16 kg (~35 lb) Caddo County IAB iron meteorite was found in Oklahoma in 1987 (Takeda et al. 2000). Petrologic and chemical studies showed that there were silicate inclusions in this meteorite, including small basaltic inclusions and even larger basaltic/gabbroic segregations up to several centimeters (an inch or so) wide. These segregations are not clasts like those within a breccia, as they have transitional margins located mainly at silicate/metal alloy boundaries only a few centimeters (an inch or so) from the fine-grained ultramafic silicates. This is similar to what occurs in winonaites, which is why the IAB iron meteorites like Caddo County are classified with the winonaites in the winonaites-IAB-IIICD silicates clan. Even the metal compositions of IAB and IIICD iron meteorites are similar enough to warrant the inclusion of these meteorites in this clam within the primitive achondrites (Mittlefehldt 2005).
The winonaites-IAB-IIICD silicate inclusions clan contains members with “primitive” chondritic compositions, basalt-depleted lithologies, and lithologies containing mafic segregations (Benedix et al. 2000). Modal plagioclase contents are like those of O chondrites, and some winonaites contain relict chondrules. Texturally winonaites show internal heterogeneity and substantial differences between members (Mittlefehldt 2005). The textures are typically metamorphic. Winonaites contain olivine, orthopyroxene, clinopyroxene, plagioclase, troilite, Fe-Ni alloys, chromite, apatite, graphite, K-feldspar, daubreelite [FeCr2S4], schreibersite [(Fe,Ni)3P], and alabandite [MnS]. The silicate and other inclusions in IAB iron meteorites are heterogeneous and have been subdivided into five types—(i) angular, chondritic silicate; (ii) non-chondritic silicate-rich; (iii) sulfide-rich; (iv) rounded, often graphite-rich; and (v) phosphate-bearing (Benedix et al. 2000). The graphite-rich inclusions only rarely contain silicates, and some sulfide-rich inclusions contain silicates that are apparently angular chondritic silicates. The mineralogy of these silicate inclusions in IAB iron meteorites are diverse, likely due to reaction with the host Fe-Ni metal alloys (Mittlefehldt 2005). They contain olivine, orthopyroxene, clinopyroxene, plagioclase, troilite, Fe-Ni alloys, chromite, daubreelite, graphite, and the phosphates apatite, whitlockite, and rarely, brianite [Na2CaMg(PO4)2].
The coarse-grained gabbroic inclusions in the Caddo County IAB iron meteorite consists of chromian diopside up to 2 × 1.3 mm (~0.08 × 0.05 in) in size set between plagioclase crystals more than 3 mm (0.12 in) in length (Takeda et al. 2000). The modal abundances are plagioclase (59%) and diopside (28%), with less abundant small, rounded grains of orthopyroxene (5%) and olivine (8%) (fig. 2). The coarse-grained gabbroic material is not a clast included in the Fe-Ni metal alloys, or in the finer-grained, recrystallized chondritic material. Rather, it is within the silicate masses, with transitional boundaries with the surrounding ultramafic silicates. In the transitional region between the two lithologies medium-sized orthopyroxene and olivine grains are dominant. The olivine with Fa2.5–3.3 is strongly magnesian, as is the fairly uniform orthopyroxene with En90–94Fs5–7Wo1–2.5 (Benedix et al. 2000; Takeda et al. 2000). The plagioclase with An16–18Ab79–81Or3 is within the range typical of primitive achondrites. Diopside (clinopyroxene) crystals display some slight zoning in CaO at the expense of MgO (En53–61Fs2.5Wo37–44) and in Cr (0.5–1.2 wt% Cr2O3).

Fig. 2. Photomicrograph of a silicate inclusion in the primitive achondrite the Caddo County IAB iron meteorite in transmitted light with partially crossed polars. This inclusion is a basaltic clast composed of plagioclase (pl, gray), clinopyroxene (cpx, whitish) and olivine (ol, pink) (after Krot et al. 2005).
Uniformitarians suggest it is likely that both the IAB irons and the winonaites came from the same parent body (Norton 2002). Their textures and compositions suggest that they are a mix of chondrite and metal breccia. Since iron and silicates are immiscible and tend to separate in a melt, finding silicates associated with irons would not be expected. In fact, the relative rarity of silicate inclusions in iron meteorites would seem to be a strong argument for iron meteorites formed by magmatic processes within differentiated asteroids resulting in metallic core formation. However, these silicated irons, such as the IAB iron meteorites, do not seem to have been associated with magmatic processes that led to differentiation because their trace elements show only weak trends or slopes that are inconsistent with fractional crystallization. Yet the silicates were heated sufficiently to recrystallize them or in some cases melt them (hence the metamorphic textures), but the metal was not completely melted. Thus there is no consensus yet on how the metal phase was formed—impact melting, fractional melting with melt segregation, batch melting with melt segregation, and fractional crystallization are all suggested mechanisms (Mittlefehldt 2005). But all these suggested processes have serious difficulties, as they are not supported by the observed textures and compositions of these meteorites, or experimentally derived solid metal/liquid metal partition coefficients. The most comprehensive model attempting to explain the origin of these meteorites proposes the breakup and reassembly of a hot, partially differentiated parent body (Benedix et al. 2000). The breakup and reassembly event is supposed to have effectively frozen the evolution of the parent body, and mixed different materials together.
The Other Achondrites
The other achondrites are sometimes referred to as the differentiated achondrites because they would appear to represent the products of classical (as understood here on the earth) igneous processes acting on the silicate-oxide system of asteroidal bodies—partial to complete melting, and magmatic crystallization (Mittlefehldt 2005). Iron meteorites represent the complementary metal-sulfide system products of this process. These other achondrites therefore include the so-called asteroidal meteorites— the HED or Vesta clan (howardites, eucrites, and diogenites), the angrites, and the aubrites; the so-called stony-irons—the mesosiderites and pallasites; and all the other groups of iron meteorites (fig. 1). Snelling (2014c) has already discussed the HED clan meteorites, particularly the eucrites (basaltic achondrites). The martian (SNC) and lunar meteorites are also achondrites.
Angrites—Angra dos Reis (ADOR) and D’Orbigny
The angrite group currently consists of 13 meteorites, the best known of which are Angra dos Reis (ADOR), LEW (Lewis Cliffs) 86010, Asuka 881371, D’Orbigny, and Sahara 99555 (Mittlefehldt 2005). These meteorites are linked by identical oxygen-isotopic compositions, similar unusual mineralogy, and several distinctive geochemical characteristics. Although some are petrologically anomalous, the preponderance of distinctive characteristics indicates that they plausibly originated on a common parent body. The angrites thus appear to be medium- to coarse-grained mafic igneous rocks of basaltic composition (though critically silica undersaturated and highly alkali-depleted) from the crust of a differentiated asteroid (Krot et al. 2005; Mittlefehldt 2005). They are believed by uniformitarians to have formed as partial melts of primitive source materials under relatively oxidizing conditions. As the angrites do not seem to form a single fractionation sequence, several parent melts are required, although many could have followed a similar partial melting/crystallization path. The unusual pyroxene compositions in angrites (see below) appear familiar to the Ca-Al-Ti inclusions in CV chondrites (carbonaceous, Vigarano-like chondrites) such as Allende (Snelling 2014a). If there was a chondritic precursor for these angrites, the closest would likely be the CV chondrites.
The first recognized and most famous angrite is the Angra dos Reis (ADOR), a single stone weighing about 1.5 kg (3.3 lb) which fell into Angra dos Reis Bay in the State of Rio de Janeiro, Brazil, in January 1869 (Norton 2002). It remained a unique meteorite for over a century. Then over three consecutive years three more angrites were found and recognized from the Antarctic ice-fields—LEW 86010 (found in 1986), LEW 87051 (found in 1987), and Asuka 881371 (found in 1988). Subsequently the remarkable specimen Sahara 99555, a single stone weighing 2.71 kg (5.97 lb), was found in May 1999 in the Libyan Sahara Desert. And recently (in 2001) the new angrite D’Orbigny, a 16.55 kg (36.49 lb) stone, became available for study, though it had been found back in July 1979 by a farmer in a corn field near Buenos Aires, Argentina (Grossman and Zipfel 2001; Kurat et al. 2004).
The Angra dos Reis (ADOR) meteorite is an ultramafic igneous rock, 95% composed by an unusual variety of clinopyroxene with a Ca-Al-Ti-rich and very low Fe content commonly called fassaite [Ca(Mg,Fe,Ti,Al)(Si,Al)2O6], but formerly named by the International Mineralogical Association as aluminian-ferrain-diopside (Mittlefehldt 2005; Norton 2002). This pyroxene in ADOR is homogeneous with a composition of En33Fs12Wo55. Accessory minerals include olivine and the Ca-rich olivine kirschsteinite [Ca(Mg,Fe)SiO4] with uniform compositions of Fo53 and Fo38 respectively. Only trace plagioclase (An>99) is sometimes present. Aluminous spinel, troilite, whitlockite, ulvöspinel [Fe2TiO4]-magnetite [Fe3O4] solid solution, and Ni-rich metal are common minor or trace phases. ADOR has an equilibrated texture, with groundmass olivine and pyroxene occurring as small xenomorphic, equidimensional grains, and larger poikilitic pyroxene grains enclosing smaller grains.
D’Orbigny when found was mostly covered with dark gray-brown fusion crust, and had a somewhat unusual shape (Kurat et al. 2004) (fig. 3). This meteorite had a shape like a loaf of bread, with front and back shields curved in a semi-parallel way and intergrown on one side with the opposite side opening like a clam. The compact rock macroscopically had a medium-grained micro-gabbroic texture with abundant scattered vesicles or druses sometimes lined with glasses (Grossman and Zipfel 2001; Kurat et al. 2004; Mikouchi and MacKay 2001) (fig. 4). Its texture is a quenched igneous texture— ophitic to subophitic, common graphic intergrowths of olivine and plagioclase, and some are porphyritic with coarse subhedral to euhedral olivine grains up to 1 cm (0.4 in) in diameter (fig. 5). The modal abundance is plagioclase 39.4%, fassaite 27.7%, Mg-rich olivine 19.4%, kirschsteinite [CaFeSiO4] 11.9%, spinel (mostly ulvöspinel) 0.6%, troilite 0.5%, and Ca silico-phosphate 0.5% (Kurat et al. 2004; Mikouchi and McKay 2001). Trace phases include the Fe-Ni metal taenite, Ni-pyrrhotite and pentlandite. The fassaite and olivine show extensive chemical zoning, whereas the plagioclase is homogeneous at An100. The fassaite has more magnesian cores at En63, but rims are zoned down to En0,. Its Al2O3 and TiO2 contents are high and variable. The TiO2 content increases from 1.4 wt% in the cores to 5 wt% in the rims, whereas Al2O3 shows complex variation with En content, usually being homogeneous at 8 wt% in the cores, but dropping to 6 wt% at the outer rims. Olivine phenocrysts have cores with Fo63 but are zoned, the CaO content increasing as Fo decreases from 0.8 wt% in the cores to ~8 wt% at Fo10, at which point the olivine becomes an intergrowth of sub-calcic kirschsteinite and Ca-rich fayalite (Fe-rich olivine). Granoblastic olivine grains that are interpreted to be xenocrystic are much more Mg-rich at Fo90.



Fig. 3. The D’Orbigny angrite meteorite as it was found. (A) Front shield with elongated regmaglypts (small, well-defined indentations or pits on the surface produced by selective erosion during passage through the atmosphere) pointing radially from the stagnation point (center left) and open round vugs of variable sizes. Scars are from partially removed fusion crust gouged by the plow. Specimens for first identification and bulk analysis were taken from the compact lithology of the front shield at upper right (visible cut). (B) Side view at lower part in (A) showing the edge at which the front shield and the back side pan, both consisting of compact rock, are intergrown. Note the open round vugs and the porous lithology to the right and left of the pan (large irregularly shaped open spaces). Pan and shield are covered by fusion crust, edges are worn and without fusion crust. (C) Inclined view at back side pan from upper side in (A), opposite side from (B), depicting the pan and the porous lithology situated between the compact shield and pan lithologies. Note the abundant irregularly shaped open spaces, some of which are partially filled by caliche (white). Length of the meteorite was 34 cm (13.4 in) (after Kurat et al. 2004).

Fig. 4. Small rock chips of the D’Orbigny angrite meteorite (after Mikouchi and McKay 2001). (a) Note fine-grained lithology at the right end of chip. (b) Note pale green olivine megacryst.

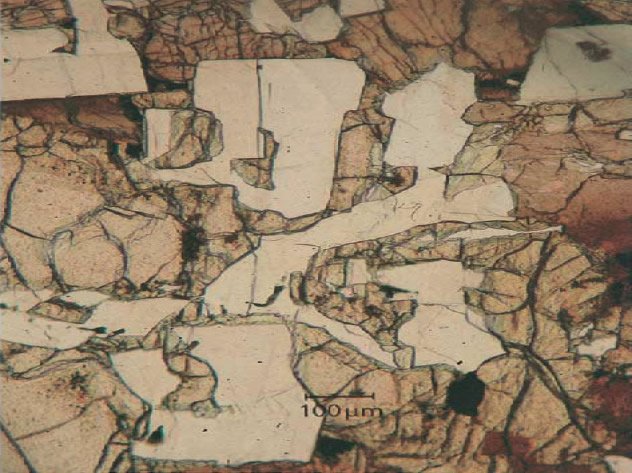
Fig. 5. Anorthite (white)-olivine (brown) intergrowth in transmitted light. (a) Both phases are intimately intergrown, olivine is in places replaced by augite (dark brown). (b) Same as (a) but under uncrossed polarizers. Olivine has mostly the same optical orientation (consists of a single crystal) and anorthite forms many twinned individual grains (after Kurat et al. 2004).
Aubrites—Norton County and Shallowater
About 8% of all known achondrites are aubrites, named after the fall of the type specimen near Aubres, France, on September 14, 1836 (Keil 2010; Norton 2002). The aubrites (or enstatite achondrites) are highly reduced enstatite pyroxenites, their silicate minerals being essentially FeO free (Krot et al. 2005; Mittlefehldt 2005). They are nearly all monomict breccias, with very coarse fragments of enstatite many millimeters across, embedded in a matrix of crushed and comminuted enstatite. They consist of 75–98 vol. % FeO-free enstatite (En>99), with variable lesser amounts of albitic plagioclase (Ab88–95), and virtually FeO-free, high-Ca diopside and Mg-olivine (forsterite). Aubrites have only small amounts of iron averaging <2 wt% Si-bearing Fe-Ni metal, with the remainder as troilite. Other phases include a host of accessory unusual sulfides in which elements that are normally lithophile are chalcophile, such as Ti in heidite [FeTi2S4], Na in caswellsilverite [NaCrS2], and Ca in oldhamite [CaS]. Clasts of both igneous and impact-melt origin are common, and the precursors to the breccias were mostly coarse-grained (probably plutonic) orthopyroxenites, that is, very coarse enstatite grains exhibiting remnant original igneous textures.
Aubrites share nearly identical unusual silicate mineralogy and oxygen-isotopic composition, and a similar highly reduced nature with enstatite (E) chondrites. This suggests to uniformitarians that they may be closely related to enstatite chondrite-like parent bodies (Krot et al. 2005; Mittlefehldt 2005; Norton 2002). However, even though the aubrites perhaps formed from the partial melting of a single body, they are not thought to have formed on the same parent bodies as the E chondrites. Yet to uniformitarians the aubrites are clearly of igneous origin in which large enstatite crystals formed by extensive fractional crystallization of an Mg-rich ultramafic magma, although some suggest that while the aubrite parent body was melted sufficiently to segregate metal and sulfide into a core, large-scale silicate fractionation did not occur. Nevertheless, since the aubrites now are nearly all breccias, their parent body must have melted, recrystallized, and then fragmented by collision, later to be reassembled into an enstatite rubble pile (asteroid).
The Norton County aubrite fell on September 14, 1948 in Norton County, Kansas, and when recovered the stone weighed 998 kg (2200 lb) (Keil 2010). It is the largest known and thus probably the best-studied aubrite, largely because of the ample material available for research (fig. 6). It is a fragmental impact breccia consisting of coarse and angular enstatite crystal fragments, embedded into a clastic matrix, mostly made of comminuted enstatite (fig. 7), together with a host of mineral and lithic fragments of igneous and impact-melt origin (Keil 2010). The precursor materials were most likely plutonic igneous rocks (fig. 8), which formed (in uniformitarian thinking) through extensive melting and igneous differentiation of the aubrite parent body, a process responsible for the formation of the observed diverse lithologies. These include dunites (represented by forsterite crystals), plutonic orthopyroxenites (represented by most enstatite crystals in the matrix), plutonic pyroxenites (the pyroxenitic clasts; fig. 8), plagioclase-silica rocks (like the feldspathic clasts), and impact-melt breccias (the micro-porphyritic clasts and the diopside-plagioclase-silica-clast). The most noteworthy are the pyroxenitic clasts, which occur in large, crystalline masses up to 8 cm (3 in) in size in many hand specimens and are clearly of plutonic origin. They consist of xenomorphic-granular and tightly intergrown, coarse enstatite (En99Wo1), with minor primary diopside (En55Wo45), and accessory kamacite, taenite, titanoan troilite, oldhamite [CaS], and caswellsilverite (fig. 8). Xenomorphic plagioclase (Ab78–88) occurs in the interstices between enstatite grains and forms myrmekitic intergrowths with silica (probably tridymite). Casanova, Keil, and Newsom (1993) studied the mineralogy and trace element composition of metal and metal nodules, up to 1.5 cm (0.6 in) in size, in the Norton County aubrite and concluded that the metal did not undergo fractional crystallization in a core but, rather, is a fraction of the metallic Fe-Ni which, during partial melting of an enstatite chondrite-like precursor lithology, was not completely segregated from the silicates. A remarkable, oldhamite-dominated igneous lithology consists of clasts containing oldhamite [CaS] single crystals up to 2 cm (0.8 in) in size, with inclusions of ferromagnesian alabandite [MnS], troilite, daubreelite [FeCr2S4], caswellsilverite [NaCrS2], and metallic Fe-Ni, which are usually in intimate contact with a silicate portion consisting of enstatite, forsterite, and/or plagioclase and testify to the formation of the oldhamite from a melt (Wheelock et al. 1994).

Fig. 6. The Norton County aubrite meteorite (after Norton 2002). This is a monomict breccia with coarse clasts of enstatite and a comminuted matrix of the same mineral. Fe-Ni metal is present as rusty brown grains on the edges of the specimen. The face measures 32 mm (1.26 in) across.

Fig. 7. Photomicrograph of the brecciated texture of the Norton County aubrite meteorite, which is typical for the common aubrite fragmental impact breccias, consisting mostly of fragments of enstatite and enstatite-dominated lithic clasts of igneous parentage. Transmitted light, polars partially crossed. Longest horizontal dimension equals 6 cm (2.4 in) (after Keil 2010).

Fig. 8. Photomicrograph of the plutonic, xenomorphic-granular texture of a pyroxenitic clast from the Norton County aubrite, dominated by large enstatite crystals with diopside exsolution and minor diopside with enstatite exsolution. Transmitted light, polars partially crossed. Longest horizontal dimension is 3.5 cm (1.37 in) (after Keil 2010).
The Shallowater aubrite was found in Lubbock County, Texas in 1936 and weighed in at 4.6 kg (10 lb) (Keil 2010; Keil et al. 1989). It is a non-brecciated, coarse-grained orthopyroxenite that consists of large, up to 4.5 cm (1.8 in), subhedral poikilitic orthoenstatite crystals (up to 80 vol.%) which enclose smaller orthoenstatite crystals of identical composition (En>99) and which also contain as inclusions and in the interstices xenoliths of an assemblage of low-Ca clinoenstatite (1 vol.%), forsterite (Mg-olivine) (2.9 vol.%), plagioclase (Ab80–88An0–17Or3–11) (2.5 vol.%), metallic Fe-Ni (3.3 vol.%, which is more than five times more than in other aubrites), troilite (2.9 vol.%), schreibersite [(Fe,Ni)3P] (0.4 vol.%), weathered opaques (8 vol.%), and traces of niningerite (a magnesium-iron-manganese sulfide) and oldhamite [CaS] (Keil et al. 1989). Regarded as definitely of igneous origin, the rock is deduced to have experienced a complex, three-stage cooling history, which prompted Keil et al. (1989) to suggest that its distinct parent body formed by the reassembly of the debris from the collision of a partly or totally molten enstatite-rich aubrite-like body with a solid, enstatite-chondrite-like body (represented by the xenoliths), followed by rapid cooling and crystallization. They also suggested that this was not the main aubrite parent body (asteroid) from which other aubrites came, but rather that Shallowater represents a sample from a second different aubrite parent body.
Mesosiderites—Estherville
Mesosiderites are stony-iron polymict breccias composed of angular fragments of different mineral compositions, an unusual mixture of silicates mostly from basaltic, gabbroic and orthopyroxenite sources with Fe-Ni metal and troilite (Mittlefehldt 1990, 2005; Norton 2002). The silicate fragments and Fe-Ni metal are in nearly equal proportions, which is why these meteorites were originally classified as stony-irons. The Fe-Ni metal may be uniformly distributed within a silicate matrix with large silicate fragments enclosed by the Fe-Ni metal, or the Fe-Ni metal may be found in clumps or aggregates surrounded by silicates, as in the case of the Estherville mesosiderite (fig. 9). Superficially they appear distinct from the basaltic (HED) achondrites because of their large Fe- Ni metal content, but mineral, chemical, and oxygen isotopic connections are recognized between the basaltic (HED) achondrites and the mesosiderites. It is because the silicate fragments are of crustal rocks from a differentiated body with both the rock fragments and silicate minerals very similar to those in the HED suite basaltic achondrite meteorites that both the mesosiderites and HED achondrites were the result of impact mixing on different parent asteroidal bodies.

Fig. 9. The mesosiderite meteorite Estherville (after Norton 2002). This slab is about 10 cm (about 4 in) across at its longest dimension. The Fe-Ni metal is fragmental and in aggregates scattered through the dark silicate matrix.
The silicates consist of mineral and lithic clasts set in a fine-grained fragmental to impact-melt matrix (Mittlefehldt 2005). The most common lithic clasts are basalts, gabbros, and orthopyroxenites, while dunites are minor and anorthosites are rare. The most common mineral clasts are centimeter-sized orthopyroxene and olivine fragments, while millimeter-sized plagioclase fragments are less common. Olivine clasts are typically single-crystal fragments, varying in composition from Fo92 to Fo58. Mesosiderites also contain fine-grained olivine whose compositions may have been altered by metamorphic equilibration. The pyroxene clasts are dominated by the low-Ca orthopyroxene hypersthene with smaller amounts of low-Ca pigeonite. Compositional ranges are from about Fs20 to Fs40. Plagioclase grains are calcic, almost pure anorthite. Basaltic and gabbroic clasts are composed of ferroan pigeonite and calcic plagioclase, with minor to accessory silica, whitlockite, augite, chromite, and ilmenite. Fe-Ni metal and troilite are common, but these have been added during brecciation. The matrix varies from cataclastic texture with highly angular mineral fragments to igneous-textured. The Fe-Ni metal portion of mesosiderites has a uniform composition unlike iron meteorites, with a Ni content of between 7 and 10%.
The fall of the Estherville mesosiderite in Iowa, USA occurred on May 10, 1879 (Powell 1971). After a brilliant fireball had been seen, a shower of several large masses and many small fragments fell, totaling 320 kg (710 lb). Estherville’s Fe-Ni metal distribution is patchy surrounded by silicates so that the Fe-Ni metal clumps and aggregates which total 56 wt% of this meteorite appear isolated in planar sections (fig. 9). Its texture is characterized by brecciated, angular silicate lithic fragments and individual silicate crystals. The major silicate mineral constituents (> 10 vol.%) are calcic plagioclase and orthopyroxene, while pigeonite and olivine are minor constituents (2–10 vol.%), and accessories (< 2 vol.%) include augite, tridymite, apatite, whitlockite, schreibersite, chromite, and ilmenite (Powell 1971). Lamellae of rutile occur within the ilmenite, and troilite is also an accessory phase as small irregular blebs (small, usually rounded inclusions) interstitial to the silicate grains within the lithic fragments.
