
Should Sediba Sashay to the Throne for Oldest Human Evolutionary Ancestor?
Should sediba overshadow Lucy, or are both pretenders to the throne in human origins?
Abstract
Since Australopithecus sediba’s “hat” was tossed in the ring as a candidate for the oldest human evolutionary ancestor (so-called) by its discoverer Lee Berger, evolutionary paleontologists have debated several of Berger’s claims. A recent new special issue of Science magazine devoted to the latest research on sediba fossils (reminiscent of a similar one in 2011) hails sediba as an exceptional ape-human (australopithecine-Homo) mosaic. Yet a closer look at the latest analyses reveal that sediba is a fine illustration of ape diversity, but the claims of Homo-mosaicism are clearly in the eye of the beholder. In particular, the latest results reveal numerous problems with the Berger team’s claim of sediba bipedality.
Introduction
Since the discovery of Australopithecus sediba fossils in 2008 at Malapa Cave, some collapsed pits north of Johannesburg, South Africa, anthropologist Lee Berger has been making a valiant effort to place sediba—a name meaning “wellspring”—squarely in the ancestral line of humans. Reports in the 9 April 2010 and 9 September 2011 issues of Science met with mixed reviews from fellow evolutionists. Even the replica of sediba’s skull—now popularly known as “Karabo”—donated to London’s Natural History Museum was, at the time of my visit there in November 2011, relegated to a side hallway far from the human evolution display.1 Will the latest series of analyses just published in the 12 April 2013 issue of Science finally enable sediba to supplant Lucy as the queen mother of human origins?
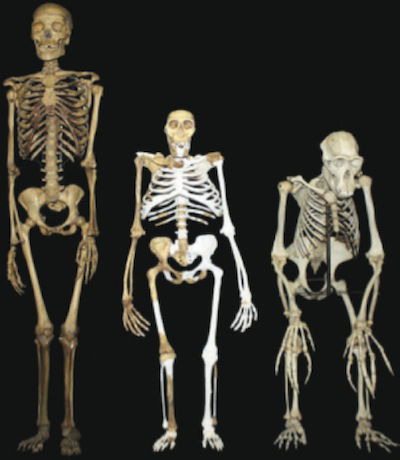
Standing proudly between a human skeleton to the left and a chimp skeleton to the right is a composite reconstruction of Australopithecus sediba. The light gray areas represent the areas filled in. Determined to make sediba walk upright, the researchers on Berger’s team have had to do some acrobatics of their own to reconcile various anatomic features incompatible with bipedal locomotion with a supposed stroll across ancient southern Africa. They have come up with a swaying, sashaying, awkward, “hyperpronating” gait unlike anything proposed for any creature—living or extinct—and completely unlike the way human beings walk. Image by Lee R. Berger and the University of the Witwatersrand, via Science.2
Australopithecus sediba fossils analyzed in the six new articles just published in Science consist of a number of bones from a headless female adult, a juvenile male, and an isolated tibia (shin bone). In addition to an introductory article by Berger (“The Mosaic Nature of Australopithecus sediba”) and an editorial summary by Ann Gibbons (“A Human Smile and Funny Walk for Australopithecus sediba”), there are individual analyses of the mandible, teeth, thorax, vertebrae, and upper and lower limbs. Some critics have suggested that Australopithecus africanus and Australopithecus sediba were the same species. These articles reveal, the researchers conclude, that sediba resembled but was distinct from Australopithecus africanus.
Borrowing Berger’s and Gibbons’s descriptives, when sediba “sashayed” with a “swag”3 across the sands of time with a peculiar gait assigned to her by Berger’s team, she apparently shuffled the deck of australopithecine diversity. But did she shuffle along with the seeds of human evolution? The Berger team maintains that sediba possesses a mosaic of traits including many human features. They are sure these features secure sediba’s rightful place as the oldest human ancestor.

Karabo—“the answer” Lee Berger proposes to the search for humanity’s evolutionary link to an ape-like past—is the name paleontologist Lee Berger has given to the male juvenile Australopithecus sediba, here provided with a number of imaginative features in a “forensic” portrait. Image from National Geographic, via The Daily Mail.

