The views expressed in this paper are those of the writer(s) and are not necessarily those of the ARJ Editor or Answers in Genesis.
Abstract
Here I provide a compendium of extinct “amphibian” groups, representatives of which may have been carried aboard Noah’s Ark. Following previous work by the Ark Encounter team, I selected the taxonomic rank of family as a first-order proxy for the biblical “kind.” The resulting tabulation places 54 extinct “amphibian” families/kinds on board the Ark. While this number hinges upon taxonomies built upon fossil data (and its inherent shortcomings compared to extant forms), it serves as a reasonable approximation of the number of fossil “amphibians” taken aboard the Ark. When added to previously determined kinds of extant anurans, caudates, and gymnophionans, a total of 248 “amphibian” kinds may have been brought on board the Ark.
Keywords: paleontology, Ark Encounter, baraminology, “kinds,” cladistics, amphibians, tetrapods
Introduction
The subject of the organisms contained in Noah’s Ark extends over centuries. From Buteo’s early tabulation to the recent work of the Ark Encounter team, there have been numerous attempts to tabulate not only how many, but what sorts of animals would be preserved through the Flood on the Ark.
The “amphibians” in particular have received little attention in these tabulations, and are often not included at all. Buteo specifically excluded them in his tabulation of 300 animals from 100 species brought aboard the Ark. Indeed, these 100 species were only mammals, and Buteo did not specifically tabulate any reptiles or birds, thinking that only mammals were large enough to bother counting, and that the others would simply find room among the floors, stalls, and posts (Buteo 1554; Griffith and Miller 2008). Whitcomb and Morris (1961) allowed for a maximum of 35,000 individuals, but did not separate this into the various tetrapod classes. Jones’ (1973) tabulation of roughly 2000 individual animals allowed for a few of the most terrestrial-adapted “amphibians.” None were tabulated in Woodmorappe’s (1996) extensive evaluation of Noah’s Ark (16,000 individuals, maximum), nor were they included by Beech and Ross (2012; 1606 individuals), though Ross (2013) included 70 “amphibian” kinds. In most cases, the authors reasoned that the semiaquatic nature of most “amphibians” permitted their exclusion among the Ark-borne vertebrates. Other authors assumed that the small size of most amphibians resulted in a negligible impact on occupancy space and resources, and so ignored tabulating them and focused instead on those organisms whose larger size has a more measurable effect on the Ark and Noah’s family.
It is now clear that the many “amphibians” (both fossil and living) cannot be excluded from the Ark solely on the basis of semi-aquatic behavior. As will be discussed below, some obligate aquatic forms can be excluded, but many were partially or dominantly terrestrial. And if they are to be included, they should be counted just like any of the other groups and not dismissed due to small size. In fact, there are a good number of very large (>1 m [3.2 ft]) amphibians from the fossil record. The inclusion of all “amphibians,” save fully aquatic forms, in the tally of Ark-borne animals is shared across the Ark Encounter team (e.g., Hennigan, 2013a, b).
This paper’s focus is the extinct “amphibians.” The fossil record of “amphibians” is far greater and more diverse than commonly perceived. When one uses the term “amphibian,” images of frogs, toads, newts, and salamanders are quick to come to mind. Few outside of conservation or academic biology are aware of the other living group: the limbless, burrowing caecilians/gymnophionans. Few fossil forms could be named by members of the general public.
While tremendously specious and consisting of over 200 kinds (see Hennigan 2013a,b), from a morphological point of view, the extant amphibians are rather depauperate, having just three basic body styles (frog-like, salamander-like, and worm-like), and the largest members are at most 1.5 m (4.9 ft) (e.g., the Japanese giant salamander and some aquatic caecilians). In contrast, the “amphibians” of the fossil record display a far wider range of physical forms and sizes. Some were superficially lizard-like; others were large-headed, limbless, and aquatic. Some had boomerang-shaped skulls, while others were snake-like, and still others resembled crocodiles and oversized salamanders. Some resembled fish more than land-dwelling organisms and some were nearly fully terrestrial. Most had an array of sharp teeth, though some had none and at least one possessed a dentition for crushing hard-shelled invertebrates. The smallest were only a few centimeters in length, while the largest were 9 m (29.5 ft) long.
Taxonomic Hierarchies:
Linnaeus and Cladistics
To properly understand the compilation of “amphibians” that follows, and my use of quotation marks for the group, we must first understand the changes in taxonomy that have occurred in the past 50 years in biology (and paleontology specifically).
Biological taxonomy has been dominated by Linnaeus’ hierarchical system for nearly three centuries, and the familiar categories of Phylum, Class, Order, Family, Genus, and Species are still used by biologists. The Linnaean system groups organisms according to their possession of shared characters, such as limbs and non-amniotic eggs in amphibians, amniotic eggs and scales in reptiles, and hair and mammary glands in mammals.
In Linnaean taxonomy, Amphibia is one of the four classes of terrestrial vertebrates (the others being Reptilia, Mammalia, and Aves). The continued discovery of new organisms (both living and fossil) has also led to additional categories modified from the classical ones above, such as Infraclass, Suborder, and Superfamily. So while a typical biology textbook will only list and discuss the classical members, the existing realm of taxonomy is considerably more complex, and the delineations between the multitude of groupings can be hazy, at best.