IIE Irons—Colomera, Kodaïkanal, and Weekeroo Station
Of the meteorites seen to fall, only about 4% are irons (Norton 2002). Yet some 40% of all finds are irons, because they are stronger and more easily pass through the earth’s atmosphere with less fragmenting than stony meteorites so they tend to be larger and far easier to recognize and find than stony meteorites. Even though they are subject to rusting, being composed of Fe-Ni alloys, they are still much more resistant to weathering than stony meteorites. The cut face of an iron meteorite is usually featureless, its apparent uniformity only broken by occasional inclusions. However, if the surface is etched with dilute nitric acid a unique texture appears. Called Widmanstätten structure, it is the result of the intergrowth of lamellae or plates of two Fe-Ni alloys, the low-Ni kamacite and high-Ni taenite. Iron meteorites that display this Widmanstätten structure are called octahedrites, since the intergrown kamacite/taenite plates are arranged parallel to the eight equilateral triangular faces of a back-to-back pyramidal structure called an octahedral dipyramid or simply an octahedron.
Irons are regarded by uniformitarians as differentiated meteorites that represent the cores of parent asteroidal bodies. In their view, precursor refractory minerals first condensed out of the solar nebula. After a brief period of heating, chondrules formed and crystallized. Then the original chondritic parent body formed through accretion and agglomeration of these chondrules and other solid condensates such as Fe-Ni metal grains. After building up to perhaps 100–200 km (about 60–120 mi) in diameter, the chondritic body supposedly melted and differentiated to produce a layered differentiated parent body with a metallic core surrounded by a largely silicate mantle and crust. Subsequently, the crust and mantle rocks were stripped away by multiple impacts to expose and eventually denude the metallic core. Finally, the Fe-Ni metallic core which may have measured several kilometers (a few miles) across would have been fragmented.
There are two classifications of iron meteorites based on either structural or chemical criteria. Earlier classifications were based on the structural appearance of the cut and etched surfaces of irons, especially the texture of the Widmanstätten structure, that is, on how wide or narrow are the kamacite plates (Norton 2002). The width depends on the bulk Ni content, so the structural classification is based on band width of the kamacite lamellae and Ni content (table 1). This is primarily an octahedrite classification with seven sub-groups ranging from coarsest to finest kamacite plate widths as their Ni content increases. Two other iron meteorite groups define the low and high end of the Ni content range, the hexahedrites and ataxites respectively. However, there are a few unique iron meteorites that do not conform to any of these classification groups, as they display unusual structures and/or chemistry, such as a random-appearing polycrystalline texture, troilite nodules, and/or silicate inclusions. Furthermore, the textural groups in this structural classification give the impression they are distinct from each other. Yet they are not well-defined groups but grade smoothly from one texture into another. The Ni content boundaries for each textural group are also somewhat arbitrary.
Table 1. Structural classification of iron meteorites according to their internal structure (after Norton 2002).
| Group | Symbol | Band Width (mm) | Nickel (%) |
|---|---|---|---|
| Hexahedrite | H | >50 | 4.5–6.5 |
| Octahedrites | O | ||
| Coarsest | 0gg | 3.3–50 | 6.5–7.2 |
| Coarse | Og | 1.3–3.3 | 6.5–7.2 |
| Medium | Orn | 0.5–1.3 | 7.4–10.3 |
| Fine | Of | 0.2–0.5 | 7.8–12.7 |
| Finest | Off | <0.2 | 7.8–12.7 |
| Plessitic | Opl | <0.2 | Kamacite spindles |
| Ataxite | D | No structure | >16.0 |
The structural parameters in that structural classification have been combined with several chemical parameters such as nickel and trace element contents to produce a more definite classification with meaningful distinct genetic groups that could represent different parent bodies (table 2). There are certain trace elements such as gallium (Ga), germanium (Ge), and iridium (Ir) that like Ni are siderophile (or iron-loving), so they are used to sub-divide the iron meteorites into distinct chemical groups. Experiments have shown that because Ni tends to accumulate and concentrate in the liquid phase, then the first solid Fe-Ni alloy accumulating, presumably at the developing core of a differentiated asteroid, is relatively low in Ni. As the crystallization process continues both the melt and crystallizing solid metal become richer in Ni, although the metal is still less rich in Ni than the melt. This fractional crystallization results in metal alloys with various Ni contents. These three siderophile trace elements have different affinities for Fe as a solid versus Fe as a liquid, so fractional crystallization affects them much more strongly. For example, Ir prefers to combine with the first crystallizing Fe metal which is low in Ni. With increasing Ni concentration in the melt, the amount of Ir in the crystallizing metal decreases since most of it is partitioned into the first metal to crystallize.
Table 2. Structural and chemical relationships in iron meteorites (after Norton 2002).
| Chemical Groups | Frequency (%) | Band Width (mm) | Ni (wt%) | Structural Groups |
|---|---|---|---|---|
| IA | 17.0 | 1.0–3 | 6.4–8.7 | Ogg, Og, Om |
| IB | 1.7 | 0.01–1.0 | 8.7–25 | Om → D |
| IC | 2.1 | <3 | 6.1–6.8 | Anom, Og |
| IIA | 8.1 | >50 | 5.3–5.7 | H |
| IIB | 2.7 | 5–15 | 5.7–6.4 | Ogg |
| IIC | 1.4 | 0.06–0.07 | 9.3–11.5 | Opl |
| IID | 2.7 | 0.4–0.8 | 9.6–11.3 | Om, Of |
| IIE | 2.5 | 0.7–2 | 7.5–9.7 | Ogg → Om (Anom) |
| IIF | 1.0 | 0.05–0.21 | 8.4–10 | Of → D |
| IIIA | 24.8 | 0.9–1.3 | 7.1–9.3 | Om |
| IIIB | 7.5 | 0.6–1.3 | 8.4–10.5 | Om |
| IIIC | 1.4 | 0.2–3 | 10–13 | Ogg → Off |
| IIID | 1.0 | 0.01–0.05 | 16–23 | Off → D |
| IIIE | 1.7 | 1.3–1.6 | 8.2–9.0 | Ogg |
| IIIF | 1.0 | 0.5–1.5 | 6.8–7.8 | Ogg → Of |
| IVA | 8.3 | 0.25–0.45 | 7.4–9.4 | Of |
| IVB | 2.3 | 0.006–0.03 | 16–26 | D |
Therefore, when Ir abundances in the crystallized Fe-Ni metals in iron meteorites are plotted against their Ni contents (which have resulted from melts with different initial bulk Ni contents) distinct narrow fields appear, often with negative slopes (fig. 10). These small but distinct fields, made up of five or more meteorites, initially allowed most iron meteorites to be classified into one of 17 chemical groups (table 2). A few of these groups were subsequently found to be related to each other. For example, IIIA and IIIB irons both have very narrow structural and Ni content variations, and both are in the Om structural group, so they are now usually combined as IIIAB irons. Other chemical groups have been similarly paired, thus reducing the number of distinct groups to 13. About 15% of all known iron meteorites do not fall into one of these groups and remain anomalous. A detailed description of the individual iron meteorite groups is found in Scott and Wasson (1975).
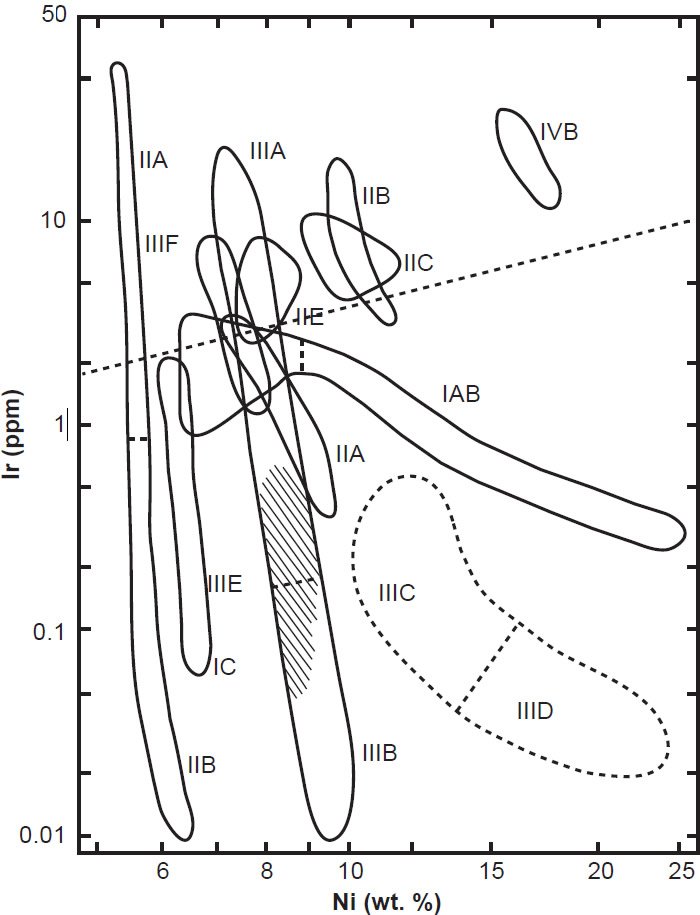
Fig. 10. A log-log plot of Ni (nickel) abundance versus Ir (iridium) abundance in iron meteorites. This allows for resolution of different compositional fields so that the meteorites are divided into differently labelled groups that can be distinguished from each other based on their Ir concentrations (after Scott and Wasson 1975).
Because the purpose of this study was to focus on individual meteorites dated by more than one radioisotope method, the only group of iron meteorites that contained representatives which met that criterion was the IIE irons. They are a small group with very diverse characteristics in terms of metal textures and the mineralogy of their silicate inclusions, which are primarily what has been radioisotope dated. The chemical composition of their Fe-Ni metals is very restricted (figs. 10 and 11) and inconsistent with fractional crystallization (Scott and Wasson 1975; Wasson and Wang 1986). Their silicate inclusions range from angular metamorphosed chondrites (containing chondrules) to highly differentiated silicates in small feldspar-rich globules (Bogard, Garrison, and McCoy 2000).
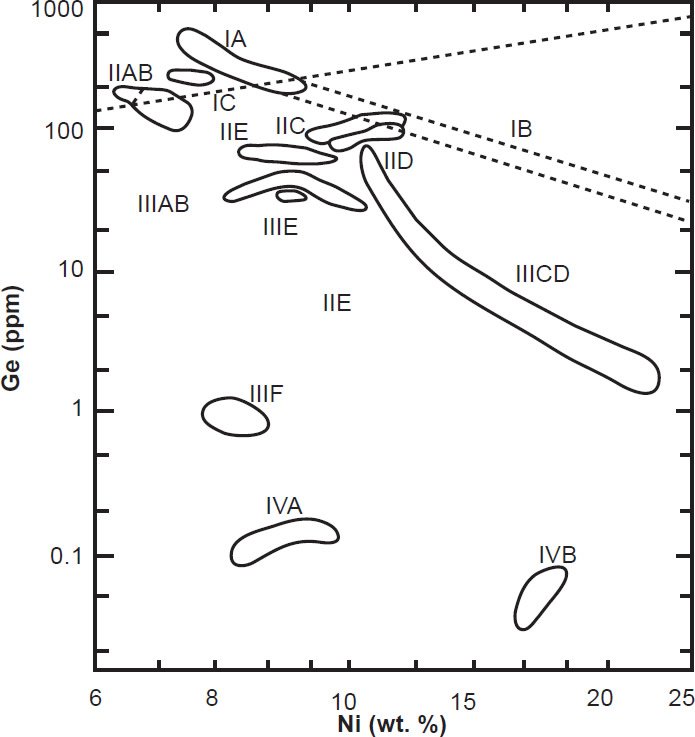
Fig. 11. A log-log plot of the abundance of Ni (nickel) versus Ge (germanium) in iron meteorites. Thirteen fields are distinguished, each representing a different chemical group and a postulated different parent body (after Scott and Wasson 1975).
The IIE irons Colomera, Kodaïkanal, and Weekeroo Station (fig. 12) all contain globular differentiated silicate inclusions (Bogard, Garrison, and McCoy 2000). They are rounded to elongated and typically can reach 1 cm (0.4 in) and comprise about 10 vol. % of the bulk of each meteorite. Inclusions in Weekeroo Station are dominantly orthopyroxene (24 vol. %), clinopyroxene (16 vol. %) and plagioclase (59 vol. %) in a ratio of 1:1:2, whereas Colomera and Kodaïkanal inclusions contain major clinopyroxene (28 and 21 vol. % respectively) and plagioclase (67 and 73 vol. % respectively) in a ratio of ~1:2 or 1:3 with only minor orthopyroxene (both 3 vol. %). Colomera, Kodaïkanal, and Weekeroo Station contain coarse-grained (up to 5 mm or 0.2 in grain size) gabbroic inclusions, partially to wholly cryptocrystalline (consisting of crystals that are too small to be distinguishable under a microscope) inclusions, and glassy inclusions. The ratio of these types can differ significantly. Most inclusions in Weekeroo Station are at least partially cryptocrystalline, and are typified by a corona structure in which large (up to several millimeters) corroded augite (Fs17.66) is rimmed by orthopyroxene (bronzite, Fs22.2) and surrounded by a fine-grained radiating structure of acicular feldspar, tridymite, and glass (Bogard, Garrison, and McCoy 2000; Bunch, Keil, and Olsen 1970). Colomera also contains similar cryptocrystalline, plus gabbroic inclusions consisting of 1–5 mm (0.03–0.19 in) grains of feldspar (both plagioclase and K-feldspar), augite (Fs8–12Wo40–45), and orthopyroxene (Fs19–23), along with millimeter-sized chromite grains, plus accessory rutile, schreibersite, troilite, and whitlockite. Glassy inclusions are found in both Colomera and Kodaïkanal. A variety of shock features are evident in the silicates of these three IIE iron meteorites, including deformation twins, planar fractures, and undulatory extinction.

Fig. 12. A polished and etched slab of the coarse octahedrite iron IIE silicate-bearing meteorite Weekeroo Station. The silicate inclusions consist of plagioclase, orthopyroxene, and clinopyroxene (after Krot et al. 2005).
The Radioisotope Dating of the Primitive and Other Achondrite Meteorites
To thoroughly investigate the radioisotope dating of the primitive and other achondrites all the relevant literature was searched. The objective was to find primitive and other achondrites that have been dated by more than one radioisotope method, and a convenient place to start was Dalrymple (1991, 2004), who compiled lists of such data. The ten primitive and other achondrite meteorites that were found to have been dated multiple times by more than one radioisotope method—the primitive achondrites Acapulco and Caddo County IAB iron, the angrites Angra dos Reis (ADOR) and D’Orbigny, the aubrites Norton County and Shallowater, the mesosiderite Estherville, and the IIE irons Colomera, Kodaïkanal, and Weekeroo Station— thus became the focus of this study. When papers containing radioisotope dating results for these achondrites were found, the reference lists were also scanned to find further relevant papers. In this way a comprehensive set of papers, articles, and abstracts on radioisotope dating of these achondrite meteorites was collected. While it cannot be claimed that all the papers, articles, and abstracts which have ever been published containing radioisotope dating results for these achondrites have thus been obtained, the cross-checking undertaken between these publications does indicate the data set obtained is very comprehensive.
All the radioisotope dating results of these ten primitive and other achondrites were then compiled and tabulated. For ease of viewing and comparing the radioisotope dating data, the isochron and model ages for some or all components of each of these ten achondrites were tabulated separately for each group—the isochron ages and the model ages for the primitive achondrites in Tables 3 and 4 respectively, the isochron ages and the model ages for the angrites in Tables 5 and 6 respectively, the isochron ages and the model ages for the aubrites in Tables 7 and 8 respectively, the isochron ages and the model ages for the mesosiderite Estherville in Tables 9 and 10 respectively, and the isochron ages and the model ages for the IIE irons in Tables 11 and 12 respectively. The data in these tables were then plotted on frequency versus age histogram diagrams, with the same color coding being used to show the ages obtained by the different radioisotope dating methods—the isochron and the model ages for whole-rock samples and some or all components of each of these ten achondrites—the primitive achondrites (figs. 13 and 14 respectively), the angrites (figs. 15 and 16 respectively), the aubrites (figs. 17 and 18 respectively), the mesosiderite Estherville (figs. 19 and 20 respectively), and the IIE irons (figs. 21 and 22 respectively).
Discussion
In contrast to the Allende CV3 carbonaceous chondrite meteorite (Snelling 2014a), there have been fewer radioisotope ages obtained for these five groups of primitive and other achondrite meteorites, because except for the angrite D’Orbigny fewer radioisotope methods have been used on them, and fewer radioisotope determinations have been undertaken. Yet the outcome is similar to that found for the ordinary and enstatite chondrites (Snelling 2014b) and the eucrites (basaltic achondrites) (Snelling 2014c). There is no consistent pattern evident of the β-decay mode K-Ar (and Ar-Ar), Rb-Sr, Lu-Hf, and Re-Os isochron ages increasing in that order according to the parents’ atomic weights or their decay rates (half-lives), or of the α-decay mode Sm-Nd isochron ages always being younger than the U-Th-Pb isochron ages according to the parents’ atomic weights or their decay rates (half-lives). Such a pattern would be potentially indicative of a past episode of accelerated radioisotope decay, as suggested by Snelling (2005c) and Vardiman, Snelling, and Chaffin (2005) from their radioisotope investigations of earth rocks and minerals. Thus it could be concluded from these data that no accelerated radioisotope decay event has occurred on the asteroids which parented these achondrite meteorites.
Table 3. Isochron ages for whole-rock samples and some or all components of the two primitive achondrites Acapulco and Caddo County IAB iron, with the details and literature sources.
| Sample | Method | Date | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
ACAPULCO (acapulcoite) |
||||||
| thirty-three analyses of two aliquots of plagioclases | Ar-Ar | 4.507 | 0.018 | Renne 2000 | isochron age | |
| phosphates | Pb-Pb | 4.557 | 0.002 | Göpel, Manhès, and Allègre 1992 abstract | isochron age | |
| sixteen samples | 207Pb-206Pb | 4.5565 | 0.0013 | phosphates—external normalization (EN) | Amelin 2005; Amelin, Wadhwa, and Lugmair 2006 | isochron age |
| sixteen samples | 207Pb-206Pb | 4.5565 | 0.001 | phosphates—double spike (DS) | Amelin 2005; Amelin, Wadhwa, and Lugmair 2006 | isochron age |
| sixteen samples | 207Pb-206Pb | 4.55652 | 0.00078 | phosphates—EN+DS | Amelin 2005; Amelin, Wadhwa, and Lugmair 2006 | isochron age |
| three phosphate and three multigrain fractions | 207Pb-206Pb | 4.5551 | 0.0013 | Amelin and Pravdivtseva 2005 | isochron age | |
| three phosphate and three multigrain fractions | U-Pb | 4.555 | 0.0032 | concordia-constrained linear 3D isochron | Amelin and Pravdivtseva 2005 | isochron age |
| four minerals + leachate + residue (fines) | Sm-Nd | 4.605 | 0.032 | Prinzhofer, Papanastassiou, and Wasserburg 1992 | isochron age | |
| Mn-Cr | 4.555 | 0.0012 | Zipfel et al. 1995 | isochron age | ||
| Mn-Cr | 4.551 | 0.0012 | Lugmair and Shukolyukov 1998 | isochron age | ||
| phosphates | I-Xe | 4.557 | 0.002 | Brazzle et al. 1999 | isochron age | |
| feldspars | I-Xe | 4.562 | 0.003 | Brazzle et al. 1999 | isochron age | |
CADDO COUNTY (IAB iron) |
||||||
| fifteen extractions from one sample | Ar-Ar | 4.504 | 0.012 | Bogard, Garrison, and Takeda 2005 | isochron age | |
| twenty-five extractions from one sample | Ar-Ar | 4.51 | 0.008 | Bogard Garrison, and Takeda 2005 | isochron age | |
| twenty extractions from one sample | Ar-Ar | 4.483 | 0.012 | Bogard Garrison, and Takeda 2005 | isochron age | |
| mineral separates plus whole rock | Rb-Sr | 4.57 | 0.23 | plagioclase, diopside, impure diopside, and whole rock | Liu et al. 2002a | isochron age |
| seven mineral separates (plag., diop.) plus whole rock | Rb-Sr | 4.52 | 0.03 | Liu et al. 2002a separates plus two each more plag and diop. | Liu et al. 2002b | isochron age |
| Plag and cpx separates (silicate inclusion) | Sm-Nd | 4.41 | 0.03 | Stewart, Papanastassiou, and Wasserburg 1993 | isochron age | |
| three samples of cpx and plag. | Sm-Nd | 4.53 | 0.02 | Stewart, Papanastassiou, and Wasserburg 1996 | isochron age | |
| mineral separates plus whole rock | Sm-Nd | 4.5 | 0.04 | plagioclase, diopside, impure diopside, and whole rock | Liu et al. 2002a | isochron age |
| eight extractions from one sample | I-Xe | 4.5579 | 0.0001 | Bogard, Garrison, and Takeda 2005 | isochron age | |
Table 4. Model ages for whole-rock samples and some or all components of the two primitive achondrites Acapulco and Caddo County IAB iron, with the details and literature sources.
| Sample | Method | Date | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
ACAPULCO |
||||||
| six extractions | Ar-Ar | 4.5 | 0.01 | Bogard et al. 1993 | plateau age | |
| Ar-Ar | 4.503 | 0.011 | McCoy et al. 1996 | step heating age | ||
| Ar-Ar | 4.51 | 0.02 | McCoy et al. 1996 | plateau age | ||
| Ar-Ar | 4.514 | 0.016 | Pellas et al. 1997 | plateau age | ||
| Ar-Ar | 4.554 | Renne 2000 | model age | |||
| two plagioclases | Ar-Ar | 4.509 | 0.016 | Renne 2000 | step heating plateau age | |
| Ar-Ar | 4.501 | 0.031 | Renne 2000 | step heating plateau age | ||
| Pellas et al. 1997 age, revised | Ar-Ar | 4.502 | 0.005 | Trieloff, Jessberger, and Fiéni 2001 | plateau age | |
| average of McCoy et al. (1996), Pellas et al. (1997), and Renne (2000) revised | Ar-Ar | 4.504 | 0.003 | Trieloff, Jessberger, and Fiéni 2001 | plateau age | |
| updated decay constants | Ar-Ar | 4.518 | 0.01 | Bogard 2011 | model age | |
| K-Ar | 4.7 | 0.3 | Palme et al. 1981 | model age | ||
| apatite grains (73 measurements) | Pb-Pb | 4.5554 | 0.0052 | weighted mean | Zhou et al. 2012 | model age |
| weighted mean of five oldest of twelve samples | U-Th/He | 4.538 | 0.032 | Min et al. 2003 | model age | |
| weighted mean of three oldest of twelve samples | U-Th/He | 4.576 | 0.03 | Min et al. 2003 | model age | |
| merrilite | Pu-Xe | 4.41 | 0.016 | Pellas et al. 1997 | model age | |
| apatite | Pu-Xe | 4.4 | 0.016 | Pellas et al. 1997 | model age | |
CADDO COUNTY (IAB iron) |
||||||
| mean value of seventeen extractions | Ar-Ar | 4.52 | 0.005 | average | Takeda et al. 2000 | plateau age |
| fifteen extractions from one sample | Ar-Ar | 4.508 | 0.013 | (or 4.528) | Bogard et al. 2005 | plateau age |
| twenty-five extractions from one sample | Ar-Ar | 4.506 | 0.01 | (or 4.527) | Bogard et al. 2005 | plateau age |
| twenty extractions from one sample | Ar-Ar | 4.489 | 0.023 | (or 4.487) | Bogard et al. 2005 | plateau age |
| Ar-Ar | 4.528 | 0.013 | using latest decay constants on Bogard et al. 2005 ages | Vogel and Renne 2008 | plateau age | |
| Ar-Ar | 4.527 | 0.01 | using latest decay constants on Bogard et al. 2005 ages | Vogel and Renne 2008 | plateau age | |
| Ar-Ar | 4.487 | 0.023 | using latest decay constants on Bogard et al. 2005 ages | Vogel and Renne 2008 | plateau age | |
| plagioclase separates from silicate inclusion | Ar-Ar | 4.472 | 0.02 | Vogel and Renne 2008 | plateau age | |
| Ar-Ar | 4.542 | 0.02 | Vogel and Renne 2008 | plateau age | ||
| Ar-Ar | 4.552 | 0.02 | Vogel and Renne 2008 | plateau age | ||
| Ar-Ar | 4.552 | 0.02 | Vogel and Renne 2008 | plateau age | ||
| Ar-Ar | 4.532 | 0.02 | Vogel and Renne 2008 | plateau age | ||
| Ar-Ar | 4.562 | 0.02 | Vogel and Renne 2008 | plateau age | ||
| mineral separates | Rb-Sr | 4.55 | Liu et al. 2002a | model age | ||
| mineral separates | Rb-Sr | 4.59 | Liu et al. 2002a | model age | ||
| Hf-W | 4.561 | 0.002 | Markowski et al. 2006 in Vogel and Renne 2008 | model age | ||
Table 5. Isochron ages for whole-rock samples and some or all components of the two angrite achondrites Angra dos Reis and D’Orbigny, with the details and literature sources.