The skull of Australopithecus sediba is fairly typical of apes, having brow ridges, a jutting lower face, and no significant prominence to the nasal bones. Image from AFP/Getty Images, via The Daily Mail.
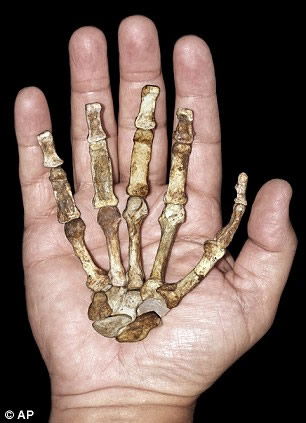
Despite claims the sediba’s hand is a very Homo-like feature, the hand is clearly australopithecine in the opinion of paleoanthropologist William Kimbel, who has examined the original fossils. The thumb is disproportionately long, a feature that may have been useful for sediba’s arboreal lifestyle. (Read more about the hand under “MH2’s Right Hand of Fellowship” at “Sediba with a Little Sleight of Hand.”) Image from AP, via The Daily Mail
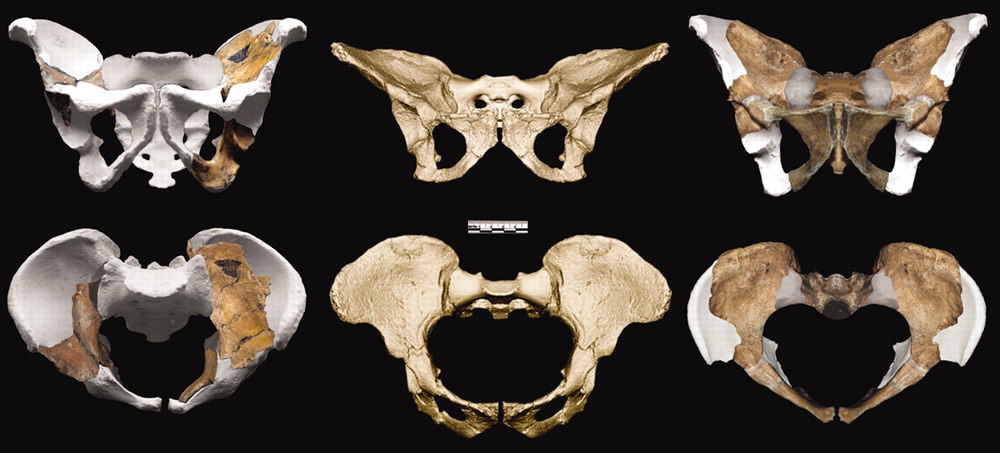
Australopithecus sediba’s pelvis is reconstructed on the left (adult female) and the right (juvenile male). In the center is a pelvis from another australopithecine, Australopithecus africanus. The missing and therefore reconstructed portions of the sediba pelvis are depicted in gray. These also happen to be the portions required for bipedal locomotion. The actual pelvic pieces do not contain the relevant parts, as we discussed in “Sediba with a Little Sleight of Hand.” There is therefore a great potential for observer bias, and, largely on the basis of these pelvic fossils, Berger’s team maintains that sediba was bipedal. Image from Science.4
The Answer?