| Sample | Method | Reading | Err +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
ANGRA DOS REIS (ADOR) |
||||||
| six total meteorite, a pyroxene, and two whitlockite samples | Rb-Sr | 4.47 | 0.24 | Wasserburg et al. 1977 | isochron age | |
| two whole rock samples plotted with one D’Orbigny whole rock sample | Rb-Sr | 4.56 | Hans, Kleine, and Bourdon 2010 | isochron age | ||
| Pb-Pb | 4.5566 | 0.0002 | Kleine et al. 2012 | isochron age | ||
| three samples plotted with St. Severin phosphates | 207Pb-204Pb | 4.558 | 0.006 | Tera and Carlson 1999 | isochron age | |
| isochron with several other meteorites and Canyon Diablo | 207Pb-206Pb | 4.555 | 0.005 | Tatsumoto, Knight, and Allègre 1973 | isochron age | |
| two samples on isochron wih 11 Alende and two St Severin samples | 207Pb-206Pb | 4.551 | 0.004 | Chen and Wasserburg 1981 | isochron age | |
| pyroxene and plagioclase separates | -207Pb-206Pb | 4.5586 | 0.0006 | Zartman, Jagoutz, and Bowring 2006 | isochron age | |
| pyroxene fractions 1R-3R + 5R | 207Pb-206Pb | 4.5571 | 0.0036 | Amelin 2008 | isochron age | |
| all five pyroxene fractions | 207Pb-206Pb | 4.55766 | 0.00013 | Amelin 2008 | isochron age | |
| pyroxene separate plotted with four mined separates from another meteorite | 207Pb/206Pb-204Pb/206Pb | 4.5578 | 0.00042 | Lugmair and Galer 1992 | isochron age | |
| pyroxene fractions 1R-3R + 5R + 5R wash | U-Pb | 4.5576 | 0.00015 | Amelin 2008 | isochron age | |
| all five pyroxene fractions + 4R and 5R washes | U-Pb | 4.5571 | 0.0005 | Amelin 2008 | isochron age | |
| three phosphate and four pyroxene samples | Sm-Nd | 4.55 | 0.04 | Lugmair and Marti 1977 | isochron age | |
| four total rock, four whitlockite, and one pyroxene analeses | Sm-Nd | 4.564 | 0.037 | Jacobsen and Wasserburg 1984 | isochron age | |
| whole rock, pyroxene, and mixed fractions | Hf-W | 4.556 | 0.0009 | Kleine et al. 2012 | isochron age | |
D’ORBIGNY |
||||||
| plagioclase separates, relative to Allende CAI’s | Al-Mg | 4.5603 | 0.0004 | Nyquist et al. 2003 | isochron age | |
| whole rock, olivene, pyroxene and plagioclase separates (relative to LEW 86010) | Al-Mg | 4.5627 | 0.0011 | Spivak-Birndorf, Wadhwa, and Janney 2005 | isochron age | |
| whole rock, olivene, pyroxene and plagioclase (2) separates, relative to E60 Pb-Pb | Al-Mg | 4.56242 | 0.00029 | Spivak-Birndorf, Wadhwa, and Janney 2009 | isochron age | |
| whole rock, olivene, pyroxene and plagioclase (2) separates, relative to CAI Pb-Pb | Al-Mg | 4.5625 | 0.0003 | Schiller, Baker, and Bizzarro 2010 | isochron age | |
| Spivak-Birndorf, Wadhwa, and Janney (2009) data relative to Allende CAI Pb-Pb | Al-Mg | 4.5628 | 0.0004 | Schiller, Baker and Bizzarro 2010 | isochron age | |
| six separates (whole rock and pyroxene); relative to CAIs | Hf-W | 4.5624 | 0.0013 | Markowski et al. 2007 | isochron age | |
| relative to E60 CAI Pb-Pb using Markowski et al (2007) data | Hf-W | 4.5631 | 0.0008 | Spivak-Birndorf, Wadhwa, and Janney 2009 | isochron age | |
| Markowski et al. (2007) data relative to Allende CAI Pb-Pb | Hf-W | 4.5637 | 0.001 | Schiller, Baker, and Bizzarro 2010 | isochron age | |
| relative to Brennecka and Wadhwa (2011) Pb-Pb age: whole rock (2); pyroxene (3); and olivene separates (1) | Hf-W | 4.5634 | 0.0003 | Kleine et al. 2012 | isochron age | |
| recalibration of Kleine et al. (2012) value relative to Connelly et al (2012) Pb-Pb age of CAIs | Hf-W | 4.5628 | 0.0006 | Kruijer et al. 2014 | isochron age | |
| whole rock, pyroxene (2) fractions, relative to LEW 86010 | Mn-Cr | 4.5616 | 0.0005 | Nyquist et al. 2003 | isochron age | |
| whole rock, chromite and two glass fractions | Mn-Cr | 4.5622 | 0.0006 | Glavin, Jagoutz, and Lugmair 2003 | isochron age | |
| eleven fractions—total rock, silicates, spinel (2), olivine (4), pyroxene and glass (2) relative to LEW 86010 | Mn-Cr | 4.5629 | 0.0006 | Glavin et al. 2004 | isochron age | |
| whole rock, olivine, pyroxene and plagioclase separates (relative to LEW 86010) | Mn-Cr | 4.5625 | 0.0005 | Spivak-Birndorf, Wadhwa, and Janney 2005 | isochron age | |
| relative to LEW 86010 | Mn-Cr | 4.5622 | 0.0005 | Sugiura, Miyazaki, and Yanai 2005 | isochron age | |
| whole rock | Mn-Cr | 4.5632 | 0.0006 | Shukolyukov and Lugmair 2007 | isochron age | |
| relative to LEW 86010 Pb-Pb and Glavin et al. (2004) data | Mn-Cr | 4.5637 | 0.0004 | Spivak-Birndorf, Wadhwa, and Janney 2009 | isochron age | |
| relative to LEW 86010 Pb-Pb and Nyquist et al. (2003) and Sugiura, Miyazaki, and Yanai (2005) data | Mn-Cr | 4.5629 | 0.0007 | Spivak-Birndorf, Wadhwa, and Janney 2009 | isochron age | |
| Sugiura, Miyazaki, and Yanai (2005) data relative to Allende CAI Pb-Pb | Mn-Cr | 4.5638 | 0.0007 | Schiller, Baker, and Bizzarro 2010 | isochron age | |
| Glavin et al. (2004) data relative to Allende CAI Pb-Pb | Mn-Cr | 4.5646 | 0.0003 | Schiller, Baker, and Bizzarro 2010 | isochron age | |
| four-step dissolution of whole rock (minus drusy pyroxenes) | Pb-Pb | 4.563 | 0.0025 | Zartman, Jagoutz, and Bowring 2006 | isochron age | |
| pyroxene (5) and whole rock (3) separates | Pb-Pb | 4.56463 | 0.00028 | Amelin 2007 | isochron age | |
| pyroxene (4) and whole rock (5) separates | Pb-Pb | 4.56424 | 0.00029 | Amelin 2008 | isochron age | |
| pyroxene (4) and whole rock (6) separates | Pb-Pb | 4.56453 | 0.0002 | Amelin 2008 | isochron age | |
| plagioclase separate plotted with eleven other samples, relative to LEW 86010 | Rb-Sr | 4.56 | Nyquist et al. 2003 | isochron age | ||
| one whole rock sample plotted with two ADOR whole rock samples | Rb-Sr | 4.56 | Hans, Kleine, and Bourdon 2010 | isochron age | ||
| whole rock sample plotted with whole rock samples of four other angrites | Lu-Hf | 4.576 | 0.049 | Amelin, Wimpenny, and Yin 2011 | isochron age | |
| four mineral fractions and two whole rock samples | Lu-Hf | 4.52 | 0.097 | Sanborn, Carlson, and Wadhwa 2012 | isochron age | |
| whole rock with mafic minerals (2) | Sm-Nd | 4.6 | 0.07 | Nyquist et al. 2003 | isochron age | |
| pyroxene and plagioclase separates | Sm-Nd | 3.08 | 0.05 | Tonui, Ngo, and Papanastassiou 2003 | isochron age | |
| whole rock, pyroxene and plagioclase fractions | Sm-Nd | 4.507 | 0.089 | Sanborn Carlson, and Wadhwa 2011 | isochron age | |
| pyroxene (5) and whole rock (3) separates (3D-linear) | U-Pb | 4.565 | 0.0023 | Amelin 2007 | isochron age | |
| pyroxene (4) and whole rock (5) separates | U-Pb | 4.56464 | 0.00027 | Amelin 2008 | isochron age | |
Table 6. Model ages for whole-rock samples and some or all components of the two angrite achondrites Angra dos Reis and D’Orbingy, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
ANGRA DOS REIS (ADOR) |
||||||
| total meteorite | 207Pb-206Pb | 4.546 | 0.001 | Wasserburg et al. 1977 | model age | |
| whitlockite-A | 207Pb-206Pb | 4.543 | 0.001 | Wasserburg et al. 1977 | model age | |
| whitlockite-B | 207Pb-206Pb | 4.544 | 0.001 | Wasserburg et al. 1977 | model age | |
| whitlockite-C | 207Pb-206Pb | 4.54 | Wasserburg et al. 1977 | model age | ||
| whitlockite | 207Pb-206Pb | 4.553 | 0.008 | Chen and Wasserburg 1981 | model age | |
| whole rock | 207Pb-206Pb | 4.551 | 0.004 | Chen and Wasserburg 1981 | model age | |
| pyroxene separate | 207Pb-206Pb | 4.5578 | 0.00042 | Lugmair and Galer 1992 | model age | |
| five pyroxene fractions—weighted average, primordial Pb | 207Pb-206Pb | 4.55765 | 0.00013 | Amelin 2008 | model age | |
| five pyroxene fractions—weighted average, 204Pb analytical | 207Pb-206Pb | 4.55768 | 0.00013 | Amelin 2008 | model age | |
| fraction 1R, Px | 207Pb-206Pb | 4.5577 | 0.0003 | Amelin 2008 | model age | |
| fraction 1R, Px | 207Pb-206Pb | 4.5577 | 0.0003 | Amelin 2008 | model age | |
| fraction 2R, Px | 207Pb-206Pb | 4.5577 | 0.0003 | Amelin 2008 | model age | |
| fraction 2R, Px | 207Pb-206Pb | 4.5577 | 0.0003 | Amelin 2008 | model age | |
| fraction 3R, Px | 207Pb-206Pb | 4.5575 | 0.0003 | Amelin 2008 | model age | |
| fraction 3R, Px | 207Pb-206Pb | 4.5575 | 0.0003 | Amelin 2008 | model age | |
| fraction 4R, Px | 207Pb-206Pb | 4.5567 | 0.0003 | Amelin 2008 | model age | |
| fraction 4R, Px | 207Pb-206Pb | 4.5587 | 0.0003 | Amelin 2008 | model age | |
| fraction 5R, Px | 207Pb-206Pb | 4.5578 | 0.0003 | Amelin 2008 | model age | |
| fraction 5R, Px | 207Pb-206Pb | 4.5578 | 0.0003 | Amelin 2008 | model age | |
| 4W3, Px Wash-3 | 207Pb-206Pb | 4.5565 | Amelin 2008 | model age | ||
| 5W3, Px Wash-3 | 207Pb-206Pb | 4.5588 | Amelin 2008 | model age | ||
| using 2012 U isotope compositions | Pb-Pb | 4.5566 | 0.00026 | Brennecka and Wadhwa 2012 | model age | |
| total meteorite | 206Pb-238U | 4.63 | 0.07 | Wasserburg et al. 1977 | model age | |
| whitlockite-A | 206Pb-238U | 4.61 | 0.07 | Wasserburg et al. 1977 | model age | |
| whitlockite-B | 206Pb-238U | 4.546 | 0.038 | Wasserburg et al. 1977 | model age | |
| whitlockite-C | 206Pb-238U | 4.5 | Wasserburg et al. 1977 | model age | ||
| whitlockite | 206Pb-238U | 4.5 | 0.2 | Chen and Wasserburg 1981 | model age | |
| whole rock | 206Pb-238U | 4.55 | 0.06 | Chen and Wasserburg 1981 | model age | |
| pyroxene separate | 206Pb-238U | 4.586 | 0.015 | Lugmair and Galer 1992 | model age | |
| fraction 1R, Px | 206Pb-238U | 4.5547 | Amelin 2008 | model age | ||
| fraction 2R, Px | 206Pb-238U | 4.553 | Amelin 2008 | model age | ||
| fraction 3R, Px | 206Pb-238U | 4.556 | Amelin 2008 | model age | ||
| fraction 4R, Px | 206Pb-238U | 4.5685 | Amelin 2008 | model age | ||
| fraction 5R, Px | 206Pb-238U | 4.3984 | Amelin 2008 | model age | ||
| 4W3, Px Wash-3 | 206Pb-238U | 5.5349 | Amelin 2008 | model age | ||
| 5W3, Px Wash-3 | 206Pb-238U | 5.6581 | Amelin 2008 | model age | ||
| total meteorite | 208Pb-232Th | 4.53 | 0.1 | Wasserburg et al. 1977 | model age | |
| whitlockite-A | 208Pb-232Th | 4.6 | 0.11 | Wasserburg et al. 1977 | model age | |
| whitlockite-B | 208Pb-232Th | 4.54 | 0.053 | Wasserburg et al. 1977 | model age | |
| whitlockite-C | 208Pb-232Th | 4.54 | Wasserburg et al. 1977 | model age | ||
| pyroxene separate | 235U-207Pb | 4.566 | 0.0047 | Lugmair and Galer 1992 | model age | |
D’ORBIGNY |
||||||
| whole rock | Al-Mg | 4.56601 | 0.00025 | Baker et al. 2005 | model age | |
| relative to CAI Pb-Pb | Al-Mg | 4.56375 | 0.0004 | Schiller, Baker, and Bizzarro 2010 | model age | |
| relative to D’Orbigny Pb-Pb | Al-Mg | 4.5656 | 0.0004 | Schiller, Baker, and Bizzarro 2010 | model age | |
| whole rock | Pb-Pb | 4.51808 | 0.00009 | Baker et al. 2005 | model age | |
| hand-picked druse pyroxene fractions subjected to leaches of differing severity | Pb-Pb | 4.5632 | 0.0011 | Zartman, Jagoutz, and Bowring 2006 | model age | |
| hand-picked druse pyroxene fractions subjected to leaches of differing severity | Pb-Pb | 4.5643 | 0.0008 | Zartman, Jagoutz, and Bowring 2006 | model age | |
| hand-picked druse pyroxene fractions subjected to leaches of differing severity | Pb-Pb | 4.5638 | 0.0008 | Zartman, Jagoutz, and Bowring 2006 | model age | |
| hand-picked druse pyroxene fractions subjected to leaches of differing severity | Pb-Pb | 4.5694 | 0.0028 | Zartman, Jagoutz, and Bowring 2006 | model age | |
| hand-picked druse pyroxene fractions subjected to leaches of differing severity | Pb-Pb | 4.5585 | 0.0033 | Zartman, Jagoutz, and Bowring 2006 | model age | |
| weighted mean of the first three hand-picked druse pyroxene fractions (above) with two other designated as “aberrant” | Pb-Pb | 4.5639 | 0.0006 | Zartman, Jagoutz, and Bowring 2006 | model age | |
| Pb-Pb | 4.563 | 0.001 | Spivak-Birndorf, Wadhwa, and Janney 2005 | model age | ||
| pyroxene (5) and whole rock (3) separates (weighted average) | Pb-Pb | 4.56448 | 0.00024 | Amelin 2007 | model age | |
| pyroxene (5) and whole rock (8) separates (weighted average assuming primordial Pb) | Pb-Pb | 4.56442 | 0.00012 | Amelin 2008 | model age | |
| pyroxene (5) and whole rock (8) separates (weighted average assuming analytical Pb) | Pb-Pb | 4.56453 | 0.00019 | Amelin 2008 | model age | |
| recalculated Amelin (2008) weighted average (primordial Pb) due to new 238U-235U ratio value | Pb-Pb | 4.5638 | 0.0004 | Brennecka et al. 2010 | model age | |
| corrected for new 238U-235U ratio determination | Pb-Pb | 4.56336 | 0.00034 | Bouvier and Wadhwa 2010 | model age | |
| Pb-Pb | 4.56334 | 0.0003 | Brennecka and Wadhwa 2011 | model age | ||
| Using 2012 U isotope compositions | Pb-Pb | 4.56337 | 0.00025 | Brennecka and Wadhwa 2012 | model age | |
| druse pyroxene fraction | 207Pb-206Pb | 4.559 | 0.0011 | Jagoutz et al. 2002 | model age | |
| groundmass pyroxene fraction | 207Pb-206Pb | 4.548 | 0.0011 | Jagoutz et al. 2002 | model age | |
| weighted mean | 207Pb-206Pb | 4.557 | 0.0015 | Jagoutz et al. 2002 | model age | |
| matrix pyroxene fraction | 207Pb-206Pb | 4.549 | 0.002 | Jagoutz et al. 2003 | model age | |
| matrix pyroxene fraction | 207Pb-206Pb | 4.557 | 0.002 | Jagoutz et al. 2003 | model age | |
| druse pyroxene fraction | 207Pb-206Pb | 4.5554 | 0.0019 | Jagoutz et al. 2003 | model age | |
| druse pyroxene fraction | 207Pb-206Pb | 4.556 | 0.004 | Jagourtz et al. 2003 | model age | |
| druse pyroxene fraction | 207Pb-206Pb | 4.557 | 0.001 | Jagourtz et al. 2003 | model age | |
| 12R Px | 207Pb-206Pb | 4.5666 | 0.0014 | assuming primordial Pb | Amelin 2008 | model age |
| 13R Px | 207Pb-206Pb | 4.5649 | 0.0006 | assuming primordial Pb | Amelin 2008 | model age |
| 15 R WR (fragment) | 207Pb-206Pb | 4.5646 | 0.0004 | assuming primordial Pb | Amelin 2008 | model age |
| 16R WR (fines) | 207Pb-206Pb | 4.5621 | 0.0004 | assuming primordial Pb | Amelin 2008 | model age |
| 17R Px | 207Pb-206Pb | 4.5647 | 0.0004 | assuming primordial Pb | Amelin 2008 | model age |
| 18R Px | 207Pb-206Pb | 4.5642 | 0.0003 | assuming primordial Pb | Amelin 2008 | model age |
| 19R WR minus Px | 207Pb-206Pb | 4.5645 | 0.0003 | assuming primordial Pb | Amelin 2008 | model age |
| 20R WR (rel. fine) | 207Pb-206Pb | 4.5644 | 0.0003 | assuming primordial Pb | Amelin 2008 | model age |
| 22R WR | 207Pb-206Pb | 4.57 | 0.0015 | assuming primordial Pb | Amelin 2008 | model age |
| 23R WR | 207Pb-206Pb | 4.5636 | 0.0013 | assuming primordial Pb | Amelin 2008 | model age |
| 21W3 Px | 207Pb-206Pb | 4.5644 | 0.0004 | assuming primordial Pb | Amelin 2008 | model age |
| 22W3 WR | 207Pb-206Pb | 4.5643 | 0.0003 | assuming primordial Pb | Amelin 2008 | model age |
| 22W3 WR | 207Pb-206Pb | 4.5644 | 0.0004 | assuming primordial Pb | Amelin 2008 | model age |
| 12R Px | 207Pb-206Pb | 4.5673 | 0.0014 | assuming analytical Pb | Amelin 2008 | model age |
| 13R Px | 207Pb-206Pb | 4.5649 | 0.0006 | assuming analytical Pb | Amelin 2008 | model age |
| 15 R WR (fragment) | 207Pb-206Pb | 4.5648 | 0.0004 | assuming analytical Pb | Amelin 2008 | model age |
| 16R WR (fines) | 207Pb-206Pb | 4.5642 | 0.0004 | assuming analytical Pb | Amelin 2008 | model age |
| 17R Px | 207Pb-206Pb | 4.5648 | 0.0004 | assuming analytical Pb | Amelin 2008 | model age |
| 18R Px | 207Pb-206Pb | 4.5642 | 0.0003 | assuming analytical Pb | Amelin 2008 | model age |
| 19R WR minus Px | 207Pb-206Pb | 4.5645 | 0.0003 | assuming analytical Pb | Amelin 2008 | model age |
| 20R WR (rel. fine) | 207Pb-206Pb | 4.5646 | 0.0003 | assuming analytical Pb | Amelin 2008 | model age |
| 22R WR | 207Pb-206Pb | 4.5741 | 0.0015 | assuming analytical Pb | Amelin 2008 | model age |
| 23R WR | 207Pb-206Pb | 4.5642 | 0.0013 | assuming analytical Pb | Amelin 2008 | model age |
| 21W3 Px | 207Pb-206Pb | 4.5645 | 0.0004 | assuming analytical Pb | Amelin 2008 | model age |
| 22W3 WR | 207Pb-206Pb | 4.5643 | 0.0003 | assuming analytical Pb | Amelin 2008 | model age |
| 22W3 WR | 207Pb-206Pb | 4.5644 | 0.0004 | assuming analytical Pb | Amelin 2008 | model age |
| 12R Px | 206Pb-238U | 4.4916 | assuming primordial Pb | Amelin 2008 | model age | |
| 13R Px | 206Pb-238U | 4.6133 | assuming primordial Pb | Amelin 2008 | model age | |
| 15 R WR (fragment) | 206Pb-238U | 4.8416 | assuming primordial Pb | Amelin 2008 | model age | |
| 16R WR (fines) | 206Pb-238U | 4.8929 | assuming primordial Pb | Amelin 2008 | model age | |
| 17R Px | 206Pb-238U | 4.6946 | assuming primordial Pb | Amelin 2008 | model age | |
| 18R Px | 206Pb-238U | 4.9719 | assuming primordial Pb | Amelin 2008 | model age | |
| 19R WR minus Px | 206Pb-238U | 5.0804 | assuming primordial Pb | Amelin 2008 | model age | |
| 20R WR (rel. fine) | 206Pb-238U | 4.9574 | assuming primordial Pb | Amelin 2008 | model age | |
| 22R WR | 206Pb-238U | 3.6722 | assuming primordial Pb | Amelin 2008 | model age | |
| 23R WR | 206Pb-238U | 2.8337 | assuming primordial Pb | Amelin 2008 | model age | |
| 21W3 Px | 206Pb-238U | 5.471 | assuming primordial Pb | Amelin 2008 | model age | |
| 22W3 WR | 206Pb-238U | 5.2906 | assuming primordial Pb | Amelin 2008 | model age | |
| 22W3 WR | 206Pb-238U | 5.5678 | assuming primordial Pb | Amelin 2008 | model age | |
Table 7. Isochron ages for whole-rock samples and some or all components of the two aubrite achondrites Norton County and Shallowater, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
NORTON COUNTY |
||||||
| eight fractions—three breccias and four splits, three enstatites | Rb-Sr | 4.7 | 0.1 | Bogard et al. 1967a | isochron age | |
| samples of feldspar (2), pyroxenes (4), matrix (2), and whole rock (1) | Rb-Sr | 4.48 | 0.04 | Minster and Allègre 1976 | isochron age | |
| whole rock and feldspar (2) samples plotted with eight samples from four other meteorites | Rb-Sr | 4.516 | 0.029 | Minster, Rickard, and Allègre 1979 | isochron age | |
| revised decay constant | Rb-Sr | 4.44 | 0.04 | Bogard, Dixon, and Garrison 2010 | isochron age | |
| Pb-Pb | 4.55 | 0.003 | Huey and Kohman 1973 | isochron age | ||
SHALLOWATER |
||||||
| Ar-Ar | 4.55 | 0.05 | McCoy et al. 1995 | isochron age | ||
Table 8. Model ages for whole-rock samples and some or all components of the two aubrite achondrites Norton County and Shallowater, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
NORTON COUNTY |
||||||
| K-Ar | 4.4 | 0.65 | Geiss and Hess 1958 | model age | ||
| K-Ar | 5.09 | Kirsten, Krankowsky, and Zähringer 1963 | model age | |||
| K-Ar | 4.68 | Kirsten, Krankowsky, and Zähringer 1963 | model age | |||
| 523.3X breccia IV | K-Ar | 4.5 | Bogard et al. 1967a | model age | ||
| 4965F breccia, split A | K-Ar | 4.2 | Bogard et al. 1967a | model age | ||
| 4965F breccia, split B | K-Ar | 4.3 | Bogard et al. 1967a | model age | ||
| 4965F breccia, enstatite | K-Ar | 4.5 | Bogard et al. 1967a | model age | ||
| samples NC-15961, 1300° celsius extraction | Ar-Ar | 4.454 | 0.018 | Bogard, Dixon, and Garrison 2010 | maximum spectrum age | |
| extractions releasing up to 87% of Ar from sample NC-Okada | Ar-Ar | 4.676 | Bogard, Dixon, and Garrison 2010 | average spectrum age | ||
| summed across all extractions from sample NC-Okada | Ar-Ar | 4.198 | Bogard, Dixon, and Garrison 2010 | total age | ||
| Pb-Pb | 4.55 | 0.003 | Huey and Kohman 1973 | model age | ||
SHALLOWATER |
||||||
| Ar-Ar | 4.53 | 0.05 | McCoy et al. 1995 | total age | ||
| five extractions releasing 6–45% Ar | Ar-Ar | 4.539 | 0.003 | McCoy et al. 1995 | plateau age | |
| Ar-Ar | 4.535 | 0.02 | Bogard 2011 | plateau age | ||
| K-Ar | 4.53 | 0.03 | McCoy et al. 1995 | model age | ||
| I-Xe | 4.566 | 0.002 | Brazzle et al. 1999 | absolute age | ||
| I-Xe | 4.5633 | 0.0004 | Gilmour et al. 2006 | closure age | ||
| I-Xe | 4.5623 | 0.0004 | Gilmour et al. 2009; Hohenberg and Pravdivtseva 2008 | closure age | ||
Table 9. Isochron ages for whole-rock samples and some or all components of the mesosiderite achondrite Estherville, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
ESTHERVILLE |
||||||
| Ar-Ar | 3.55 | 0.096 | Murthy, Alexander, and Saito 1978 | isochron age | ||
| Rb-Sr | 4.411 | 0.088 | one sample fits with five other meteorites | Cumming 1969 | isochron age | |
| Rb-Sr | 4.26 | 0.1 | Murthy, Coscio, and Sabelin 1977 | isochron age | ||
| Rb-Sr | 4.24 | 0.03 | Murthy, Alexander, and Saito 1978 | isochron age | ||
| nine separates | Rb-Sr | 4.542 | 0.203 | Sets A and C | Brouxel and Tatsumoto 1991 | isochron age |
| Re-Os | 4.25 | 0.5 | one sample fits with 21 other meteorites | Luck and Allègre 1983 | isochron age | |
| Re-Os | 4.6 | 0.05 | fits to this isochron | Shen, Papanastassiou, and Wasserburg 1998 | isochron age | |
| Pb-Pb | 4.556 | 0.035 | Set A | Brouxel and Tatsumoto 1990 | isochron age | |
| Pb-Pb | 4.553 | 0.035 | Set B—fourteen analyses | Brouxel and Tatsumoto 1991 | isochron age | |
| eleven separates | Sm-Nd | 4.533 | 0.094 | Sets A and C | Brouxel and Tatsumoto 1991 | isochron age |
Table 10. Model ages for whole-rock samples and some or all components of the mesosiderite achondrite Estherville, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
ESTHERVILLE |
||||||
| Ar-Ar | 3.62 | Bogard et al. 1990 | plateau age | |||
| Ar-Ar | 3.94 | 0.1 | two samples averaged with 18 other meteorite samples | Bogard and Garrison 1998 | plateau age | |
| U-Pb | 4.571 | 0.018 | Set A | Brouxel and Tatsumoto 1990 | concordia age | |
| U-Pb | 4.569 | 0.061 | Set B—fourteen analyses | Brouxel and Tatsumoto 1991 | concordia age | |
Table 11. Isochron ages for whole-rock samples and some or all components of the three iron IIE achondrites Colomera, Kodaïkanal, and Weekeroo Station, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
COLOMERA |
||||||
| sixteen inclusions (feldspar, diopside, glass) | Rb-Sr | 4.61 | 0.04 | Sanz, Burnett, and Wasserburg 1970 | isochron age | |
| fifteen inclusions only | Rb-Sr | 4.64 | 0.14 | Sanz, Burnett, and Wasserburg 1970 | isochron age | |
| twenty inclusions (feldspar, diopside, glass) | Rb-Sr | 4.64 | 0.04 | Sanz, Burnett, and Wasserburg 1970 | isochron age | |
| revised decay constant | Rb-Sr | 4.51 | 0.04 | Bogard, Garrison, and McCoy 2000 | isochron age | |
| revised decay constant | Rb-Sr | 4.59 | 0.127 | Snyder et al. 2001 | isochron age | |
KODAÏKANAL |
||||||
| revised decay constant | Rb-Sr | 3.73 | 0.1 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| revised decay constant | Rb-Sr | 3.81 | 0.1 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| eleven density and mineral separates of Burnett and Wasserburg 1967 | Rb-Sr | 3.743 | 0.044 | Snyder et al. 2001 | isochron age | |
| four samples plotted; five samples from three other meteorites | Re-Os | 4.624 | 0.017 | Birck and Allègre 1998 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 207Pb-206Pb | 3.676 | 0.003 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 207Pb-206Pb | 3.675 | 0.003 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 238U-206Pb | 3.68 | 0.023 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 238U-206Pb | 3.679 | 0.005 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 238U-206Pb | 3.684 | 0.022 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 235U-207Pb | 3.677 | 0.009 | Göpel, Manhès, and Allègre 1985 | isochron age | |
| one clinopyroxene and two alkali-rich glass samples extracted from the meteorite (and leached, as well as the leachates) | 235U-207Pb | 3.678 | 0.009 | Göpel, Manhès, and Allègre 1985 | isochron age | |
WEEKEROO STATION |
||||||
| nine fractions plotted | Rb-Sr | 4.37 | 0.2 | Burnett and Wasserburg 1967 | isochron age | |
| Rb-Sr | 4.39 | 0.07 | Evensen et al. 1979 | isochron age | ||
| Rb-Sr | 4.28 | 0.23 | Bogard, Garrison, and McCoy 2000 | isochron age | ||
| Rb-Sr | 4.64 | 0.04 | Snyder et al. 2001 | isochron age | ||
| Rb-Sr | 4.35 | 0.07 | Snyder et al. 2001 | isochron age | ||
| Rb-Sr | 4.33 | 0.23 | Snyder et al. 2001 | isochron age | ||
| Sm-Nd | 0.7 | Snyder et al. 2001 | isochron age | |||
| Hf-W | 4.555 | Schulz et al. 2012 | isochron age | |||
Table 12. Model ages for whole-rock samples and some or all components of the three iron IIE achondrites Colomera, Kodaïkanal, and Weekeroo Station, with the details and literature sources.
| Sample | Method | Reading | Error +/- | Note | Source | Type |
|---|---|---|---|---|---|---|
COLOMERA |
||||||
| ten extractions releasing 25–72% Ar from feldspar sample | Ar-Ar | 4.47 | 0.01 | Bogard, Garrison, and McCoy 2000 | plateau age | |
| all extractions above 25% Ar release from feldspar | Ar-Ar | 4.48 | 0.01 | Bogard, Garrison, and McCoy 2000 | average spectrum age | |
| seven extractions above 75% Ar release from feldspar | Ar-Ar | 4.5 | 0.02 | Bogard, Garrison, and McCoy 2000 | average spectrum age | |
| feldspar sample 400°C | Ar-Ar | 4.451 | 0.017 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 500°C | Ar-Ar | 3.194 | 0.025 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 550°C | Ar-Ar | 3.499 | 0.013 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 600°C | Ar-Ar | 3.852 | 0.007 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 625°C | Ar-Ar | 4.021 | 0.006 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 650°C | Ar-Ar | 4.11 | 0.004 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 675°C | Ar-Ar | 4.142 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 705°C | Ar-Ar | 4.323 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 725°C | Ar-Ar | 4.387 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 750°C | Ar-Ar | 4.403 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 775°C | Ar-Ar | 4.437 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 800°C | Ar-Ar | 4.456 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 815°C | Ar-Ar | 4.464 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 830°C | Ar-Ar | 4.465 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 850°C | Ar-Ar | 4.465 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 870°C | Ar-Ar | 4.469 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 890°C | Ar-Ar | 4.473 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 915°C | Ar-Ar | 4.479 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 940°C | Ar-Ar | 4.48 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 965°C | Ar-Ar | 4.473 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1000°C | Ar-Ar | 4.465 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1050°C | Ar-Ar | 4.468 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1100°C | Ar-Ar | 4.482 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1125°C | Ar-Ar | 4.488 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1150°C | Ar-Ar | 4.499 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1175°C | Ar-Ar | 4.501 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1200°C | Ar-Ar | 4.507 | 0.002 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1250°C | Ar-Ar | 4.51 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1300°C | Ar-Ar | 4.507 | 0.001 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1350°C | Ar-Ar | 4.485 | 0.009 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1450°C | Ar-Ar | 4.552 | 0.015 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| 1550°C | Ar-Ar | 5.164 | 0.052 | Bogard, Garrison, and McCoy 2000 | step extraction age | |
| five analyses of one sample | Rb-Sr | 4.7 | Burnett and Wasserburg 1967 | model age | ||
| six analyses of another sample | Rb-Sr | 3.8 | Burnett and Wasserburg 1967 | model age | ||
| four analyses of another sample | Rb-Sr | 3.1 | Burnett and Wasserburg 1967 | model age | ||
KODAÏKANAL |
||||||
| two samples | K-Ar | 3.53 | 0.1 | Bogard et al 1967b | average model age | |
| glass (Me943G) | K-Ar | 2.47 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (P1159-04) | K-Ar | 3.09 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (P1159-04) | K-Ar | 3.38 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (Me574B) | K-Ar | 3.32 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (Me574B) | K-Ar | 3.27 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| feldspar (Me943-1) | K-Ar | 3.59 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| mixed phases (Me574A) | Rb-Sr | 3.83 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (Me574B) | Rb-Sr | 3.74 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (P1159-04) | Rb-Sr | 3.8 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (P1159-04) split A | Rb-Sr | 3.82 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (P1159-04) split B | Rb-Sr | 3.97 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (Me943G) | Rb-Sr | 3.76 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
| glass (Me943G) | Rb-Sr | 3.69 | 0.1 | Bogard, Burnett, and Wasserburg 1969 | model age | |
WEEKEROO STATION |
||||||
| average of seven determinations on four fractions | K-Ar | 4.49 | 0.1 | Bogard et al. 1967b | model age | |
| Weekeroo H (oligoclase) p<2.96, 105-177μ | K-Ar | 4.05 | Bogard et al. 1967b | model age | ||
| K-Ar | 4.52 | Bogard et al. 1967b | model age | |||
| K-Ar | 4.56 | Bogard et al. 1967b | model age | |||
| Weekeroo H (oligoclase) p<2.96, 62-50μ | K-Ar | 4.54 | Bogard et al. 1967b | model age | ||
| Weekeroo N-B2 <150μ | K-Ar | 4.37 | Bogard et al. 1967b | model age | ||
| K-Ar | 4.48 | Bogard et al. 1967b | model age | |||
| Weekeroo HC <150μ | K-Ar | 4.36 | Bogard et al. 1967b | model age | ||
| K-Ar | 4.51 | Bogard et al. 1967b | model age | |||
| eight extractions | K-Ar | 4.54 | 0.03 | Niemeyer 1980 | model age | |
| K-Ar | 4.3 | 0.1 | Bogard, Garrison, and McCoy 2000 | model age | ||
| eight extractions | Ar-Ar | 4.54 | 0.03 | Niemeyer 1980 | plateau age | |
| Ar-Ar | 4.49 | 0.03 | Bogard, Garrison, and McCoy 2000 | plateau age | ||
| I-Xe | 4.555 | Brazzle et al 1999; Niemeyer 1980 | model age | |||
| Re-Os | 7.1 | Niemeyer and Esser 1991; Snyder et al. 2001 | model age | |||
The Primitive Achondrites—Acapulco and Caddo County IAB Iron
The Pb-Pb and U-Pb isochron ages for Acapulco all agree on a 4.55–4.57 Ga age for this meteorite, with matching support from Mn-Cr and I-Xe isochron ages (fig. 13). One Sm-Nd isochron age is older, and one Ar-Ar isochron age is younger. Similarly, nine Ar-Ar model ages are younger by a similar amount from the one Pb-Pb model 4.55–4.57 Ga age (fig. 14). However, one Ar-Ar-model age agrees with that one Pb-Pb model age, while another Ar-Ar model age is substantially older. Additionally, two Pu-Xe model ages are younger than that one Pb-Pb model 4.55– 4.57 Ga age, while two U-Th/He model ages straddle either side of it.
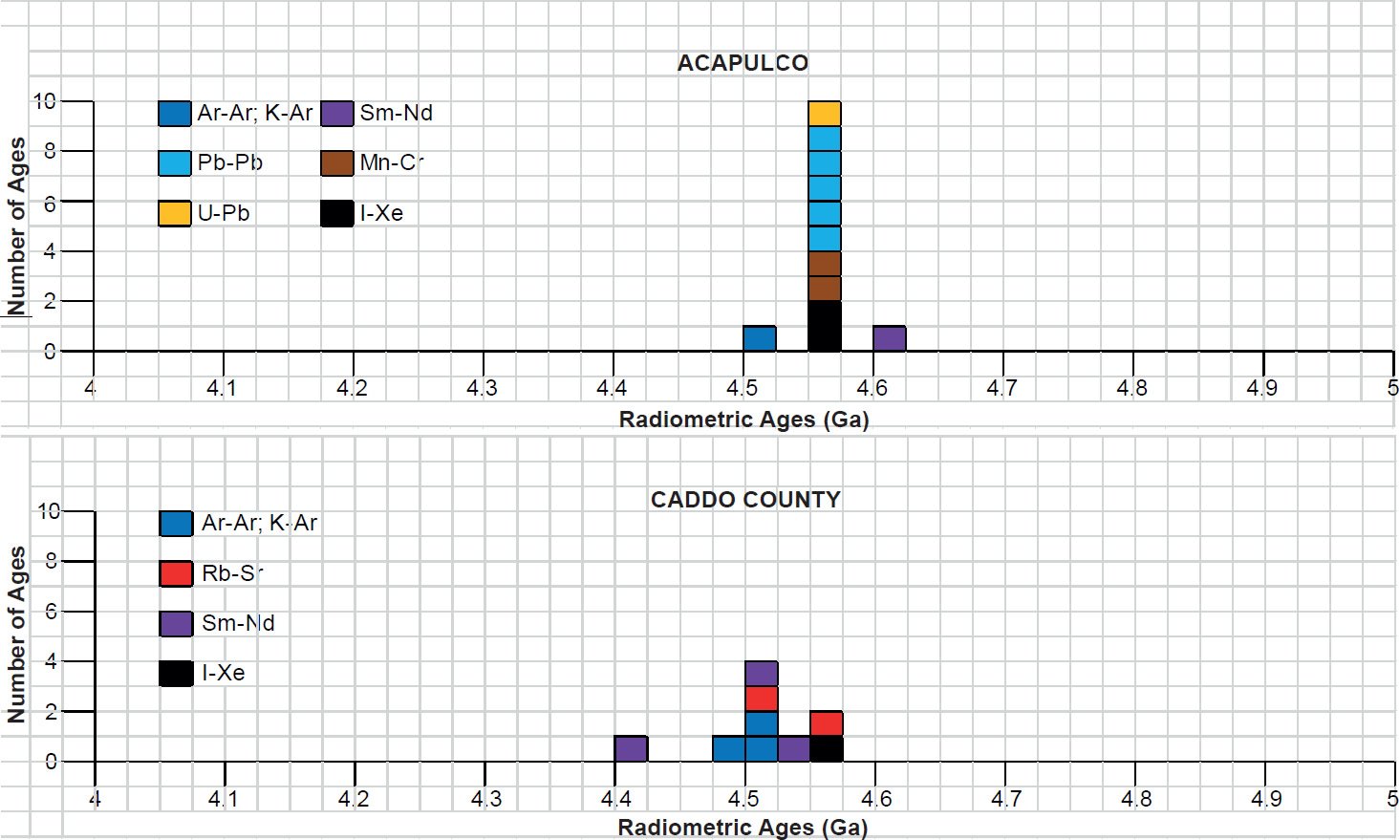
Fig. 13. Frequency versus radioisotope ages histogram diagram for the isochron ages for whole-rock samples and some or all components of the two primitive achondrites Acapulco and Caddo County IAB iron, with color coding being used to show the ages obtained by the different radioisotope dating methods.
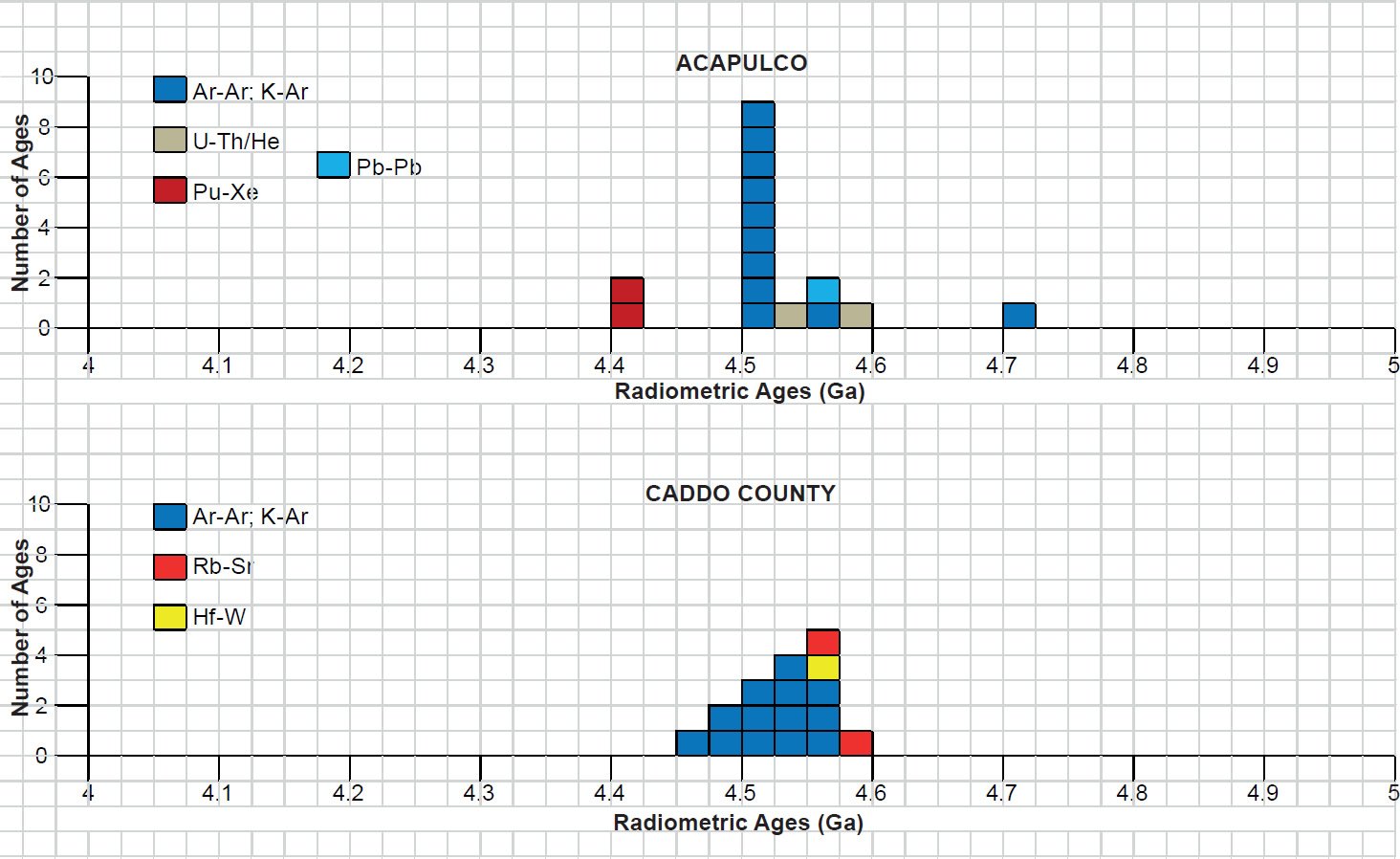
Fig. 14. Frequency versus radioisotope ages histogram diagram for the model ages for whole-rock samples and some or all components of the two primitive achondrites Acapulco and Caddo County IAB iron, with color coding being used to show the ages obtained by the different radioisotope dating methods.
The silicates in the Caddo County IAB iron meteorite yield a main grouping of two Ar-Ar, one Rb-Sr and one Sm-Nd isochron ages at a 4.50–4.525 Ga age, slightly lower than the minor grouping of one Rb-Sr and one I-Xe isochron ages at 4.55–4.57 Ga (fig. 13). Two other Sm-Nd and one Ar-Ar younger isochron ages are scattered below these groupings. On the other hand, the main grouping of model ages is at 4.55–4.57 Ga and consists of three Ar-Ar, one Rb-Sr and one Hf-W model ages (fig. 14). One Rb-Sr model age is older, but ten scattered Ar-Ar model ages are younger.
The Angrites—Angra dos Reis and D’Orbigny
Both of these angrites are strongly dated at 4.55–4.57 Ga, particularly by Pb-Pb isochron and model ages, but supported by U-Pb, Sm-Nd, Rb-Sr, and Hf-W isochron ages and U-Pb model ages (Angra dos Reis), and U-Pb, Rb-Sr, Lu-Hf, Sm-Nd, Mn-Cr, Hf-W, and Al-Mg isochron ages and Al-Mg model ages (D’Orbigny) (figs. 15 and 16). There is minimal scatter of isochron ages, with just one younger Rb-Sr isochron age for Angra dos Reis, and one older Sm-Nd isochron age and one very much younger Sm-Nd isochron age for D’Orbigny (fig. 15). There are older U-Pb and Th-Pb model ages scattered above the strong 4.55–4.57 Ga age peak for Angra dos Reis, and younger Pb-Pb, U-Pb, and Th-Pb model ages scattered below it (fig. 16). On the other hand, there are numerous U-Pb model ages widely scattered either side of the strong 4.55–4.57 Ga age peak for D’Orbigny, and just two slightly younger Pb-Pb model ages (fig. 16).