Berger notes that the difficulty in showing how sediba relates phylogenetically to other hominins is due to the fragmentary nature of the rest of the fossil record. But because his team concludes that these sediba fossils resemble genuine Homo more than any other, Berger is certain that South Africa has supplied “The Answer” to the evolutionary quest for the missing link in human evolution. (Karabo—meaning “the answer”—is the name Berger gave to the male fossil and his fetching portrait unveiled in 2011.)5
“We're getting an unprecedented look at an early hominin species in a field that is generally identified by fragments, and often very small fragments, so we're beginning to see a record that's more complete than we've ever had before,” says Berger, noting sediba’s nearly intact hand, leg, and skull.6 “Overall, the fossils provide an unprecedented insight into the anatomy and phylogenetic position of an early human ancestor,” he claims.7
“This examination of a large number of associated, often complete and undistorted elements gives us a glimpse of a hominin species that appears to be mosaic in its anatomy and that presents a suite of functional complexes that are different from both those predicted for other australopiths and those of early Homo,” Berger writes. “Such clear insight into the anatomy of an early hominin species will clearly have implications for interpreting the evolutionary processes that affected the mode and tempo of hominin evolution and the interpretation of the anatomy of less well-preserved species.”8
Berger notes, for instance, that the comparative anatomy of the jaw suggests “a possibility that Au. sediba, and perhaps Au. africanus, did not descend from the Au. afarensis [Lucy] lineage.”9 “We thought we knew how this part of human evolution worked,” Berger says, “and sediba comes along and says, ‘Here's another option.’”10 Darryl J. de Ruiter, lead author of the mandible study, says, “Australopithecus sediba is unique in size, shape and pattern of growth, though it does share features with older australopiths, as well as later specimens referred to Homo erectus. It represents a transitional form between australopiths and Homo, the genus to which we humans belong, though it looks more like Homo than any other australopith ever found,” adding that the findings “show very strong support of Darwin’s theory of evolution by natural selection.”11 De Ruiter admits that the findings do remain open to interpretation due to the incompleteness of the overall hominin fossil record but is convinced that “we are looking at the ancestor of the genus Homo, or at least a very close relative of that ancestor.”12
Building on the Past
The latest batch of anatomical analyses rests heavily on the foundation of conclusions drawn from the 2011 group, which we discussed thoroughly in “Sediba with a Little Sleight of Hand.” Though some of the claims made in 2011 remain controversial among evolutionists, Berger’s team treats those conclusions as axiomatic and takes up the march to the pivotal human ancestral spot from there.
For instance, in claiming that Au. sediba possesses a mosaic of human and ape traits, the current articles consider the hand—analyzed in 2011—to be the only Homo-like feature of the upper extremity. Yet the hand is not at all like a human hand. The thumb is proportionately much too long and the fingers too short. While Berger’s team has claimed the hand is an adaptation suitable for manipulating tools—none of which are associated with sediba, incidentally—the hand is actually quite nicely suited for arboreal life. As paleoanthropologist William Jungers of Stony Brook, who also examined the original fossils, said, “it’s ‘an australopithecine hand in essentially all relevant respects’ and didn’t manipulate objects in an advanced humanlike way.”13 (For an extensive discussion of sediba’s hand, see “Sediba with a Little Sleight of Hand.”)
Nevertheless, the hand, matched with the extreme ape-ness of the arm and shoulder currently described—perfect for swinging through trees—Berger and his colleagues dub Au. sediba a mosaic in which the other required Homo adaptations had not yet occurred, awaiting a later “full-time commitment to terrestrial environments.”14
Berger writes, “With the exception of the hand skeleton, the upper limbs of the Malapa hominins are largely primitive in their morphology. Au. sediba thus shares with other australopiths an upper limb that was well suited for arboreal climbing and possibly suspension, although perhaps more so than has previously been suggested for this genus.”15 In truth, the hand was as well-suited to an arboreal life as the rest of the arm.
“Stepping” up to the Plate?
Not surprisingly, much of the emphasis in this special sediba issue of Science focuses on showing how Au. sediba walked. As with Lucy (Australopithecus afarensis), the sine qua non for any ape or ape-like animal to claim a place in evolutionary human lineage is a bipedal gait. “Bipedalism has traditionally been regarded as the fundamental adaptation that sets hominids apart from other primates.”16 And not just an inefficient acrobatic maneuver such as any modern primate can achieve will do. A small brain (like sediba’s) is forgivable in a human ancestor, the current evolutionary presumption being that given enough time and a chance to view the world face on, the brain size and intellect will catch up. A hominin candidate must demonstrate an anatomy suited for sustained habitual bipedal locomotion to enter humanity’s hall of ancestors.