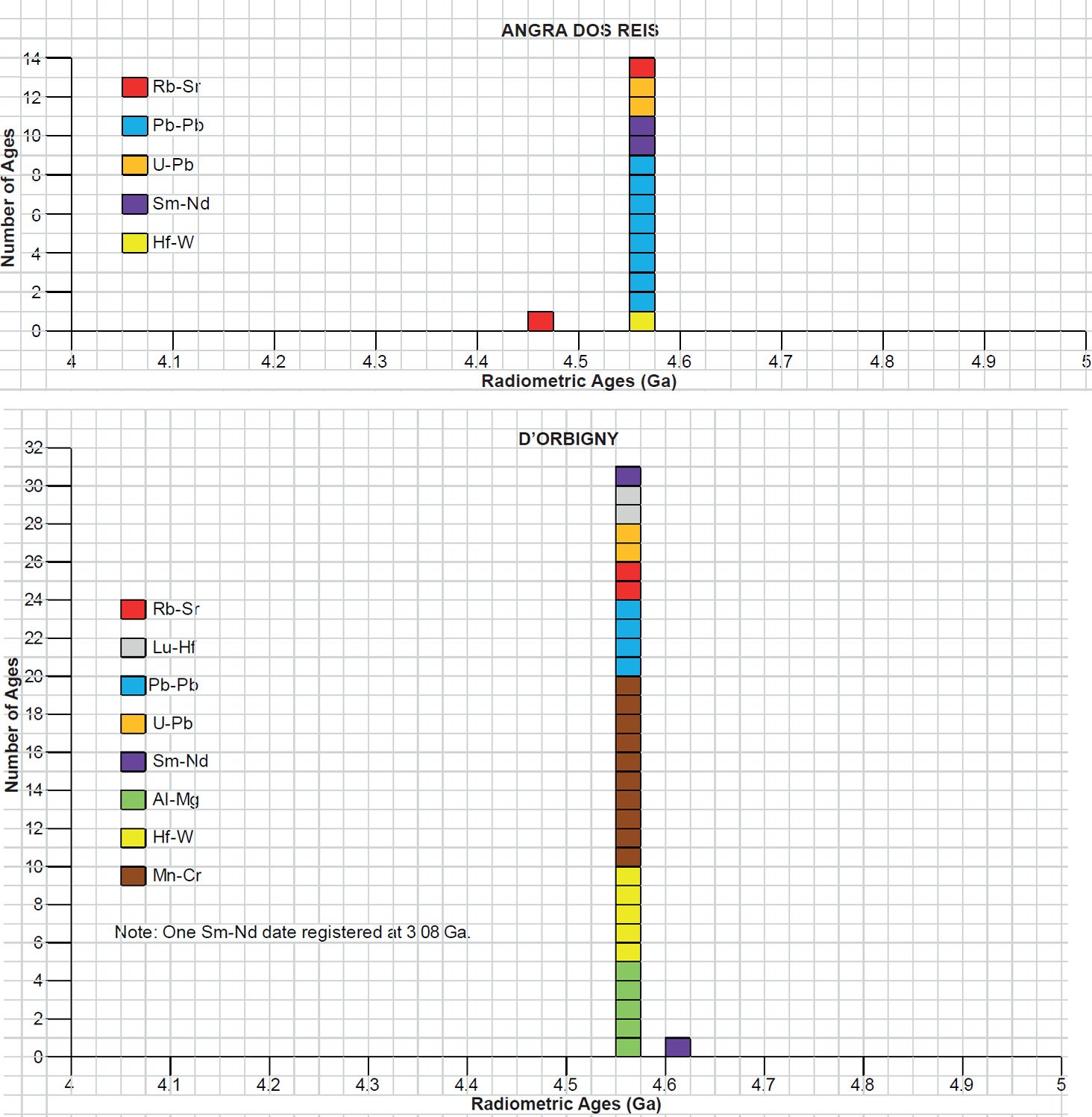
Fig. 15. Frequency versus radioisotope ages histogram diagram for the isochron ages for whole-rock samples and some or all components of the two angrite achondrites Angra dos Reis and D’Orbigny, with color coding being used to show the ages obtained by the different radioisotope dating methods.
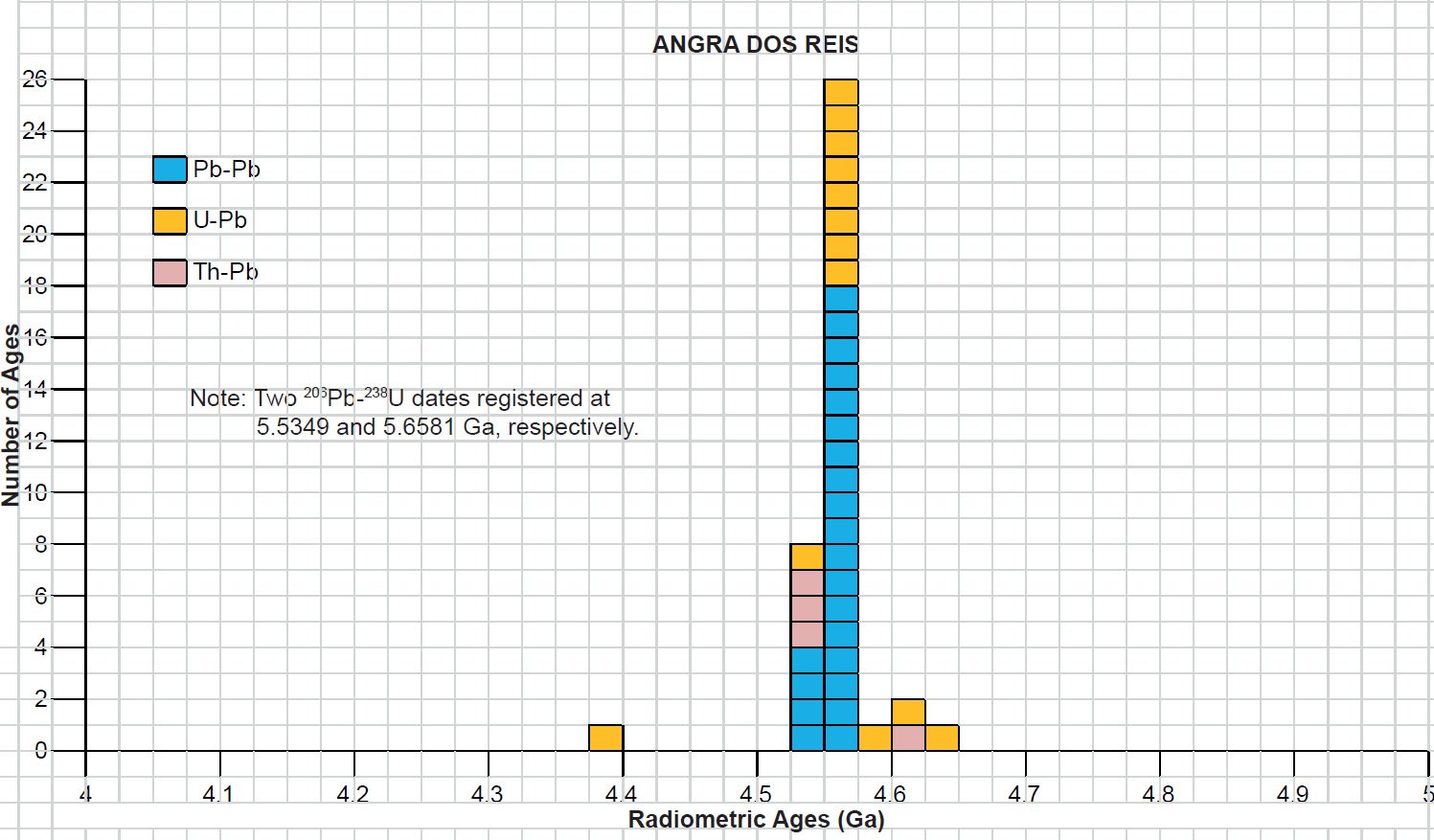
Fig. 16. Frequency versus radioisotope ages histogram diagram for the model ages for whole-rock samples and some or all components of the angrite achondrite Angra dos Reis, with color coding being used to show the ages obtained by the different radioisotope dating methods.
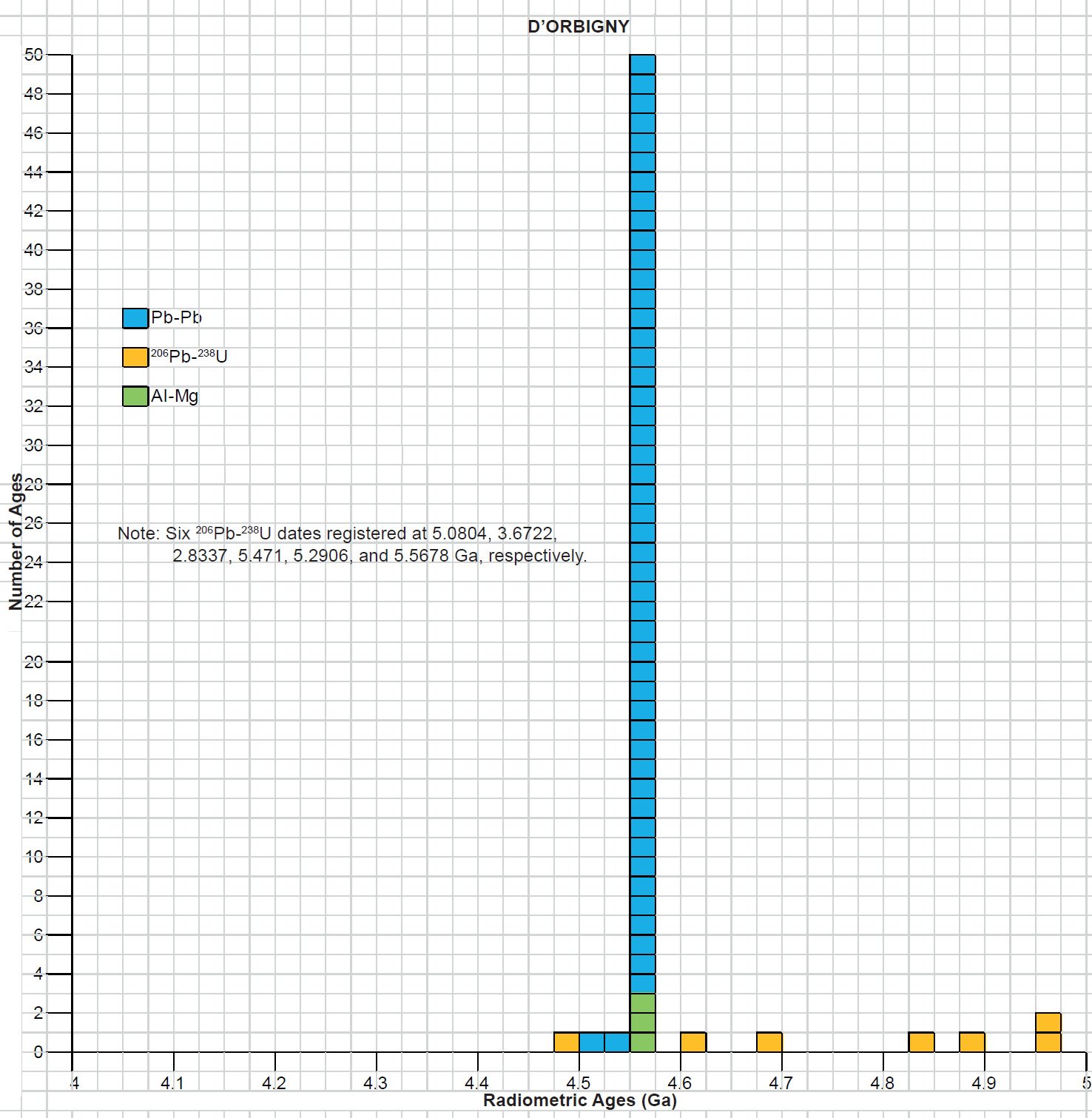
Fig. 16 (cont). Frequency versus radioisotope ages histogram diagram for the model ages for whole-rock samples and some or all components of the angrite achondrite D’Orbigny, with color coding being used to show the ages obtained by the different radioisotope dating methods.
The Aubrites—Norton County and Shallowater
While there is one Pb-Pb isochron age of 4.55 Ga for the Norton County aubrite, there are three younger Rb-Sr isochron ages scattered below it (table 7 and fig. 17). There is only one Ar-Ar isochron age of 4.55 Ga for the Shallowater aubrite (table 7 and fig. 17). Similarly, there is only one Pb-Pb model age of 4.55 Ga for Norton County, yet there are ten scattered K-Ar and Ar-Ar model ages, three older and seven younger (table 8 and fig. 18). On the other hand, there is a close grouping of model ages for Shallowater, four Ar-Ar model ages grouped at 4.53–4.539 Ga and three I-Xe model ages grouped at 4.562–4.566 Ga (table 8 and fig. 18).
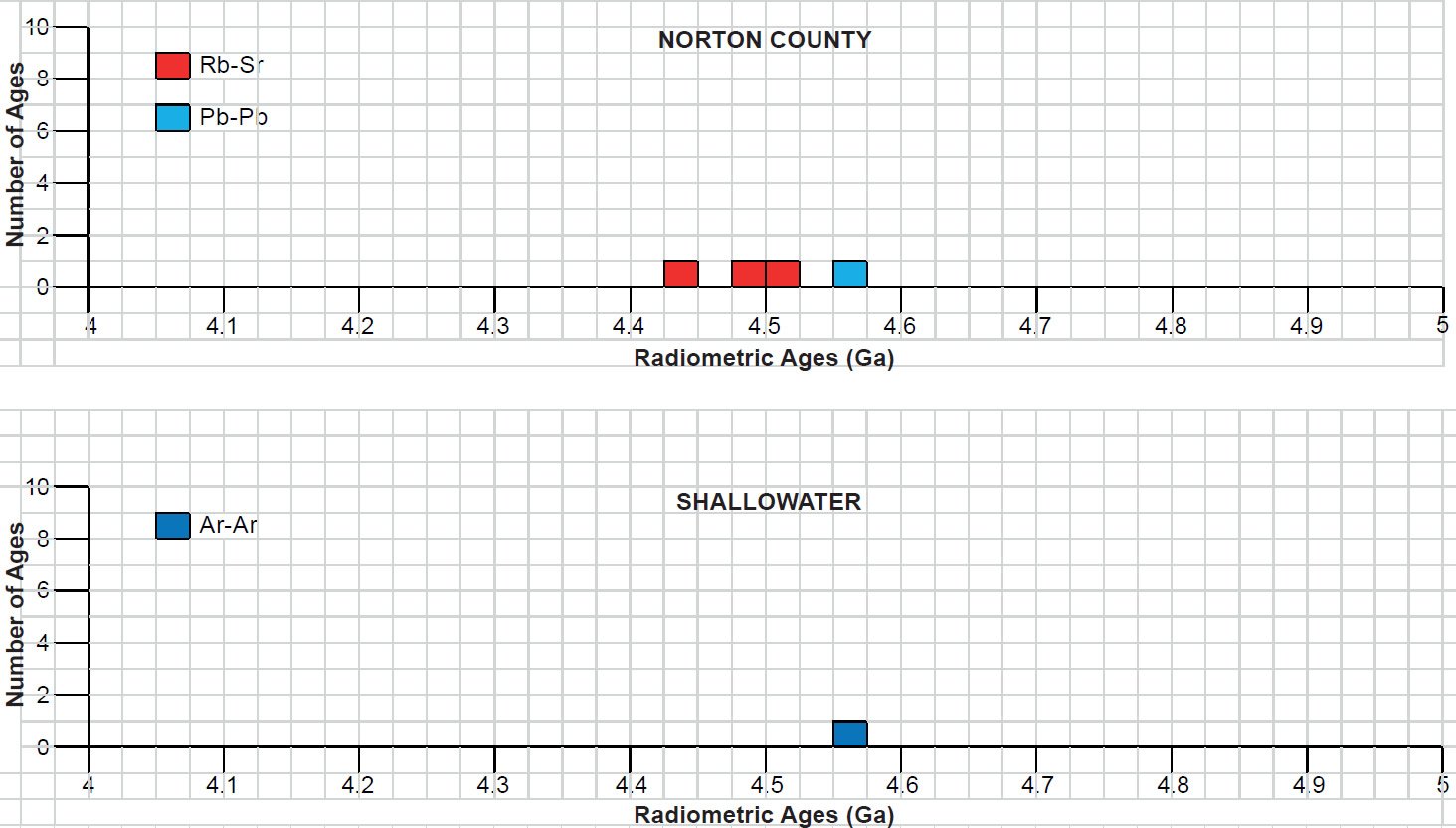
Fig. 17. Frequency versus radioisotope ages histogram diagram for the isochron ages for whole-rock samples and some or all components of the two aubrite achondrites Norton County and Shallowater, with color coding being used to show the ages obtained by the different radioisotope dating methods.
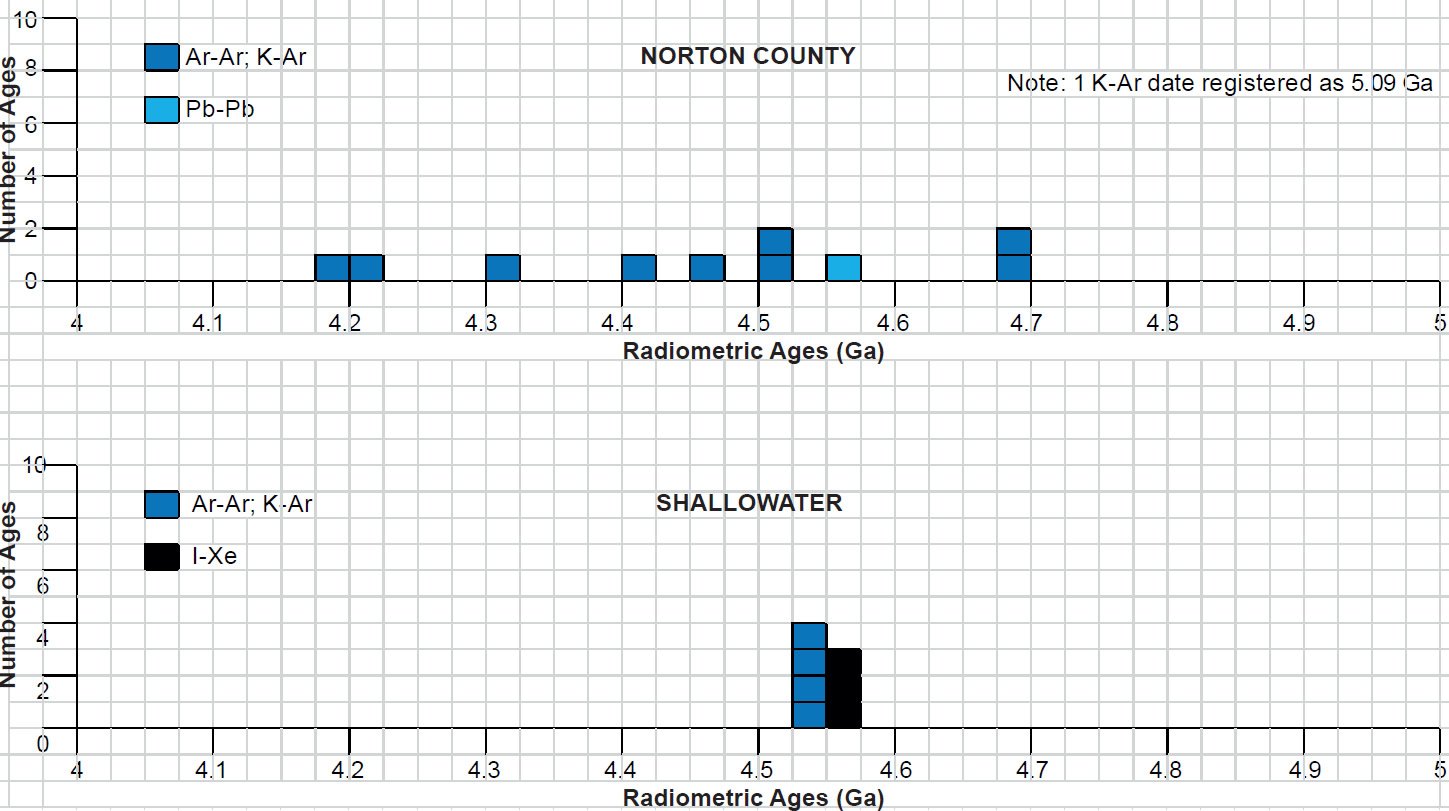
Fig. 18. Frequency versus radioisotope ages histogram diagram for the model ages for whole-rock samples and some or all components of the two aubrite achondrites Norton County and Shallowater, with color coding being used to show the ages obtained by the different radioisotope dating methods.
The Mesosiderites—Estherville
The silicates in the Estherville mesosiderite yield two Pb-Pb isochron ages grouped at 4.55–4.56 Ga, but their Ar-Ar, Rb-Sr, and Sm-Nd isochron ages are younger, the Ar-Ar and some Rb-Sr isochron ages being particularly younger (table 9 and fig. 19). For comparison, the two Re-Os isochron ages yielded by the Fe-Ni metal component of this meteorite are scattered either side of the 4.55–4.56 Ga Pb-Pb isochron ages for the silicates, one being much younger, similar to two much younger Rb-Sr isochron ages. The only two U-Pb model ages are at 4.57 Ga, whereas the only two Ar-Ar model ages are very much younger (table 10 and fig. 20).
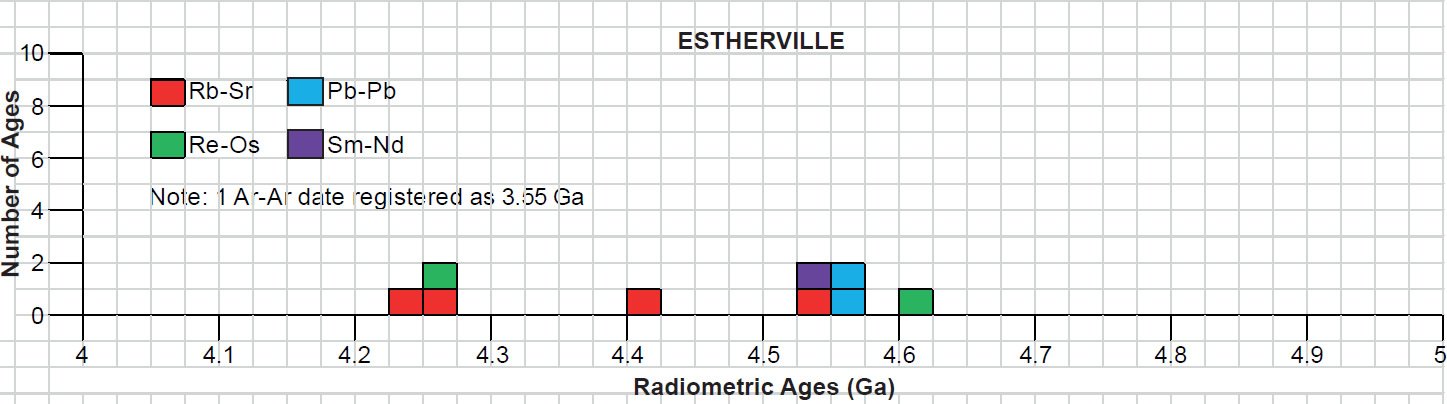
Fig. 19. Frequency versus radioisotope ages histogram diagram for the isochron ages for whole-rock samples and some or all components of the mesosiderite achondrite Estherville, with color coding being used to show the ages obtained by the different radioisotope dating methods.
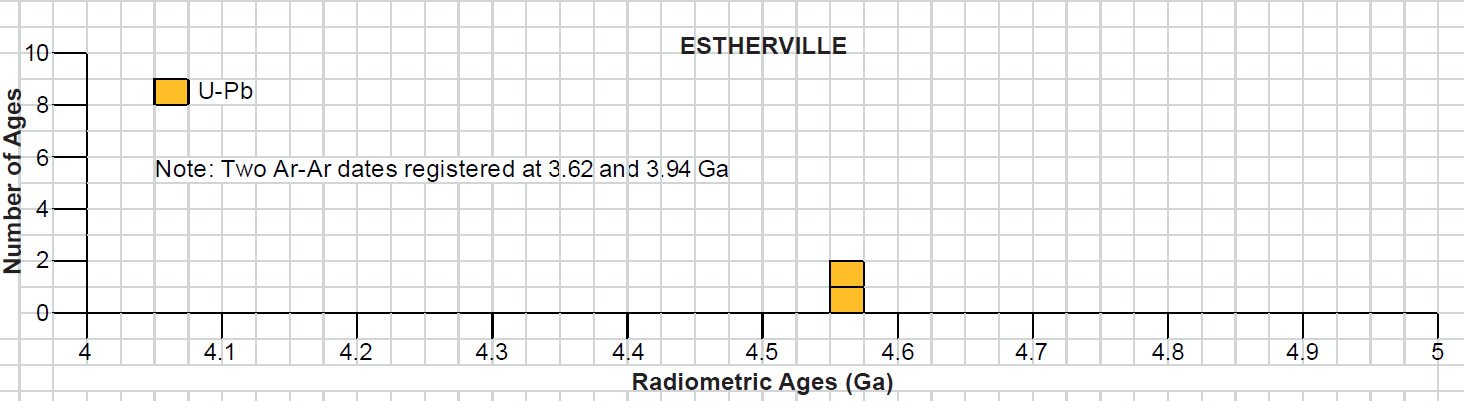
Fig. 20. Frequency versus radioisotope ages histogram diagram for the model ages for whole-rock samples and some or all components of the mesosiderite achondrite Estherville, with color coding being used to show the ages obtained by the different radioisotope dating methods.