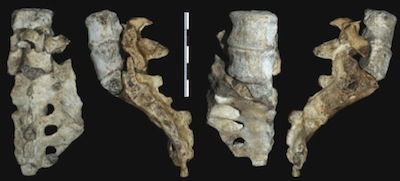
These views show the two lower lumbar vertebrae and the sacrum of the adult Australopithecus sediba from Malapa Cave. The lowest lumbar vertebra has a great deal more dorsal wedging than that found in other australopithecines or in ordinary humans. That, coupled with other features, leads the researchers from sediba-discoverer Lee Berger’s team to conclude that this specimen had a much longer and more flexible lower back than humans, in addition to an extreme lumbar curve and a pelvis that was tilted far forward. Image by Scott A. Williams et al., via Science.17
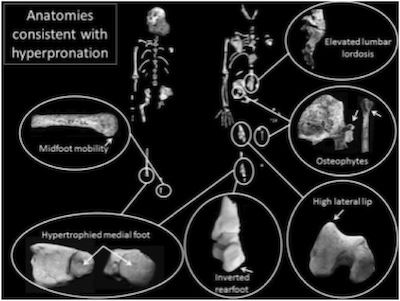
Because the Berger team is convinced—largely on the basis of “reconstructed” (missing) parts of the Au. sediba pelvis—that sediba was bipedal, they had to come up with some sort of pseudo-bipedal gait to account for a number of anatomical features that are inconsistent with bipedal locomotion. This collection of skeletal features found on the juvenile and a random tibia (left) and the adult (right) are used to support the awkward “hyperpronating” sashaying gait they propose. Image by Jeremy M. DeSilva et al., via Science.18
Therein lies sediba’s problem. Berger’s team has always claimed, largely on the basis of “reconstructed” (i.e., missing) pelvic parts, that Au. sediba must have been bipedal. However, many aspects of sediba’s anatomy, as revealed in the current issue of Science, testify loudly against the possibility of sediba walking around South Africa on two legs. In an effort to reconcile several inconsistencies related to the claimed bipedal locomotion, the researchers conclude that this animal walked bipedally in a unique and awkward way unseen in any other known animal, extant or extinct, and certainly very different from the bipedal gait of humans. (We have seen the same maneuver—invention of a unique sort of “australopithecine bipedality”—in Lucy’s history, as discussed in A Look at Lucy’s Legacy and Lucy, the Knuckle-Walking “Abomination”? but the gait the Berger team has created for sediba strains credulity even further.)
Much of the problem in ascribing an upright gait to sediba lies in the inadequate surface area of the heel. The bottom of the heel just isn’t flat enough to provide a stable, stress-dissipating surface at the beginning of a step. Think of trying to walk on ice skates, and imagine the unnatural, unstable, and awkward contortions you would need to make to remain upright walking around on two such feet. In “Body un-designed” we discussed the well-designed human foot anatomy that stabilizes the truly bipedal human gait. Au. sediba’s foot lacks even this most foundational beginning for bipedal walking, a stable start for her step. Ever since Berger’s team revealed the heel was “‘exquisitely small,’ twisted, and pointed as in a chimp,”19 many experts have questioned how such an animal could possibly walk upright. Therefore, in order to keep sediba walking upright, the researchers propose the animal had to strike the ground with the side of its foot and then roll the foot inward (“hyperpronating” it).
Humans normally “pronate” the foot after the heel strikes the ground by simultaneously everting, abducting, and dorsiflexing it. This transforms the rigidity of the foot necessary for stability when the heel strikes to a looser configuration in order to absorb stress and adapt to uneven surfaces.20 The authors define “hyperpronation” as “a continued pronation into the part of stance phase when the foot should be resupinating (usually at the later part of midstance and during the entire propulsive phase).”21
This proposed hyperpronating gait—described by paleanthropologist Brian Richmond as “provocative”22—would place a great deal of stress on the lower back, the knee, and “any muscle crossing both the knee and the hip joints (e.g., the rectus femoris), particularly those inserting laterally in the leg.”23 Richmond, who was not involved in the study, and others wonder if this particular animal walked in an unusual way due to injury, noting “The key question is whether this was typical for the species.”24 The fossil exhibits evidence of osteophytic change—a bony overgrowth associated with stress—in the region of the knee. The researchers interpret this finding as evidence the “hyperpronating” bipedal gait was normal for sediba as well as evidence for great diversity in the australopithecine proto-human evolution of bipedality. However, such bony changes may have just been a manifestation of chronic stress induced by an unnatural, awkward gait in an individual, possibly injured, animal. And certainly nothing about this anatomy demonstrates evolution toward un-ape-ness.