The IIE Irons—Colomera, Kodaïkanal and Weekeroo Station
The silicates in the Colomera and Weekeroo Station IIE irons yield a few results at the 4.55–4.57 Ga “target” age indicated by the strong peaks and main groupings of isochron and model ages for many other meteorites (tables 11 and 12). However, four of the five Rb-Sr isochron ages for Colomera are older than the “target” age, and the other is younger (fig. 21). For Weekeroo Station only the sole Hf-W isochron age matches the “target” age, whereas five of the six Rb-Sr isochron ages are much younger, and the other is older, plus the one Sm-Nd isochron age is extremely young (fig. 21). There is one Ar-Ar model age for Colomera that matches the “target” age, whereas many other Ar-Ar model ages are younger, and some very much younger (fig. 22). Additionally, Colomera yields one much older Rb-Sr model age and two very much younger Rb-Sr model ages. Weekeroo Station yields a K-Ar model age that matches the “target” age, supported by an I-Xe model age, whereas apart from one much older K-Ar model age, all its other K-Ar model ages are either younger or much younger (table 12 and fig. 22). Interestingly, the Fe-Ni metal in Weekeroo Station yields a much older Re-Os model age.
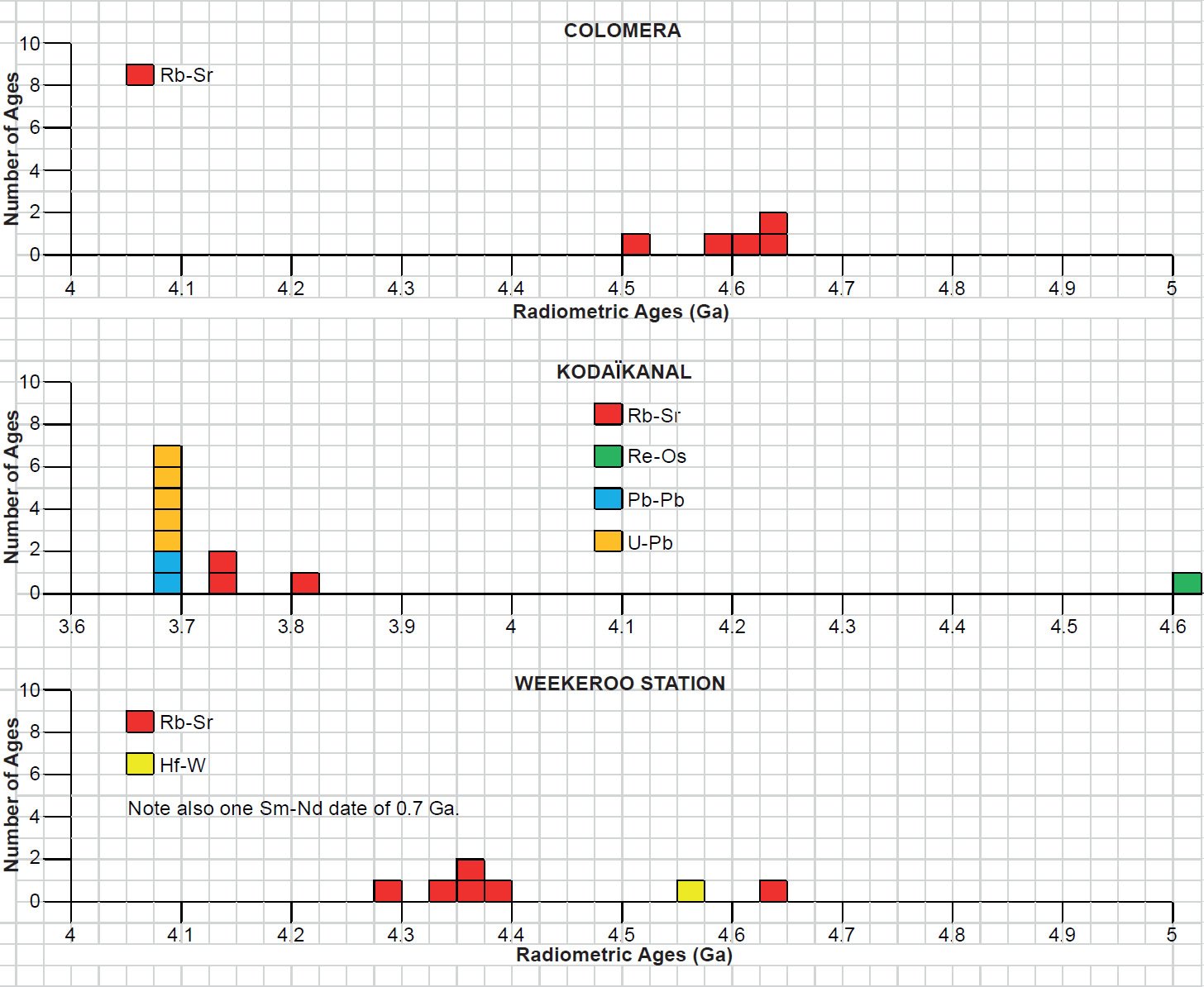
Fig. 21. Frequency versus radioisotope ages histogram diagram for the isochron ages for whole-rock samples and some or all components of the three iron IIE achondrites Colomera, Kodaïkanal, and Weekeroo Station, with color coding being used to show the ages obtained by the different radioisotope dating methods.
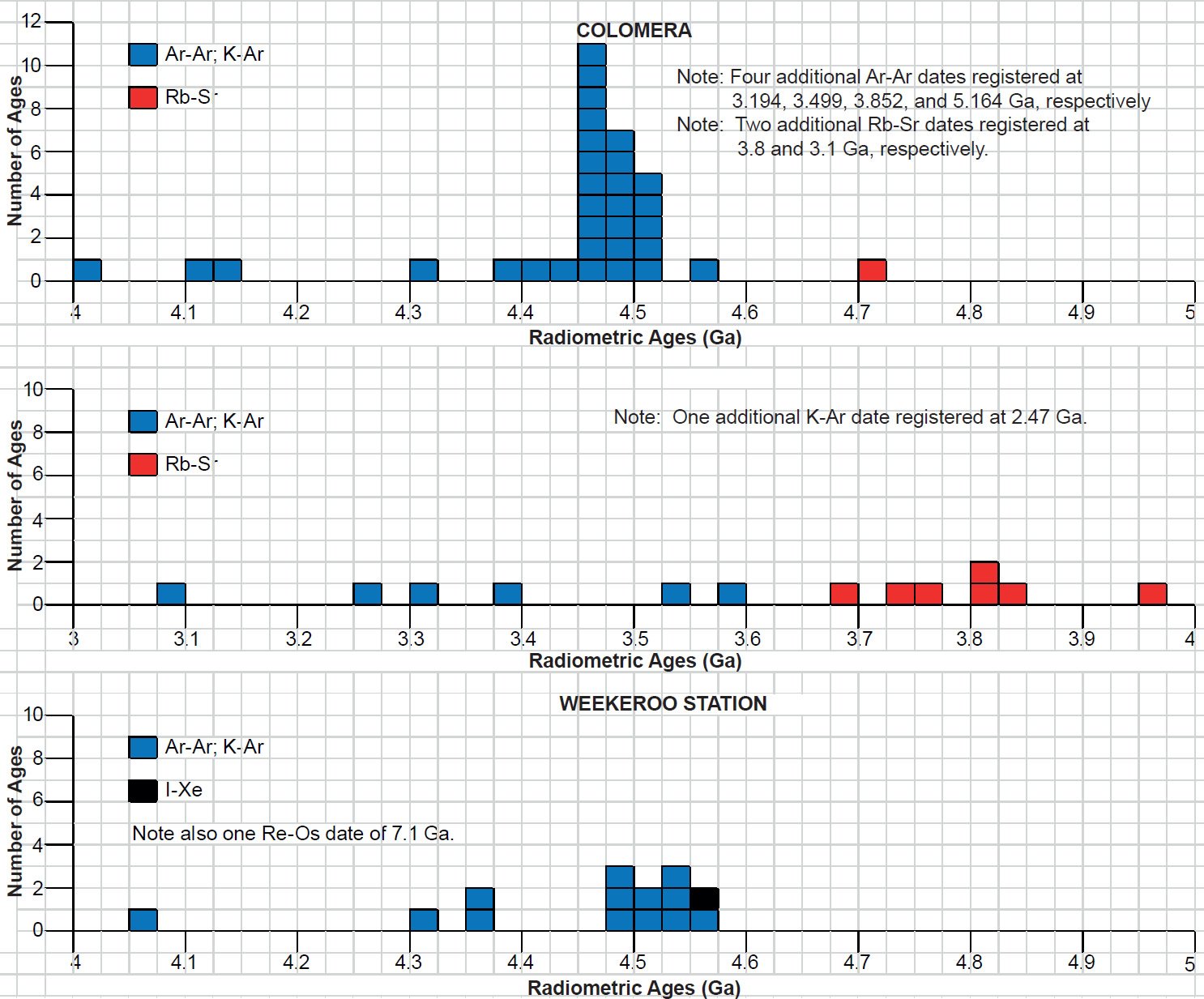
Fig. 22. Frequency versus radioisotope ages histogram diagram for the model ages for whole-rock samples and some or all components of the three iron IIE achondrites Colomera, Kodaïkanal, and Weekeroo Station, with color coding being used to show the ages obtained by the different radioisotope dating methods.
In stark contrast, the silicates in the Kodaïkanal IIE iron meteorite yield both isochron and model ages that indicate the radioisotope systems in its silicates have been greatly disturbed, as all of this meteorite’s isochron and model ages are very much younger than the 4.55–4.57 Ga “target” age (note the adjusted scales of radiometric ages for this meteorite in figs 21 and 22). Interestingly, all the U-Pb and Pb-Pb isochron ages peak at 3.675–3.7 Ga, and the Rb- Sr isochron ages are slightly older, whereas its Fe-Ni metal component yields a sole Re-Os isochron 4.62 Ga age, which might be closer to the “target” age if this isochron age was recalculated using the latest Re decay constant (Birck and Allègre 1998) (table 11 and fig. 21). The K-Ar and Rb-Sr model ages in Kodaïkanal’s silicates are all scattered between 2.74 Ga and 3.97 Ga, but the Rb-Sr model ages are all older than the K-Ar model ages (table 12 and fig. 22).
Agreement of Ages
As noted previously by Snelling (2014a, b, c) for chondrite and eucrite meteorites, there is generally strong agreement among most of the Pb-Pb isochron and model ages for most of these meteorites in these five different groups for a common 4.55–4.57 Ga age. Occasionally, where used, some U-Pb, Rb-Sr, Ar-Ar, Sm-Nd, Lu-Hf, and maybe Re-Os, isochron ages, and some U-Pb, Rb-Sr, Ar-Ar, and K-Ar model ages match this common 4.55–4.57 Ga age. This is to be expected, since the Rb, Lu, Re, Sm, and K decay constants are all ultimately calibrated against the U-Pb method and thus the U decay constants (Dickin 2005, pp. 70–71; Lugmair, Scheinin, and Marti 1975; Nebel, Scherer, and Mezger 2011; Renne et al. 2010; Selby et al. 2007; Söderlund et al. 2004). This ultimately means that all meteorite ages are dependent on the reliability of determinations of the 238U and 235U decay constants, and the critical 238U/235U ratio. However, such calibrations are dependent on assuming the crucial 238U/235U ratio is constant in all rocks and minerals, but recently significant variations in this ratio have been measured in both terrestrial minerals and meteorites (Brennecka and Wadhwa 2012; Hiess et al. 2012). It also may be significant that the estimated half-life of 238U is 4.468 Ga, which is almost identical to the claimed 4.55–4.57 Ga age of the earth, the asteroids, and the meteorites derived from them.
Additionally, whenever I-Xe, Hf-W, Mn-Cr, and Al-Mg isochron ages, and I-Xe, Hf-W, and Al-Mg model ages were determined they always matched the “target” 4.55–4.57 Ga age defined by the Pb-Pb isochron and model ages. This is because all these methods are calibrated against the 4.55–4.57 Ga Pb-Pb isochron and model ages of other meteorites and their components. The I-Xe method is calibrated against the 4.557 Ga Pb-Pb model age of phosphate separates from the Acapulco meteorite and normalized against the I-Xe determinations of the Shallowater aubrite (Bogard Garrison, and Takeda 2005; Brazzle et al. 1999; Göpel, Manhes, and Allègre 1994). The Hf-W method is calibrated by an Hf-W isochron obtained on the H5 chondrite St. Marguerite being calibrated against the Pb-Pb model ages of whole-rock fragments and phosphates from St. Marguerite (Göpel, Manhes, and Allègre 1994; Kleine et al. 2002, 2004, 2005; Markowski et al. 2006), and also by calibrating the Hf-W isochron obtained on the angrite D’Orbigny against its Pb-Pb isochron age corrected for U isotopic variations (Amelin 2008; Brennecka and Wadhwa 2011; Kleine et al. 2012). The Mn-Cr method is calibrated by an Mn-Cr isochron obtained on the H5 chondrite St. Marguerite being calibrated its Pb-Pb model age and the Pb-Pb isochron age of the angrite LEW 86010, or calibrated by the Mn-Cr isochrons of measured meteorites being calibrated against the Mn-Cr isochron for the angrite LEW 86010, which is in turn calibrated against its Pb-Pb isochron age (Göpel, Manhes, and Allègre 1994; Lugmair and Galer 1992; Lugmair and Shukolyukov 1998; Polnau and Lugmair 2001). The Al-Mg method is calibrated against the Al-Mg age of the Ca-Al inclusions (CAIs) in the CV chondrite Efremovka E60 calibrated against their Pb-Pb isochron age or is calibrated by the Al-Mg isochron for the angrite D’Orbigny calibrated against either the Pb-Pb isochron age of the CAIs in the Allende CV chondrite or the Pb-Pb isochron age of D’Orbigny (Amelin et al. 2002; Amelin 2008; Jacobsen et al. 2008; Schiller, Baker, and Bizzarro 2010; Spivak-Birndorf, Wadhwa, and Janney 2009).
Scattering of Ages
To explain the scattering of ages either side of the 4.55–4.57 Ga “target” age appeal is usually made to the effects of thermal disturbance of the radioisotope systems, either during the supposed cooling of the parent asteroid body from its formation in the aftermath of the supposed solar nebula, or during subsequent fragmentation and reassembly of the parent asteroid, and/or as a result of impact cratering of the parent asteroids (Bogard 2011; Bogard, Dixon, and Garrison 2010; Bogard et al. 1990, 1993; Bogard and Garrison 1998; Bogard, Garrison, and McCoy 2000; Bogard, Garrison, and Takeda 2005; Lewis 1997). This must be especially the case where among the meteorites studied here the scattering of ages is common in the K-Ar, Ar-Ar, I-Xe, Pu-Xe, Rb-Sr, Sm-Nd, Re-Os, U-Th/He, and U-Th-Pb systems. Ar and He being gases are readily mobile within and between minerals, and metamorphism is known to reset all these radioisotope systems (Faure and Mensing 2005; Snelling 2000).
Thermal disturbances tend to reset the radioisotope systems so that the resultant ages are usually lower than their original ages (Lewis 1997), which is very evident for the Kodaïkanal iron IIE meteorite. On the other hand, for many of these meteorites the radioisotope ages are both younger and older than the believed formation age of the parent bodies, determined as 4.55–4.57 Ga from the clustering of all the radioisotope systems at that age. Birck and Allègre (1998) noted that the older Re-Os age for the Kodaïkanal iron IIE meteorite could be reconciled by simply adjusting the 187Re decay constant to thus force the older Re-Os age to agree with the expected 4.55–4.57 Ga age. Indeed, Snelling (2014d, e, 2015) has documented how decay constants have repeatedly been adjusted so as to bring Rb-Sr, Lu-Hf, and Re-Os ages into agreement with Pb-Pb and U-Pb ages, including for meteorites. Thus some of the scattering of ages of these meteorites could be due to the decay constants used to calculate the respective ages. Nevertheless, many of the much older ages in the U-Th-Pb isotopic system, and perhaps even the few in the K-Ar, Ar-Ar, and Rb-Sr isotopic systems, could be due to parent radioisotope migration out of, or daughter isotope migration into, minerals and meteorites during thermal disturbances.
A Biblical Perspective
Any postulated history for the formation of the parent asteroids of these meteorites, and of course for the solar system itself, is completely invalidated by the divinely provided biblical account of the six normal days of God creating during the Creation Week. On Day One God ex nihilo created the earth (Genesis 1:1), and only on Day Four did He make the sun and the moon to provide light on the earth during the day and night respectively (Genesis 1:14–16). We are not specifically told that the rest of the solar system was also created on Day Four, but He did make the stars also on that day, and lights were placed in the expanse of the heavens to be for signs and seasons. From this description it is not unreasonable to conclude that the rest of the solar system was made on Day Four, including the asteroids. Furthermore, each entity God created and made during these six normal days of the Creation Week was formed exceedingly rapidly within the time and space of each normal day, so by the end of Day Four asteroids were completely formed entities, with iron cores, ultramafic mantles, and basaltic crusts, all the necessary supposed silicate-metal fractionation and crust-mantle differentiation happening exceedingly rapidly within hours, and thus not requiring the millions of years postulated by uniformitarians.
Given the general consensus that the asteroids consist of residual material from the formation of the solar system, Snelling (2014a, b, c) proposed that the accepted coincident 4.55–4.57 Ga ages for the earth and many meteorites could be due to the earth and the parent asteroids having been created by God from the same primordial material, which He had created on Day One, as already proposed by Faulkner (1999, 2013). His proposal is based on the usages in Genesis 1 of the Hebrew words āśâ (meaning to do or to make) and bārā’ (meaning to create). Because it is indisputably evident that āśâ is commonly used to refer to the act of fashioning something out of already-existing material (for example, the creation of man in Genesis 1:26; cf. 2:7), Faulkner (2013) suggests that, apart from any contextual clues to suggest that it must bear the sense of creation out of nothing, there is a distinct possibility that the making of the astronomical bodies was instead a matter of fashioning them from material previously created on Day One. Just as the description of the earth in Genesis 1:2 is of something unfinished that God returned to over the next several days to shape and prepare, perhaps the matter that would become the astronomical bodies was created on Day One but was shaped on Day Four, whereupon God brought forth their light to the earth.
Thus the simplest unifying assumption would therefore be that all such primordial material may have had the same created isotopic endowment. This assumption seems to be borne out by the earth apparently having the same time-integrated Pb isotopic endowment and thus being the same Pb-Pb “age” as the meteorites plotted on the geochron (Patterson 1956). The earth’s current Pb isotopic endowment was represented on that geochron by the Pb isotopic composition of a modern oceanic sediment sample, which would appear to contain the time-integrated Pb isotopic endowment from the earth’s beginning which was then processed through the earth’s subsequent rock cycle (Tyler 1990). However, while the possibility that the created initial ratios of parent to daughter elements were different for the earth compared to those created for other solar system objects could be considered, that possibility seems unwarranted if God made all the solar system objects (planets, moons, and asteroids) from the same primordial material He had created on Day One, which is consistent with them all having a common Designer. Nevertheless, if there were created differences in the initial isotopic ratios, then it would not have been possible to plot the meteorites and the earth on the same Pb-Pb geochron, or meteorites on the same Pb-Pb, U-Pb, Rb-Sr, Sm-Nd, Lu-Hf, and Re-Os isochrons (Snelling in prep.).
It would also seem reasonable to propose that God created some of all the isotopes of each element at the beginning in the primordial material, including those isotopes that subsequently also formed by radioisotope decay as daughter isotopes from parent isotopes, regardless of when radioisotope decay started. In other words, when God made the primordial material He included in it 206Pb, 207Pb, and 208Pb atoms along with 238U, 235U, and 232Th atoms. It is reasonable to posit that He did, given that when He created the “primordial material” it likely had to have some initial isotopic ratios. Whether it will be possible to develop a model for initial isotope ratios that will explain the current data must be the goal of future research. In any case, initial ratios in the primordial material need not be required to be in secular equilibrium, because these initial ratios are solely the ratios in the daughter Pb isotopes, without any of the other isotopes in the 238U, 235U, and 232Th decay “chains” being relevant. Indeed, even the conventional scientific community has assumed the initial material of the solar system had the “primeval” Pb isotopic ratios as measured in the troilite (iron sulfide) in the Canyon Diablo iron meteorite, without reference to any of the intermediate daughters (Faure and Mensing 2005). This is consistent with God creating a fully-functioning universe, as typified by Him creating fruit trees already bearing fruit in fully-functioning soil on land, all during Day Three, and the sun, moon, asteroids and stars fully-functioning in their ordained positions and roles on Day Four. Thus when by the end of Day Four the asteroids had been formed, all the supposed silicate-metal fractionation and crust-mantle differentiation of that primordial material necessary to produce their internal layering had happened exceedingly rapidly within hours. That silicate-metal fractionation and crust-mantle differentiation may have also resulted in some redistribution or mixing of parent and daughter atoms, the latter having been originally created rather than derived via radioactive decay. These processes may also explain some of the scattering in the radioisotope ages for these meteorites, especially those older than the 4.55–4.57 Ga clustering.
At what point in time radioactive decay began is unclear from Scripture, and is still a matter of debate among creationists. The RATE project considered the possibility of a large amount of accelerated decay occurring during the Creation Week, as radioisotope decay was not regarded as decay in the sense of deterioration of matter (Vardiman, Snelling, and Chaffin 2005). It is instead a transmutation process, by which one element is changed into another. The daughter element is certainly not inferior to the parent element. However, it is the radiation given off which is harmful that causes concern as to whether the radioisotope decay processes meet the standard of God’s declaration of His completed creation being “very good” (Genesis 1:31).
In the context of the decay evident today due to the operation of the second law of thermodynamics, Anderson (2103) contended that there is no real biblical evidence to suggest that the second law was inoperable prior to the curse, and so argued that rather the second law was in effect from the beginning of creation. He thus also suggested that the tendency toward entropy implicit in the second law was never of a kind that conflicted with God’s declaration that the creation was “very good,” or that eventuated in the death of any sentient creature. On the other hand, it could be argued that radioisotope decay is more than the operation of the second law of thermodynamics; the additional outcome being the radiation produced which is harmful to life’s biological and chemical makeup. Indeed, if billions of years of accelerated radioisotope decay had occurred mainly during the early part of the Creation Week, as considered a possibility by the RATE team (Vardiman, Snelling, and Chaffin 2005), the enormous burst of radiation would surely have been detrimental to all life on the earth, for example, the plants of Day Three. It is for this reason that many creationists are not comfortable with postulating that accelerated radioisotope decay happened during the Creation Week, so maybe there was no radioisotope decay at all until it was started as part of the curse.
The lack of any evidence of a pattern of isochron ages in these ten primitive and other achondrite meteorites, as well as in the 28 chondrite and basaltic achondrite (eucrite) meteorites previously studied (Snelling 2014a, b, c), that matches the pattern found during the RATE project (Snelling 2005c; Vardiman, Snelling, and Chaffin 2005) would therefore strongly suggest that all these meteorites and their parent asteroids have not experienced any episode of accelerated radioisotope decay, either at the time of the creation of the primordial material on Day One or of their formation on Day Four, or since. This could then be taken to infer that no accelerated radioisotope decay occurred anywhere in the solar system during the Creation Week, including on the earth during Days One–Three. Such a conclusion is based on the assumption that the mechanism of small changes to the binding forces in the nuclei of the parent radioisotopes proposed as the cause of a past episode of accelerated radioisotope decay (Vardiman, Snelling, and Chaffin 2005) would thus have affected every atom making up the earth, and by logical extension every atom of the universe at the same time, because God appears to have created the physical laws governing the universe to operate consistently through time and space.
On the other hand, if this assumption is true, and it is consistent with God’s work of providentially maintaining the universe after He created it, then why is there evidence of an episode of accelerated radioisotope decay in the earth, but not in these parent asteroids and their meteorite fragments? The answer would seem to be that the accelerated radioisotope decay only occurred during the catastrophic global Flood event on the earth and that the parent asteroids were not similarly affected. However, if the earth’s atoms were affected by accelerated radioisotope decay during the Flood, then surely every other atom in the universe would have been similarly affected. However, God is not bound by the physical laws He put in place at creation, as He can change them at any time anywhere or everywhere. After all, when Jesus Christ the Creator locally suspended the law of gravity as He walked on the stormy waters of the Sea of Galilee, the law of gravity was still operating at the same time to keep the disciples in their boat, their boat on the water and the earth in space in orbit around the sun. Thus God could have made small changes to the binding forces of the nuclei of only the earth’s atoms during the Flood to cause accelerated radioisotope decay only on the earth, while leaving the atoms making up the rest of the solar system and universe untouched. Perhaps the reason God initiated accelerated radioisotope decay only on the earth was to generate the heat necessary to initiate and drive the catastrophic plate tectonics which reshaped the earth’s surface during the Flood (Baumgardner 2003).
Another reason for arguing that accelerated radioisotope decay occurred in earth’s rocks only during the Flood is that the earth’s rocks contain the physical evidence of only 500–600 million years’ worth of radioisotope decay (as calculated using today’s measured decay rates), which equates to the same time period postulated by uniformitarians during which the geologic record of the Flood accumulated (Snelling 2009). This physical evidence of radioisotope decay in earth’s rocks is provided by radiohalos and fission tracks (Snelling 2005a, b). Each fully-formed U radiohalo, no matter where in the geologic record it occurs or the supposed age of its host rock, only represents up to 100 million years’ worth of U decay, so even if it is in a Precambrian (pre- Flood or Creation Week) granite it still only records up to 100 million or so years’ worth of U decay that occurred during the Flood, at the same time as new granites containing U radiohalos were forming in plutons which had intruded into fossil-bearing (and therefore Flood-deposited) sedimentary strata. On the other hand, up to 600 or so million years’ worth of fission tracks are found in zircon grains matching the radioisotope ages of those same zircon grains at the base of the strata record of the Flood (Snelling 2005b).
This still does not fully explain why there is such a spread of radioisotope “ages” from 4.03 Ga to the present in the earth’s rocks, or why the radioisotope “ages” of the oldest earth rock (the Acasta Gneiss, Canada) (Bowring, Williams, and Compston 1989; Stern and Bleeker 1998) and the oldest mineral in an earth rock (a zircon grain in the Jack Hills sandstone, Western Australia) (Valley et al. 2014; Wilde at al. 2001) are 4.03 Ga and 4.4 Ga respectively rather than 4.56 Ga, the supposed age of the earth. The answer might be that the earth’s rocks, subsequent to their creation on Day One, first suffered from the processes of mixing of isotopes and resetting of radioisotope “clocks” in the mantle and crust during the Day Three “Great Upheaval” when God formed the dry land, and then further suffered from mixing of isotopes and resetting of radioisotope “clocks” in the mantle and crust as a consequence of the catastrophic plate tectonics during the Flood, as well as the concurrent accelerated radioisotope decay during the Flood. These mixing processes would not just have affected isochron ages in that the mixing and adding or removing of parent and/ or daughter isotopes produces mixing lines that are then interpreted as isochrons, but the same mixing, adding and/or subtracting of parent and/or daughter isotopes would have reset the radioisotope model ages. In these ways the radioisotope “clocks” would have been reset during both the “Great Upheaval” and the Flood, just as they are when pre-Flood crustal rocks (with older radioisotope ages) were melted to form granite magmas, which when they crystallized had their radioisotope “clocks” reset to record the younger granite formation ages.
By comparison, the Day Three “Great Upheaval” which the Scriptures describe as occurring on the earth could not have affected other bodies in the solar system, including the asteroids which were the source of the meteorites in this study, because these other bodies were not created until Day Four. Thus we can be dogmatic that this event was earth-specific, as it was designed to produce the continental crust and the dry land on the earth in readiness for the subsequent creation of plants, birds, animals, and man. In any case, so far our observations of the surfaces of asteroids do not indicate any continental crust on them akin to that which formed on the earth on Day Three. However, when they were formed on Day Four their formation could have included incredibly rapid silicate-metal fractionation and mantle-crust differentiation with accompanying redistribution of the previously created parent and (what uniformitarians now interpret as) daughter isotopes.
The scatter in the radioisotope ages of the 38 meteorites studied thus far, the ten in this study and the 28 in the previous studies, would then be due to processes subsequent to the initial creation of the parent asteroids from the primordial material on Day Four of the Creation Week which have reset the radioisotope “clocks” at various times. Such processes would include the initial fractionation and differentiation, heating due to impact cratering, impact disintegration, and re-coalescing of the asteroids, space weathering, and heating on passage of the meteorites through the earth’s atmosphere (Bogard and Garrison 2009; Cloutis, Binzel, and Gaffey 2014; Michel 2014; Norton 2002, ).
Where to From Here?
As concluded by Snelling (2014a, b, c) from his studies of 16 chondrite meteorites and 12 basaltic achondrites (eucrites), based on the assumptions made the 4.55–4.57 Ga radioisotope “ages” for the Acapulco, Caddo County, Angra dos Reis, D’Orbigny, Norton County, Shallowater, Estherville, and Weekeroo Station achondrites obtained primarily by Pb-Pb and U-Pb radioisotope isochron and model age dating of various constituent minerals and fractions, but also by radioisotope methods directly calibrated against Pb-Pb meteorite ages (for example, Amelin 2007, 2008; Amelin and Pravdivtseva 2005; Brazzle et al. 1999; Brouxel and Tatsumoto 1990, 1991; Gilmour et al. 2006; Huey and Kohman 1973; Kleine et al. 2012; Markowski et al. 2006; Schulz et al. 2012; Zhou et al. 2012) are likely not their true real-time ages. The assumptions on which the radioisotope dating methods are based are simply unprovable, and in the light of the possibility of an inherited primordial geochemical signature, subsequent resetting of radioisotope “clocks” due to impact cratering of asteroids, and the evidence in earth rocks for past accelerated radioisotope decay, mixing of isotopes and resetting of radioisotope “clocks,” these assumptions are unreasonable.
However, we still need to develop a coherent and comprehensive explanation of what these radioisotope compositions in both meteorites and earth rocks really represent and mean within our biblical young-age creation-Flood framework for earth and universe history. We have some possible clues already, as discussed here, and a clearer picture may yet emerge from continued investigations now in progress. Examination of the radioisotope dating data for groups of meteorites is necessary to complete our understanding of what the meteorite radioisotope “ages” might mean. If only the earth was affected by accelerated radioisotope decay during the Flood, then it is also necessary to examine the radioisotope dating data for martian meteorites, and lunar meteorites and rocks to determine whether Mars and the Moon were affected by that aspect of the Flood catastrophe or not. Additionally, the radioisotope dating data for many more earth rocks from all levels of the geologic record need to be collated and examined. If accelerated radioisotope decay only occurred during the Flood, then it might be expected that the radioisotope “ages” of pre-Flood (mostly Precambrian) strata determined by the different methods would be noticeably discordant (Snelling 2005c), whereas the radioisotope “ages” of the strata formed during the Flood (mostly Phanerozoic) would be mostly concordant (Snelling 2005b). This difference might be expected due to the pre-Flood rocks having already been formed and their radioisotope “clocks” started before the onset of the accelerated radioisotope decay during the Flood, when their radioisotope “clocks” would have been speeded up by different amounts according to the atomic weights of the parent radioisotopes for the full duration of the Flood event. In contrast, the Flood rocks would have had their radioisotopes mixed and then reset when those rocks formed. Thus their radioisotope “clocks” only started at different times during the accelerated radioisotope decay of the Flood event, and consequently Flood rocks experienced less accelerated radioisotope decay than pre-Flood rocks. It may take the collation and examination of the huge radioisotope dating data sets of as many different earth rocks as possible from all levels of the geologic record to enable any firm conclusions to be made.
Whatever the radioisotope dating data for the earth’s rocks may reveal, it is already well-established that there are so many problems with the radioisotope dating methods which render them totally unreliable in providing time markers for the different stages in the earth’s history. Indeed, the investigations of determinations of the decay constants of each of the parent radioisotopes needs to be completed to provide further documentation of the numerous uncertainties in that key time assumption. Therefore, even though most of these primitive and other achondrites yield a consistent Pb-Pb, U-Pb, and Pb-Pb-calibrated isochron and model age of 4.55–4.57 Ga, that cannot be their true real-time age, which according to the biblical paradigm is only about 6000 real-time years.
Conclusions
After six decades of numerous careful radioisotope dating investigations of these achondrite meteorites their Pb-Pb, U-Pb, and Pb-Pb-calibrated isochron and model age of 4.55–4.57 Ga has been well established. This date for these achondrites is supported for most of them by a strong clustering of their Pb-Pb, U-Pb, and Pb-Pb-calibrated isochron and model ages in the 4.55–4.57 Ga range, as well as sometimes being confirmed by both isochron and model age results via the K-Ar, Ar-Ar, Rb-Sr, Lu-Hf, Re-Os, and Sm-Nd methods. The Hf-W, Mn-Cr, Al-Mg, and I-Xe methods are all calibrated against the Pb-Pb method, so their results are not objectively independent. Thus the Pb-Pb dating method stands supreme in the conventional evolutionary uniformitarian community as the ultimate, most precise tool for determining the ages of these achondrite and other meteorites.
There are no discernible patterns in the isochron and model ages for these achondrites, apart from scattering of many K-Ar, Ar-Ar, Rb-Sr, Re-Os, and Sm-Nd isochron ages and a few Pb-Pb and U-Pb isochron ages, and the considerable scattering of many K-Ar, Ar-Ar, and Rb-Sr model ages and some Pb-Pb, U-Pb, and Th-Pb model ages. The radioisotope ages for these achondrites do not follow the systematic pattern found in Precambrian rock units during the RATE project. The α-decay isochron ages are not always older than the β-decay isochron ages for particular achondrites, and among the β-decayers the isochron ages are not always older according to the increasing heaviness of the atomic weights of the parent radioisotopes, or the increasing lengths of their half-lives. Thus there appears to be no consistent evidence in these achondrite meteorites similar to that found in earth rocks of past accelerated radioisotope decay. Therefore it could be concluded from these data that no accelerated radioisotope decay event has occurred on the asteroids which parented these achondrite meteorites.
Any explanation for the 4.55–4.57 Ga age for these achondrite meteorites needs to consider their origin. These meteorites are regarded as fragments of asteroids, debris derived from impact cratering of their surfaces and collisions with other asteroids. Even in the naturalistic paradigm the asteroids, and thus the meteorites, are regarded as “primordial material” left over from the formation of the solar system. Similarly, it has been suggested the Hebrew of the Genesis text allows for God to have made “primordial material” on Day One of the Creation Week from which He made the earth on Day One and the non-earth portion of the solar system on Day Four. Thus today’s measured radioisotope compositions of these achondrites may reflect a geochemical signature of that “primordial material,” which included atoms of all elemental isotopes created by God. Therefore if some, or perhaps most, of the daughter isotopes measured today in these achondrite meteorites were thus “inherited” by them when they were formed from that “primordial material,” and the parent isotopes in these meteorites have only been subjected to some subsequent radioisotope decay (and none at accelerated rates), then the 4.55–4.57 Ga Pb-Pb, U-Pb, and Pb-Pb-calibrated isochron and model “age” for these achondrites cannot be their true real-time age, which according to the biblical paradigm is only about 6000 real-time years.
However, these conclusions and suggested explanations as discussed are still somewhat tentative, their confirmation or adjustment awaiting the examination of more radioisotope dating data for groups of meteorites, and martian and lunar meteorites. Furthermore, further extensive studies of the radioisotope dating of lunar rocks and rocks from all levels of the earth’s geologic record are required to attempt to systematize what proportions of the isotopes in each radioisotope dating system measured today are due to inheritance from the “primordial material,” to accelerated radioisotope decay during the Flood, and mixing, additions and subtractions in the earth’s mantle and crust through earth history, particularly during the Day Three Upheaval and then subsequently during the Flood.
Acknowledgments
The invaluable help of my research assistant in compiling these radioisotope dating data into the tables and then plotting the data in the color coded age versus frequency histogram diagrams is acknowledged. Also, our production assistant Laurel Hemmings is especially thanked for her painstaking work in preparing these papers for publication.
References
Amelin Y., A.N. Krot, I.D. Hutcheon, and A.A. Ulyanov. 2002. Lead isotopic ages of chondrules and calcium-aluminum- rich inclusions. Science 297, no. 5587:1678–1683.
Amelin, Y. 2005. Meteorite phosphates show constant 176Lu decay rate since 4557 million years ago. Science 310, no. 5749:839–841.
Amelin, Y., and O.V. Pravdivtseva. 2005. U-Pb age of the Acapulco phosphate: Testing the calibration of the I-Xe chronometer. Meteoritics and Planetary Science 40, no. 9 (Supplement):A16.
Amelin, Y., M. Wadhwa, and G.W. Lugmair. 2006. Pb- isotopic dating of meteorites using 202Pb-205Pb double spike: Comparison with other high-resolution chronometers. Lunar and Planetary Science Conference 37: #1970.
Amelin, Y. 2007. The ages of angrites. Lunar and Planetary Science Conference 37: #1669.
Amelin, Y. 2008. U-Pb ages of angrites. Geochimica et Cosmochimica Acta 72, no. 1:221–232.
Amelin, Y., A. Kaltenbach, T. Iizuka, C.H. Stirling, T.R. Ireland, M. Petaev, and S.B. Jacobsen. 2010. U-Pb chronology of the Solar System’s oldest solids with variable 238U/235U. Earth and Planetary Science Letters 300, no. 3–4:343–350.
Amelin, Y., J. Wimpenny, and Q.-Z. Yin. 2011. Angrite Lu-Hf whole rock data provide no direct support to accelerated decay of 176Lu by supernova irradiation. Workshop on the Formation of the First Solids in the Solar System #9014.
Anderson, L.A. Jr. 2013. Thoughts on the goodness of creation: In what sense was creation “perfect”? Answers Research Journal 6:391–397. https://answersingenesis.org/physics/thoughts-on-the-goodness-of-creation-in-what-sense-was-creation-perfect/.
Baker, J., M. Bizzarro, N. Wittig, J. Connelly, and H. Haack. 2005. Early planetesimal melting from an age of 4.5662 Gyr for differentiated meteorites. Nature 436:1127–1131.
Baumgardner, J.R. 2003. Catastrophic plate tectonics: The physics behind the Flood. In Proceedings of the Fifth International Conference on Creationism, ed. R.L. Ivey Jr., 113–126. Pittsburgh, Pennsylvania: Creation Science Fellowship.
Benedix, G.K., T.J. McCoy, K. Keil, and S.G. Love. 2000. A petrologic study of the IAB iron meteorites: Constraints on the formation of the IAB-Winonaite parent body. Meteoritics and Planetary Science 35, no. 6:1127–1141.
Birck, J.L., and C.-J. Allègre. 1998. Rhenium-187-osmium-187 in iron meteorites and the strange origin of the Kodaïkanal meteorite. Meteoritics and Planetary Science 33, no. 4:647–653.
Bogard, D.D., D.S. Burnett, P. Eberhardt, and G.J. Wasserburg. 1967a. 87Rb-87Sr isochron and 40K-40Ar ages of the Norton County achondrite. Earth and Planetary Science Letters 3:179–189.
Bogard, D.D., D.S. Burnett, P. Eberhardt, and G.J. Wasserburg. 1967b. 40K-40Ar ages of silicate inclusions in iron meteorites. Earth and Planetary Science Letters 3:275–283.
Bogard, D.D., D.S. Burnett, and G.J. Wasserburg. 1969. Cosmogenic rare gases and the 40K-40Ar age of the Kodaïkanal iron meteorite. Earth and Planetary Science Letters 5:273–281.
Bogard, D.D., D.H. Garrison, J.L. Jordan, and D. Mittlefehldt. 1990. 39Ar-40Ar dating of mesosiderites: Evidence for major parent body disruption <4Ga ago. Geochimica et Cosmochimica Acta 54, no. 9:2549–2564.
Bogard, D.D., D.H. Garrison, T.J. McCoy, and K. Keil. 1993. 39Ar-40Ar ages of acapulcoites and lodranites: Evidence for early parent body heating. Lunar and Planetary Science Conference 24:141–142.
Bogard, D.D., and D.H. Garrison. 1998. 39Ar-40Ar ages and thermal history of mesosiderites. Geochimica et Cosmochimica Acta 62, no. 8:1459–1468.
Bogard, D.D., D.H. Garrison, and T.J. McCoy. 2000. Chronology and petrology of silicates from IIE iron meteorites: Evidence of a complex parent body evolution. Geochimica et Cosmochimica Acta 64, no. 12:2133–2154.
Bogard, D.D., D.H. Garrison, and H. Takeda. 2005. Ar-Ar and I-Xe ages and the thermal history of IAB meteorites. Meteoritics and Planetary Science 40, no. 2:207–224.
Bogard, D.D., and D.H. Garrison. 2009. Ar-Ar impact heating ages of eucrites and timing of the LHB. Lunar and Planetary Science Conference 40: #1131.
Bogard, D.D., E.T. Dixon, and D.H. Garrison. 2010. Ar-Ar ages and thermal histories of enstatite meteorites. Meteoritics and Planetary Science 45, no. 5:723–742.
Bogard, D.D. 2011. K-Ar ages of meteorites: Clues to parent-body thermal histories. Chemie der Erde 71, no. 3:207–226.
Bouvier, A., and M. Wadhwa 2010. The age of the Solar System redefined by the oldest Pb–Pb age of a meteoritic inclusion. Nature Geoscience 3:637–641.
Bowring S.A., I.S. Williams, and W. Compston. 1989. 3.96Ga gneisses from the Slave Province, Northwest-Territories, Canada. Geology 17, no. 2:971–975.
Brazzle, R.H., O.V. Pravdivtseva, A.P. Meshik, and C.M. Hohenberg. 1999. Verification and interpretation of the I-Xe chronometer. Geochimica et Cosmochimica Acta 63, no. 5:739–760.
Brennecka, G.A., M. Wadhwa, P.E. Janney, and A.D. Anbar. 2010. Towards reconciling early Solar System chronometers: The 238U/235U ratios of chondrites and D’Orbigny pyroxenes. Lunar and Planetary Science Conference 41:#2117.
Brennecka, G.A., and M. Wadhwa. 2011. 238U/235U ratios of angrites: Adjusting absolute ages of anchors. Mineralogical Magazine 75, no. 3:579.
Brennecka, G.A., and M. Wadhwa. 2012. Uranium isotope compositions of the basaltic angrite meteorites and the chronological implications for the early Solar System. Proceedings of the National Academy of Sciences USA 109, no. 24:9299–9303.
Brouxel, M., and M. Tatsumoto. 1990. Age of the Estherville mesosiderite. Lunar and Planetary Science Conference 20:117–118.
Brouxel, M., and M. Tatsumoto. 1991. The Estherville mesosiderite: U-Pb, Rb-Sr, and Sm-Nd isotopic study of a polymict breccia. Geochimica et Cosmochimica Acta 55, no. 4:1121–1133.
Bunch, T.E., K. Keil, and E. Olsen. 1970. Mineralogy and petrology of silicate inclusions in iron meteorites. Contributions to Mineralogy and Petrology 25:297–340.
Burnett, D.S., and G.J. Wasserburg. 1967. 87Rb-87Sr ages of silicate inclusions in iron meteorites. Earth and Planetary Science Letters 2, no. 5:397-408.
Casanova, I., K. Keil, and H.E. Newsom. 1993. Composition of metal in aubrites: Constraints on core formation. Geochimica et Cosmochimica Acta 57, no. 3:675–682.
Chen, J.H., and G.J. Wasserburg. 1981. The isotopic composition of uranium and lead in Allende inclusions and meteoritic phosphates. Earth and Planetary Science Letters 52, no. 1:1–15.
Cloutis, E.A., R.P. Binzel, and M.J. Gaffey. 2014. Establishing asteroid-meteorite links. Elements 10, no. 1:25–30.
Cumming, G.L. 1969. A recalculation of the age of the solar system. Canadian Journal of Earth Sciences 6, no. 4:719–735. Dalrymple, G.B. 1991. The age of the earth. Stanford, California: Stanford University Press.
Dalrymple, G.B. 2004. Ancient earth, ancient skies. Stanford, California: Stanford University Press.
Dickin, A.P. 2005. Radiogenic isotope geology. 2nd ed. Cambridge, United Kingdom: Cambridge University Press.
Evensen N.M., P.J. Hamilton, G.E. Harlow, R. Klimentidis, R.K. O’Nions, and M. Prinz. 1979. Silicate inclusions in Weekeroo Station: Planetary differentiates in an iron meteorite. Lunar and Planetary Science Conference 10:376–378.
Faulkner, D.R. 1999. A biblically-based cratering theory. Creation Ex Nihilo Technical Journal 13, no. 1:100–104.
Faulkner, D.R. 2013. A proposal for a new solution to the light travel time problem. Answers Research Journal 6:295–300. https://answersingenesis.org/astronomy/starlight/a-proposal-for-a-new-solution-to-the-light-travel-time-problem/.
Faure, G. and T.M. Mensing. 2005. Isotopes: Principles and applications. 3rd ed. Hoboken, New Jersey: John Wiley & Sons.
Geiss, J., and D.C. Hess. 1958. Argon-potassium ages and the isotopic composition of argon from meteorites. Astrophysical Journal 127:224–236.
Gilmour, J.D., O.V. Pravdivtseva, A. Busfield, and C.M. Hohenberg. 2006. The I-Xe chronometer and the early solar system. Meteoritics and Planetary Science 41, no. 1:19–31.
Gilmour, J.D., S.A. Crouther, A. Busfield, G. Holland, and J.A. Whitby. 2009. An early I-Xe age for CB chondrite chondrule formation, and a re-evaluation of the closure age of Shallowater enstatite. Meteoritics and Planetary Science 44, no. 4:573–579.
Glavin, D.P., E. Jagoutz, and G.W. Lugmair. 2003. Mn- Cr chronology of the D’Orbigny angrite. Meteoritics and Planetary Science 38:A40.
Glavin, D.P., A. Kubny, E. Jagoutz, and G.W. Lugmair. 2004. Mn-Cr isotope systematics of the D’Orbigny angrite. Meteoritics and Planetary Science 39, no. 5:693–700.
Göpel, C., G. Manhès, and C.-J. Allègre. 1985. Concordant 3,676 Myr U-Pb formation age for the Kodaikanal iron meteorite. Nature 317, no. 6035:341–344.
Göpel, C., G. Manhès, and C.-J. Allègre. 1992. U-Pb study of the Acapulco meteorite. Meteoritics 27, no. 3:226.
Göpel, C., G. Manhès, and C.-J. Allègre. 1994. U-Pb systematics of phosphates from equilibrated ordinary chondrites. Earth and Planetary Science Letters 121, no. 1–2:153–171.
Grossman, J.N., and J. Zipfel. 2001. The Meteoritical Bulletin, no. 85, 2001 September. Meteoritics and Planetary Science 36:A293–A322.
Hans, U., T. Kleine, and B. Bourdon. 2010. Rb-Sr chronology of volatile depletion of the angrite and eucrite parent bodies. Lunar and Planetary Science Conference 41:#2680.
Hiess, J., D.J. Condon, N. McLean, and S.R. Noble. 2012. 238U/235U systematics in terrestrial uranium-bearing minerals. Science 335, no. 6076:1610–1614.