Furthermore, to keep from falling down while hyperpronating, an upright walker would have to shift his center of gravity backward through a “compensatory hyperlordosis”25 of the lower back. Sediba’s lowest lumbar vertebra does exhibit a dorsally wedged shape. Such dorsal wedging is associated with a lumbar curve. Humans have some dorsal wedging, most pronounced on the lowest lumbar vertebral body, and this is associated with the eventual formation of a lumbar curve (aka lumbar lordosis) as we develop an upright posture. Our lumbar lordosis enables us to shift our center of gravity over the hips.
The researchers infer that Au. sediba probably had five (or possibly six) lumbar vertebrae. The juvenile specimen has what appeared to be two higher lumbar vertebrae, and the adult female had two lower ones that matched the sacrum below it.26 As they note, most humans have five. The ability to develop a lumbar curve, however, contrary to popular belief, is fairly independent of the number of lumbar vertebrae. Great apes have only three or four, but gorillas that habitually walk with an unnatural and awkward bipedal gait can develop such a curve.27 On dissection the curve was found to result from alterations in the shapes of the intervertebral discs, presumably remodeled as a result of the animals’ behavior rather than any heritable feature. And gibbons and Old World monkeys, with six or seven lumbar vertebrae, do not have such a curve.28 We recently explored the current state of knowledge and confusion as well as many misconceptions about the number of lumbar vertebrae in primates and humans in “Lucy, the Knuckle-Walking “Abomination”?”
Even more significant than the number of vertebrae for proving the researchers’ claim of bipedality, however, is the “strongly wedged last lumbar vertebra.”29 The writers claim that the extreme lumbar dorsal wedging “demonstrates an intrinsic lumbar lordosis and adaptation to upright posture and bipedal locomotion in Au. sediba.”30 However, the lower vertebrae are actually much more wedged than typical in humans. The lowest (presumably L5) vertebral body’s angle of -10.9 degrees, “greatly surpassing lordotic wedging observed at the last lumbar vertebrae in other Australopithecus specimens,” is so extreme as to be only rarely seen in humans.31 Additional anatomical clues suggest that the sediba pelvis was tilted much farther forward than that of humans and that the lumbar portion of sediba’s back was much longer, more flexible, and more mobile than that of humans.32
Mosaic, Transitional, or Unique?
The analysis of the upper limb and shoulder confirm sediba was well-designed for an arboreal lifestyle . . . just like other apes.
Thus, it seems that sediba’s anatomy was not only totally unsuited for a human sort of stable bipedal locomotion but was instead—unless these bones belonged to a chronically injured animal—uniquely designed for something else entirely. And if the original owner of these bones made a habit of attempting to walk upright when it wasn’t swinging through trees, it placed a great deal of stress on its bones and connective tissues. Incidentally, the analysis of the upper limb and shoulder confirm sediba was well-designed for an arboreal lifestyle, “climbing trees or scrambling up rocky cliffsides”33—just like other apes.
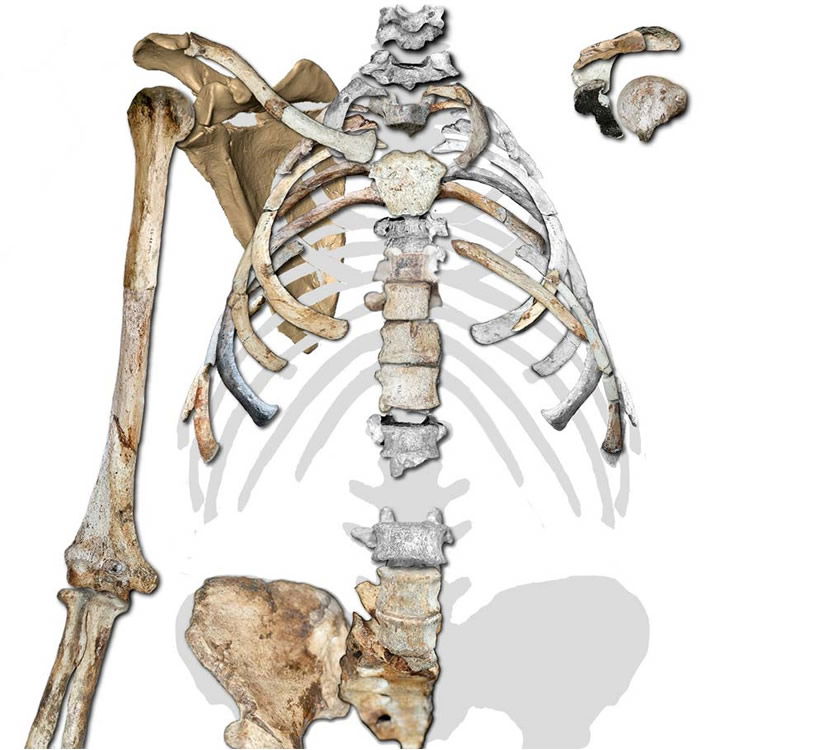
The conical thorax of the adult Australopithecus sediba fossil is quite typical of that seen in modern apes, and the upper limb and shoulder are well-suited for sediba’s arboreal lifestyle. Image by Peter Schmid et al., via Science.34
Despite the emphasis on a “mosaic” anatomy with subtle human characteristics—cited as evidence that this was a “transitional” ape making evolutionary progress toward becoming human—the new analyses primarily confirm sediba was just an ape, though possibly a different species than other australopithecine apes. The mandible and teeth study authors, for instance, based conclusions on characteristics that are known to vary greatly. The data also depended on fairly subjective, non-measurable characteristics, leading George Washington University paleoanthropologist Bernard Wood to comment, “Without more information, I don’t know whether this is something I want to bet $5 on, $50 on, or $500 dollars on.”35 Paleontologist William Kimbel, who has done extensive work with the Australopithecus afarensis (“Lucy”) fossils, comments, “These are minor differences in the crowns [of teeth]. To try to apply this to hominins that are millions of years old is fraught with difficulty.”36
An additional symptom of Au. sediba’s essential ape-ness is found in its chest. The conical shape of the reconstructed upper thorax is as ape-like as any modern ape’s. Not only is the conical thorax typical of modern apes, it is inconsistent with the anatomy needed for bipedal locomotion.37 The one unusual feature the author’s note is that a fragment thought to probably belong to a lower rib is slenderer than those above it; in living apes the lower ribs are thicker. From this they infer that the lower thorax may have been an evolutionary step toward the slenderer waist seen in humans. Yet they note the rib shows evidence of being tugged on by its attachments to the lower back.38 And as noted above, the lower back of this animal seemed to have a number of unique—not human, just unique—qualities that could have therefore produced this “tug” on the rib.
The conical shape of Au. sediba’s upper thorax is not suitable for efficient bipedal locomotion in that it does not allow for the chest rotation that occurs with arm-swinging. The authors write, “The shape of the thorax and the positioning of the scapulae are critical in bipedal gait, as they affect the pendular mechanics of the upper limbs in their role as counterbalances to trunk rotation.”39 Therefore, they conclude from the chest and shoulder anatomy—as with the foot anatomy—that this sediba specimen could not have walked with a standard bipedal gait.
Science writer Ann Gibbons, commenting on the new studies, writes, “The South African species may knock the queen of australopithecines, Lucy—a member of Au. afarensis—off her long-held perch as the most likely ancestor of Homo,” but “that’s a radical view.”40 For one thing, many evolutionary paleontologists disagree with Berger’s assertions that his fossils pre-date other presumed ancestors of modern humans. The Homo fossil A.L. 666-1 is dated at 2.33 million years by evolutionary reckoning, making Au. sediba, at just under 2 million years, “too late to the party to be the ancestor of Homo,” as George Washington University paleoanthropologist Brian Richmond explains.41 (We dealt with some controversial gymnastics Berger used to secure a desirable age for sediba in our previous article: Sediba with a Little Sleight of Hand. Furthermore, all of these millions-of-years dates are based on a number of unverifiable assumptions.42) Additionally, other evolutionary paleontologists question the methodology and interpretation of the traits selected to “prove” the human-ward mosaicism of sediba.