Hohenberg, C.M., and O.V. Pravdivtseva. 2008. I-Xe dating: From adolescence to maturity. Chemie der Erde 68, no. 4:339–351.
Huey, J.M. and T.P. Kohman. 1973. 207Pb-206Pb isochron and age of chondrites. Journal of Geophysical Research 78, no. 17:3227–3244.
Jacobsen, S.B. and G.J. Wasserburg. 1984. Sm-Nd isotopic evolution of chondrites and achondrites, II. Earth and Planetary Science Letters 67, no. 2:137–150.
Jacobsen, B., Q.-Z. Yin, F. Moynier, Y. Amelin, A.N. Krot, K. Nagashima, I.D. Hutcheon, and H. Palme. 2008. 26Al-26Mg and 207Pb-206Pb systematics of Allende CAIs: Canonical solar initial 26Al/27Al ratio reinstated. Earth and Planetary Science Letters 272, no. 1-2:353–364.
Jagoutz, E., R. Jotter, M.E. Varela, R. Zartman, G. Kurat, and G.W. Lugmair. 2002. Pb-U-Th isotopic evolution of the D’Orbigny angrite. Lunar and Planetary Science Conference 33:#1043.
Jagoutz, E., R. Jotter, M.E. Varela, R. Zartman, G. Kurat, and G.W. Lugmair. 2003. Cm?-U-Th-Pb isotopic evolution of the D’Orbigny angrite. Meteoritics and Planetary Science 38:A81.
Keil, K., T. Ntaflos, G.J. Taylor, A.J. Brearley, H.E. Newsom, and A.D. Romig Jr. 1989. The Shallowater aubrite: Evidence for origin by planetesimal impacts. Geochimica et Cosmochimica Acta 53, no. 12:3291–3307.
Keil, K. 2010. Enstatite achondrite meteorites (aubrites) and the histories of their asteroidal parent bodies. Chemie der Erde 70, no. 40:295–317.
Kirsten, T., D. Krankowsky, and J. Zähringer. 1963. Edelgas- und kalium-bestimmungen an einer grösseren zahl von steinmeteoriten. Geochimica et Cosmochimica Acta 27, no. 1:13–42.
Kleine, T., C. Münker, K. Mezger, and H. Palme. 2002. Rapid accretion and early core formation on asteroids and the terrestrial planets from Hf-W chronometry. Nature 418:952–955.
Kleine, T., K. Mezger, C. Münker, H. Palme, and A. Bischoff. 2004. 182Hf–182W isotope systematics of chondrites, eucrites, and martian meteorites: Chronology of core formation and early mantle differentiation in Vesta and Mars. Geochimica et Cosmochimica Acta 68, no. 13:2935–2946.
Kleine, T., K. Mezger, H. Palme, E. Scherer, and C. Münker. 2005. Early core formation in asteroids and late accretion of chondrite parent bodies: Evidence from 182Hf–182Win CAIs, metal-rich chondrites and iron meteorites. Geochimica et Cosmochimica Acta 69, no. 24:5805–5818.
Kleine, T., U. Hans, A.J. Irving, and B. Bourdon. 2012. Chronology of the angrite parent body and implications for core formation in protoplanets. Geochimica et Cosmochimica Acta 84, no. 1:186–203.
Krot, A.N., K. Keil, C.A. Goodrich, E.R.D. Scott, and M.K. Weisberg. 2005. Classification of meteorites. In Meteorites, comets, and planets, ed. A.M. Davis. Treatise on Geochemistry, vol.1, pp.83–128. Amsterdam, Netherlands: Elsevier.
Kruijer, T.S., T. Kleine, M. Fischer-Gödde, and C. Burkhardt. 2014. Nucleosynthetic W isotope anomalies and the Hf-W chronometry of Ca-Al-rich inclusions. Earth and Planetary Science Letters 403:317–327.
Kurat, G., M.E. Varela, F. Brandstätter, G. Weckwerth, R.N. Clayton, H.W. Weber, L. Schultz, E. Wäsch, and M.A. Nazarov. 2004. D’Orbigny: A non-igneous angritic achondrite? Geochimica et Cosmochimica Acta 68:1901–1921.
Lewis, J.S. 1997. Physics and chemistry of the solar system. Rev. ed. London, England: Academic Press.
Liu Y.Z., L.E. Nyquist, H. Wiesmann, Y. Reese, C.-Y. Shih, and H. Takeda. 2002a. Rb-Sr and Sm-Nd ages of plagioclase- diopside-rich material in Caddo County IAB iron meteorite. Lunar and Planetary Science Conference 33:#1389.
Liu Y.Z., L.E. Nyquist, H. Wiesmann, C.-Y. Shih, and H. Takeda. 2002b. The age of plagioclase-diopside inclusions in the Caddo County meteorite: Rb-Sr and Al-Mg isochrons. Meteoritics and Planetary Science 37:A89.
Luck, J.-M., and C.-J. Allègre. 1983. 187Re-187Os systematics in meteorites and cosmological consequences. Nature 302:130–132.
Lugmair, G.W., N.B. Scheinin, and K. Marti. 1975. Search of extinct 146Sm, 1. The isotopic abundance of 142Nd in the Juvinas meteorite. Earth and Planetary Science Letters 27:79–84.
Lugmair, G.W., and K. Marti. 1977. Sm-Nd-Pu timepieces in the Angra dos Reis meteorite. Earth and Planetary Science Letters 35, no. 2:273–284.
Lugmair, G.W., and S.J.G. Galer. 1992. Age and isotopic relationships among the angrites Lewis Cliff 86010 and Angra dos Reis. Geochimica et Cosmochimica Acta 56, no. 4:1673–1694.
Lugmair, G.W., and A. Shukolyukov. 1998. Early solar system timescales according to 53Mn-53Cr systematics. Geochimica et Cosmochimica Acta 62, no. 16:2863–2886.
Markowski, A., G. Quitté, A.N. Halliday, and T. Kleine. 2006. Tungsten isotopic composition of iron meteorites: Chronological constraints vs. cosmogenic effects. Earth and Planetary Science Letters 242, no. 1–2:1–15.
Markowski, A., G., Quitté, T. Kleine, A.N. Halliday, M. Bizzarro, and A.J. Irving. 2007. Hafnium–tungsten chronometry of angrites and the earliest evolution of planetary objects. Earth and Planetary Science Letters 262, no. 1–2:214–229.
McCoy, T.J., K. Keil, D.D. Bogard, D.H. Garrison, I. Casanova, M.M. Lindstrom, A.J. Brearley, K. Kehm, R.H. Nichols Jr., and C.M Hohenberg. 1995. Origin and history of impact- melt rocks of enstatite chondrite parentage. Geochimica et Cosmochimica Acta 59, no. 1:161–175.
McCoy. T.J., K. Keil, R.N. Clayton, T.K. Mayeda, D.D. Bogard, D.H. Garrison, G.R. Huss, I.D. Hutcheon, and R. Wieler. 1996. A petrologic, chemical, and isotopic study of Monument Draw and comparison with other acapulcoites: Evidence for formation by incipient partial melting. Geochimica et Cosmochimica Acta 60, no. 14:2681–2708.
Michel, P. 2014. Formation and physical properties of asteroids. Elements 10, no. 1:19–24.
Mikouchi, T., and G. McKay. 2001. Mineralogical investigation of D’Orbigny: A new angrite showing close affinities to Asuka 881371, Sahara 99555 and Lewis Cliffs 87051. Lunar and Planetary Science Conference 32:#1876.
Min, K., K.A. Farley, P.R. Renne, and K. Marti. 2003. Single grain (U-Th)/He ages from phosphates in Acapulco meteorite and implications for thermal history. Earth and Planetary Science Letters 209, no. 3–4:323–336.
Minster, J.F., and C.-J. Allègre. 1976. 87Rb-87Sr history of the Norton County enstatite achondrite. Earth and Planetary Science Letters 32, no. 2:191–198.
Minster, J.F., L.-P. Rickard, and C.-J. Allègre. 1979. 87Rb-87Sr chronology of enstatite meteorites. Earth and Planetary Science Letters 44, no. 3:420–440.
Mittlefehldt, D.W. 1990. Petrogenesis of mesosiderites: I. Origin of mafic lithologies and comparison with basaltic achondrites. Geochimica et Cosmochimica Acta 54, no. 4:1165–1173.
Mittlefehldt, D.W. 2005. Achondrites. In Meteorites, comets, and planets, ed. A.M. Davis. Treatise on Geochemistry, vol.1, pp.291–324. Amsterdam, Netherlands: Elsevier.
Morris, J.D. 2007. The young earth. Rev. ed., pp.59–61. Green Forest, Arkansas: Master Books.
Murthy, V.R., M.R. Coscio, Jr., and T. Sabelin. 1977. Proceedings of the Eighth Lunar Science Conference, 177–186.
Murthy, V.R., E.C. Alexander Jr., and K. Saito. 1978. Rb-Sr and 40Ar-39Ar systematics of the Estherville mesosiderite. Lunar and Planetary Science Conference 9:781–783.
Nebel, O., E.E. Scherer, and K. Mezger. 2011. Evaluation of the 87Rb decay constant by age comparison against the U-Pb system. Earth and Planetary Science Letters 301, no. 1-2:1–8.
Niemeyer, S. 1980. I-Xe and 40Ar-39Ar dating of silicate from Weekeroo Station and Netschaëvo IIE iron meteorites. Geochimica et Cosmochimica Acta 44, no. 1:33–44.
Niemeyer, S., and B.K. Esser. 1991. Re-Os chronology of IAB, IIE, and IIIAB iron meteorites. Meteoritics 26:378-379.
Norton, O.R. 2002. The Cambridge encyclopedia of meteorites. Cambridge, United Kingdom: Cambridge University Press.
Nyquist, L.E., C.Y. Shih, H. Wiesmann, and T. Mikouchi. 2003. Fossil 26Al and 53Mn in D’Orbigny and Sahara 99555 and the timescale for angrite magmatism. Lunar and Planetary Science Conference 34:#1388.
Palme, H., L. Schultz, B. Spettel, H.W. Weber, H. Wänke, M. Christophe Michel-Levy, and J.C. Lorin. 1981. The Acapulco meteorite: chemistry, mineralogy and irradiation effects. Geochimica et Cosmochimica Acta 45, no. 5:727–752.
Patterson, C. 1956. Age of meteorites and the earth. Geochimica et Cosmochimica Acta 10:230–237.
Pellas, P., C. Fiéni, M. Trieloff, and E.K. Jessberger. 1997. The cooling history of the Acapulco meteorite as recorded by the 244Pu and 40Ar-39Ar chronometers. Geochimica et Cosmochimica Acta 61, no. 16:3477–3501.
Polnau, E., and G.W. Lugmair. 2001. Mn-Cr isotope systematics in the two ordinary chondrites Richardton (H5) and Ste. Marguerite (H4). Lunar and Planetary Science Conference 32:1527.
Powell, B.N. 1971. Petrology and chemistry of mesosiderites—II. Silicate textures and compositions and metal-silicate relationships. Geochimica et Cosmochimica Acta 35, no. 1:5–34.
Prinzhofer, A., D.A. Papanastassiou, and G.J. Wasserburg. 1992. Samarium-neodymium evolution of meteorites. Geochimica et Cosmochimica Acta 56, no. 2:797–815.
Renne, P.R. 2000. 40Ar/39Ar age of plagioclase from Acapulco meteorite and the problem of systematic errors in cosmochronology. Earth and Planetary Science Letters 175, no. 1–2:13–26.
Renne, P.R., R. Mundil, G. Balco, K. Min, and K.R. Ludwig. 2010. Joint determination of 40K decay constants and 40Ar*/40K for the Fish Canyon sanidine standard, and improved accuracy for 40Ar/39Ar geochronology. Geochimica et Cosmochimica Acta 74, no. 18:5349–5367.
Sanborn, M.E., R.W. Carlson, and M. Wadhwa. 2011. 147,146Sm-143,142Nd and 87Rb-87Sr systematics of the angrites Northwest Africa 4590, Northwest Africa 4801, and D’Orbigny. Lunar and Planetary Science Conference 42: #2369.
Sanborn, M.E., R.W. Carlson, and M. Wadhwa. 2012. Internal Lu-Hf isochrons for the quenched and plutonic angrites and their chronological implications. Lunar and Planetary Science Conference 43:#2039.
Sanz, H.G., D.S. Burnett, and G.J. Wasserburg. 1970. A precise 87Rb/87Sr age and initial 87Sr/86Sr for the Colomera iron meteorite. Geochimica et Cosmochimica Acta 34, no. 11:1227–1239.
Schiller, M., J.A. Baker, and M. Bizzarro. 2010. 26Al-26Mg dating of asteroidal magmatism in the young Solar System. Geochimica et Cosmochimica Acta 74, no. 16:4844–4864.
Schulz, T., D. Upadhyay, C. Münker, and K. Mezger. 2012. Formation and exposure history of non-magmatic iron meteorites and winonaites: Clues from Sm and W isotopes. Geochimica et Cosmochimica Acta 85:200–212.
Scott, E.R.D., and J.T. Wasson. 1975. Classification and properties of iron meteorites. Reviews of Geophysics and Space Physics 13, no. 4:527–546.
Selby, D., R.A. Creaser, H.J. Stein, R.J. Markey, and J.L. Hannah. 2007. Assessment of the 187Re decay constant by cross calibration of Re-Os molybdenite and U-Pb chronometers in magmatic ore systems. Geochimica et Cosmochimica Acta 71, no. 8:1999–2013.
Shen, J.J., D.A. Papanastassiou, and G.J. Wasserburg. 1998. Re-Os systematics in pallasite and mesosiderite metal. Geochimica et Cosmochimica Acta 62, no. 15:2715–2723.
Shukolyukov, A., and G.W. Lugmair. 2007. The Mn-Cr isotope systematics of bulk angrites. Lunar and Planetary Science Conference 38: #1423.
Söderlund, U., P. J. Patchett, J. D. Vervoort, and C. E. Isachsen. 2004. The 176Lu decay constant determined by Lu-Hf and U-Pb isotope systematics of Precambrian mafic intrusions. Earth and Planetary Science Letters 219, no. 3–4:311–324.
Snelling, A.A. 2000. Geochemical processes in the mantle and crust. In Radioisotopes and the age of the earth: A young- earth creationist research initiative, ed. L. Vardiman, A.A. Snelling, and E.F. Chaffin, 123–304. El Cajon, California: Institute for Creation Research, and St. Joseph, Missouri: Creation Research Society. http://www.icr.org/rate/.
Snelling, A.A. 2005a. Radiohalos in granites: Evidence of accelerated nuclear decay. In Radioisotopes and the age of the earth: Results of a young-earth creationist research initiative, ed. L. Vardiman, A.A. Snelling, and E.F. Chaffin, 101–207. El Cajon, California: Institute for Creation Research, and Chino Valley, Arizona: Creation Research Society. http://www.icr.org/article/radiohalos-granites-evidence-for-accelerated/.
Snelling, A.A. 2005b. Fission tracks in zircons: Evidence for abundant nuclear decay. In Radioisotopes and the age of the earth: Results of a young-earth creationist research initiative, ed. L. Vardiman, A.A. Snelling, and E.F. Chaffin, 209–324. El Cajon, California: Institute for Creation Research, and Chino Valley, Arizona: Creation Research Society. http://www.icr.org/article/fission-tracks-zircons-evidence-for/.
Snelling, A.A. 2005c. Isochron discordances and the role of inheritance and mixing of radioisotopes in the mantle and crust. In Radioisotopes and the age of the earth: Results of a young-earth creationist research initiative, ed. L. Vardiman, A.A. Snelling, and E.F. Chaffin, 393–524. El Cajon, California: Institute for Creation Research, and Chino Valley, Arizona: Creation Research Society. http://www.icr.org/article/isochron-discordances-role-inheritance/.
Snelling, A.A. 2009. Earth’s catastrophic past: Geology, creation and the Flood. Green Forest, Arkansas: Master Books, and Petersburg, Kentucky: Answers in Genesis.
Snelling, A.A. 2014a. Radioisotope dating of meteorites: I. The Allende CV3 carbonaceous chondrite. Answers Research Journal 7:103–145. https://answersingenesis.org/astronomy/age-of-the-universe/radioisotope-dating-meteorites-i-allende-cv3-carbonaceous-chrondrite/.
Snelling, A.A. 2014b. Radioisotope dating of meteorites: II. The ordinary and enstatite chondrites. Answers Research Journal 7:239–296. https://answersingenesis.org/astronomy/age-of-the-universe/radioisotope-dating-meteorites-ii-ordinary-and-enstatite-chondrites/.
Snelling, A.A. 2014c. Radioisotope dating of meteorites: III. The eucrites (basaltic achondrites). Answers Research Journal 7:533–585. https://answersingenesis.org/astronomy/age-of-the-universe/radioisotope-dating-meteorites-iii/.
Snelling, A.A. 2014d. Determination of the radioisotope decay constants and half-lives: Rubidium-87 (87Rb). Answers Research Journal 7:311–322. https://answersingenesis.org/geology/radiometric-dating/determination-radioisotope-decay-constants-and-half-lives-rubidium-87-87rb/.
Snelling, A.A. 2014e. Determination of the radioisotope decay constants and half-lives: Lutetium-176 (176Lu). Answers Research Journal 7:483–497. https://answersingenesis.org/geology/radiometric-dating/determination-radioisotope-decay-constants-and-half-lives-lutetium-176/.
Snelling, A.A. 2015. Determination of the radioisotope decay constants and half-lives: Rhenium-187 (187Re). Answers Research Journal 8:93–109. https://answersingenesis.org/geology/radiometric-dating/determination-radioisotope-decay-constants-and-half-lives-rhenium-187/.
Snelling, A.A. n.d. Radioisotope dating of meteorites. V: Isochron ages of groups of meteorites. Answers Research Journal (in preparation).
Snyder, G.A., D.-C. Lee, A.M. Ruzicka, M. Prinz, L.A. Taylor, and A.N. Halliday. 2001. Hf-W, Sm-Nd, and Rb-Sr isotopic evidence of late impact fractionation and mixing of silicates on iron meteorite parent bodies. Earth and Planetary Science Letters 186, no. 2:311–324.
Spivak-Birndorf L., M. Wadhwa, and P.E. Janney. 2005. 26Al-26Mg chronology of the D’Orbigny and Sahara 99555 angrites. Meteoritics and Planetary Science 40:A145.
Spivak-Birndorf, L., M. Wadhwa, and P.E. Janney. 2009. 26Al-26Mg systematics in D’Orbigny and Sahara 99555 angrites: Implications for high-resolution chronology using extinct chronometers. Geochimica et Cosmochimica Acta 73, no. 17:5202–5211.
Stern, R.A., and W. Bleeker. 1998. Age of the world’s oldest rocks refined using Canada’s SHRIMP: the Acasta gneiss complex, Northwest Territories, Canada. Geoscience Canada 25, no. 1:27–31.
Stewart, B.W., D.A. Papanastassiou, and G.J. Wasserburg. 1993. S-Nd systematics of silicate inclusions in iron meteorites: Results from Caddo (IAB). Lunar and Planetary Science Conference 24:1359–1360.
Stewart, B.W., D.A. Papanastassiou, and G.J. Wasserburg. 1996. Sm-Nd systematics of a silicate inclusion in the Caddo IAB iron meteorite. Earth and Planetary Science Letters 143, no. 1–4:1–12.
Sugiura, N., A. Miyazaki, and K. Yanai. 2005. Widespread magmatic activities on the angrite parent body at 4562 Ma ago. Earth Planets Space 57:e13–e16.
Takeda, H., D.D. Bogard, D.W. Mittlefehldt, and D.H. Garrison 2000. Mineralogy, petrology, chemistry, and 39Ar-40Ar and exposure ages of the Caddo County IAB iron: Evidence for early partial melt segregation of a gabbro area rich in plagioclase-diopside. Geochimica et Cosmochimica Acta 64, no. 7:1311–1327.
Tatsumoto, M., R.J. Knight, and C.J. Allègre. 1973. Time differences in the formation of meteorites as determined from the ratio of lead-207 to lead-206. Science 180, no. 4092:1279–1283.
Tatsumoto, M., D.M. Unruh, and G.A. Desborough. 1976. U-Th-Pb and Rb-Sr systematics of Allende and U-Th-Pb of Orgueil. Geochimica et Cosmochimica Acta 40, no. 6: 617–634.
Tera, F., and R.W. Carlson. 1999. Assessment of the Pb-Pb and U-Pb chronometry of the early solar system. Geochimica et Cosmochimica Acta 63, no. 11–12:1877–1889.
Tonui, E.K., H.H. Ngo, and D.A. Papanastassiou. 2003. Rb-Sr and Sm-Nd study of the D’Orbigny angrite. Lunar and Planetary Science Conference 34:#1812.
Trieloff, M., E.K. Jessberger, and C. Fiéni. 2001. Comment on “40Ar/39Ar age of plagioclase from Acapulco meteorite and the problem of systematic errors in cosmochronology” by Paul R. Renne. Earth and Planetary Science Letters 190, no. 3:267–269.
Tyler, D.J. 1990. The tectonically-controlled rock cycle. In Proceedings of the Second International Conference on Creationism, ed. R.E. Walsh and C.L Brooks, vol.2, pp.293–301. Pittsburgh, Pennsylvania: Creation Science Fellowship.
Valley, J.W., A.J. Cavosie, T. Ushikubo, D.A. Reinhard, D.F. Lawrence, D.J. Larson, P.H. Clifton, T.F. Kelly, S.A. Wilde, D.E. Moser, and M.J. Spicuzza. 2014. Hadean age for a post-magma-ocean zircon confirmed by atom-probe tomography. Nature Geoscience 7, no. 3:219–223.
Vardiman, L., A.A. Snelling, and E.F. Chaffin, eds. 2005. Radioisotopes and the age of the earth: Results of a young- earth creationist research initiative. El Cajon, California: Institute for Creation Research, and Chino Valley, Arizona: Creation Research Society. http://www.icr.org/rate2/.
Vogel, N., and P.R. Renne. 2008. 40Ar–39Ar dating of plagioclase grain size separates from silicate inclusions in IAB iron meteorites and implications for the thermochronological evolution of the IAB parent body. Geochimica et Cosmochimica Acta 72, no. 4:1231–1255.
Wasserburg, G.J., F. Tera, D.A. Papanastassiou, and J.C. Huneke. 1977. Isotopic and chemical investigations of Angra dos Reis. Earth and Planetary Science Letters 35, no. 2:294–316.
Wasson, J.T., and J. Wang. 1986. A nonmagmatic origin of group-IIE iron meteorites. Geochimica et Cosmochimica Acta 50, no. 5:725–732.
Weisberg, M.K., T.J. McCoy, and A.N. Krot. 2006. Systematics and evaluation of meteorite classification. In Meteorites and the early solar system II, ed. D.S. Loretta and H.Y. McSween, 19–52. Tucson, Arizona: University of Arizona Press.
Wheelock, M.M., K. Keil, C. Floss, G.J. Taylor, and G. Crozaz. 1994. REE geochemistry of oldhamite-dominated clasts from the Norton County aubrite: Igneous origin of oldhamite. Geochimica et Cosmochimica Acta 58, no. 1:449–458.
Wilde, S.A., J.W. Valley, W.H. Peck, and C.M. Graham. 2001. Evidence from detrital zircons for the existence of continental crust and oceans on the Earth 4.4 Gyr ago. Nature 409:175–178.
Zartman, R.E., E. Jagoutz, and S.A. Bowring. 2006. Pb-Pb dating of the D’Orbigny and Asuka 881371 angrites and a second absolute time calibration of the Mn-Cr chronometer. Lunar and Planetary Science Conference 37:#1580.
Zipfel, J., H. Palme, A.K. Kennedy, and I.D. Hutcheon. 1995. Chemical composition and origin of the Acapulco meteorite. Geochimica et Cosmochimica Acta 59, no. 17:3607–3627.
Zhou, Q., R.A. Zeigler, Q.-Z. Liu, R.L. Korotev, B.L. Joliff, Y. Amelin, K. Marti et al. 2012. U-Pb dating of zircons and phosphates in lunar meteorites, acapulcoites and angrites. Lunar and Planetary Science Conference 43:#1554.