We would go an additional step in pointing out that there is no evidence that humans evolved from ape-like ancestors in the first place. The researchers—whether involved with sediba or “Lucy”—begin with the assumption that humans must have evolved from ape-like ancestors and then interpret any mixture of traits as mosaic evidence of evolutionary transitions-in-progress. Alternatively, however, by rejecting the presupposition that humans could only exist by evolving from less complex creatures, it becomes clear that there are certainly variations among apes—including extinct ones like the australopithecines—that demonstrate ape diversity. But those variations do not demonstrate that evolution happened. As evolutionary paleoanthropologist William Kimbel comments, “If you look at the discoveries of hominins 2 to 3 million years ago [by evolutionary reckoning], each one seems to shuffle the deck [of traits] in different ways. It adds another subtle layer of information about the diversity of African hominins.”43
For instance, the researchers are determined to make sediba walk upright even though they must hypothesize a bizarre, inefficient, and stressful gait for her. This determination persists despite anatomical evidence from the shoulder, chest, and foot that she did not walk upright. Why? Because evolutionists maintain that small-brained ape-like ancestors began to walk upright and then somehow acquired the genetic information to become something far more complex and quite different—humans. Much of evolutionary research is devoted to trying to figure out, not if, but how this could have happened, but no objective evidence that it does (or did) has been found.
Conclusion
Studies like these identify various features shared with other vertebrates and then presume to connect the dots of common designs to paint a supposed scenario of human evolution from ape-like ancestors. Yet shared traits—common designs—are precisely what we would expect from a Creator God who is the Common Designer of all living things. And His eyewitness account, recorded in Genesis, indicates He created living things to reproduce after their kinds. In other words, living organisms vary within their created kinds but, as observable biology indicates, do not change into new, complex, different kinds, there being no mechanism by which they can.
That Australopithicus sediba’s anatomy differed somewhat from that of modern apes is clear. And these studies all show that it greatly resembled Australopithicus africanus, but also that it had sufficient differences to consider it a different species. Extinct apes, like modern ones, demonstrated diversity. But that is really all this batch of work demonstrates: diversity of apes, not evolution or man’s supposed ancestry. Well, actually, the studies do illustrate one other thing: the obdurate determination, wittingly or not, of evolutionists to make the data say what they want it to say, to align with their predetermined suppositions.
For more information:
- Sediba with a Little Sleight of Hand
- News to Note, December 31, 2011: Year in Review (forensic fantasy in London)
- Can Karabo Catch Up with Lucy's Legacy?
- What Australopithecus sediba Ate
- Did Humans Really Evolve from Apelike Creatures?
- Scars of Evolution or of a Sin-Cursed World?
- Homo erectus Grew Teeth Like Modern Humans, Not Like Chimps
- A Look at Lucy’s Legacy
- Lucy, the Knuckle-Walking “Abomination”?
- New African Fossils Alter Perception of Human Origins
- Supposed Hominid Evolution
- Laetoli Footprints Revisited
- Ancient Humans Grazing?
- News to Note, September 17, 2011 (controversy continues-who came first?)
- Evolutionary Researchers: Human Hand Evolved for Dual Abilities
- Enigmatic Skull Confirmed to Have Flat Face
- Cooking: The World’s Oldest Profession
- Sweet Food For Thought
- It’s an Ape . . . It’s a Human . . . It’s . . . It’s . . . a Missing Link!
- The Problem with Australopithecus sediba
Footnotes
- For more information, see Elizabeth Mitchell, “News to Note, December 31, 2011: Year in Review,” Answers in Genesis, December 31, 2011, https://answersingenesis.org/answers/news-to-know/news-to-note-december-31-2011-year-in-review/.
- Lee R. Berger, “The Mosaic Nature of Australopithecus sediba,” Science 340, no. 6129 (April 12, 2013): 163–165, doi:10.1126/science.340.6129.163.
- Ann Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba,” Science 340, no. 6129 (April 12, 2013): 132–133, doi:10.1126/science.340.6129.132.
- Science Express on September 8, 2011, Science 333, no. 6048: 1407–1411, http://science.sciencemag.org/content/sci/333/6048/1407/F2.large.jpg.
- Mitchell, “News to Note, December 31, 2011.”
- Amar Toor, “Where Did We Come from? Prehistoric Fossils May Rewrite Human Evolution,” The Verge, April 11, 2013, http://www.theverge.com/2013/4/11/4212390/au-sediba-prehuman-fossils-could-rewrite-human-evolution.
- Enrico de Lazaro, “Research Reveals How Australopithecus sediba Walked, Chewed, Moved,” Sci-News, April 15, 2013, http://www.sci-news.com/othersciences/anthropology/article01005.html.
- Berger, “The Mosaic Nature of Australopithecus sediba.”
- Ibid.
- Toor, “Where Did We Come from?”
- Texas A&M University, “Study Confirms Au. Sediba Findings,” Newswise, April 12, 2013, http://www.newswise.com/articles/view/601631.
- Toor, “Where Did We Come from?”
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
- Steven E. Churchill et al., “The Upper Limb of Australopithecus sediba,” Science 340, no. 6129 (April 12, 2013): doi:10.1126/science.1233477.
- Berger, “The Mosaic Nature of Australopithecus sediba.”
- Brian G. Richmond and David S. Strait, “Evidence That Humans Evolved from a Knuckle-Walking Ancestor,” Nature 404 (January 10, 2000): 382–385, doi:10.1038/35006045. Evolutionary anthropologists Brian Richmond and David Scott explained that Lucy could really be a knuckle-walker but still in the human evolutionary line. See recent articles “A Look at Lucy’s Legacy” and Lucy, the Knuckle-Walking ‘Abomination’? about Lucy to learn more about the history and anatomy of bipedal claims.
- Scott A. Williams et al., “The Vertebral Column of Australopithecus sediba,” Science 340, no. 6129: doi:10.1126/science.1232996.
- Jeremy M. DeSilva et al., “The Lower Limb and Mechanics of Walking in Australopithecus sediba,” Science 340, no. 6129 (April 12, 2013), doi:10.1126/science.1232999.
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
- See “Scars of Evolution or of a Sin-Cursed World?” to learn more.
- DeSilva et al., “The Lower Limb and Mechanics of Walking in Australopithecus sediba.”
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
- DeSilva et al., “The Lower Limb and Mechanics of Walking in Australopithecus sediba.”
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
- DeSilva et al., “The Lower Limb and Mechanics of Walking in Australopithecus sediba.”
- Williams et al., “The Vertebral Column of Australopithecus sediba.”
- This was reported in Journal of Anatomy and Physiology, a publication of the Anatomical Society of Great Britain and Ireland, 24 (1890):48–50, describing previous work by D. J. Cunningham as well as original work by Dr. Johnson Symington.
- Martin Haeusler, Sandra A. Martelli, Thomas Boeni, “Vertebrae numbers of the early hominid lumbar spine,” Journal of Human Evolution 43 (2002): 621–643; and David Pilbeam, “The Anthropoid Postcranial Axial Skeleton: Comments on Development, Variation, and Evolution,” Journal of Experimental Zoology (Molecular Developmental Evolution) 302B (2004): 241–267, doi:10.1002/jez.b.22.
- Williams et al., “The Vertebral Column of Australopithecus sediba.”
- Ibid.
- Ibid.
- Ibid.
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
- Peter Schmid et al., “Mosaic Morphology in the Thorax of Australopithecus sediba,” Science 340, no. 6129 doi:10.1126/science.1234598.
- Ibid.
- Ibid.
- Schmid et al., “Mosaic Morphology in the Thorax of Australopithecus sediba.”
- Ibid.
- Ibid.
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
- Ibid.
- See “Radiometric Dating: Making Sense of the Patterns,” “Radiometric Dating: Problems with the Assumptions,” and “Radiometric Dating: Back to Basics.”
- Gibbons, “A Human Smile and Funny Walk for Australopithecus sediba.”
Recommended Resources

Answers in Genesis is an apologetics ministry, dedicated to helping Christians defend their faith and proclaim the good news of Jesus Christ.
- Customer Service 800.778.3390
- © 2024 Answers in Genesis