Importantly, modern cladistic methods are now favored by biologists and paleontologists, and scientific description of new organisms routinely includes cladistic evaluations to place the new organism in taxonomic context. These methods have introduced a strain onto the Linnaean system by multiplying exponentially the number of possible hierarchical “bins.” Briefly, cladistic methodology involves the numerical coding of physical features of an organism, such as the presence/absence of a structure, the relative length of a bone, etc. (table 1). Once a group of organisms is coded, the matrix of character states is processed by a computer program to create a branching tree diagram called a cladogram (fig. 1).
It is important to understand that in these diagrams the actual taxa are found only at the tips of the cladogram’s branches; they are not found at the nodes (connection points) of those branches. These nodes reflect the characters that unite all members whose branches share that node (fig. 1). These characters are referred to as “shared-derived characters” since evolutionary biologists and paleontologists believe that all taxa which trace back to the node derived these characters from a shared common ancestor.
Table 1. A hypothetical cladistic data set.
| Taxa | Character i | Character ii | Character iii | Character iv |
|---|---|---|---|---|
| Genus a | 0 | 0 | 0 | 0 |
| Genus b | 1 | 0 | 0 | 0 |
| Genus c | 1 | 1 | 0 | 0 |
| Genus d | 1 | 1 | 1 | 0 |
| Genus e | 1 | 1 | 1 | 1 |
The use of “shared-derived characters” is somewhat different than the “shared characters” of the Linnaean system. As noted above, Class Amphibia can be diagnosed by the possession of limbs and non-amniotic eggs under the Linnean system. However, the non-amniotic, jelly-like eggs of “amphibians” are similar to the eggs seen in many fishes. So not only are these characters shared outside of “amphibians,” they are shared with putatively more primitive members along the evolutionary tree. The possession of limbs is a shared character in common with “reptiles,” birds, and mammals, and can be used as a shared-derived character, since limbs are not seen in fish (with “fish” being another cladistically problematic term!).
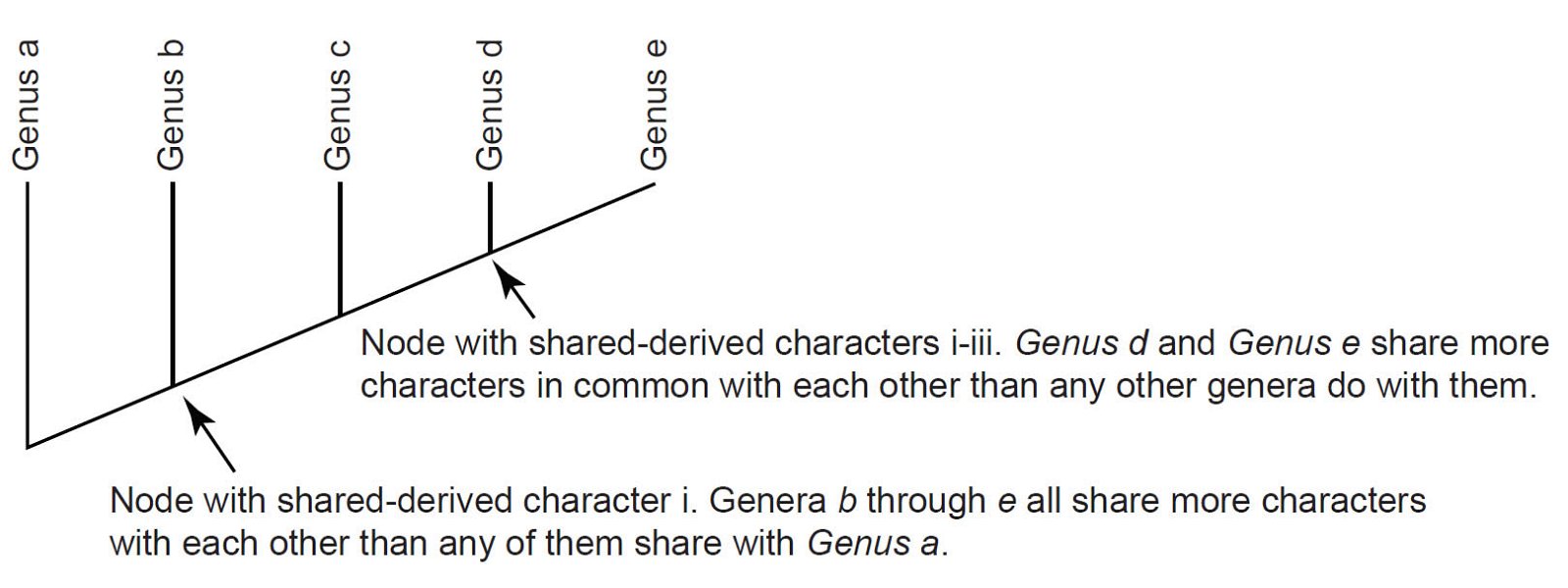
Fig. 1. A cladogram based on data from Table 1.
A cladogram thus serves as a hypothesis of evolutionary relationships among the organisms. While universally used within the evolutionary paleontological community, the underlying structure is purely mathematical; evolution is an inference from the analysis. This is why some young-earth creationists take the same datasets used in cladistic analyses and apply them to other methods, such as multidimensional scaling (see Wood and Murray 2003 for an extended discussion); the dataset is agnostic towards the methods applied to it. Strictly construed, cladograms group organisms according to their possession of successively more numerous and specific characters, within the confines of a bifurcating tree structure.
Once a cladogram is produced, biologists use this to evaluate whether groups of organisms under investigation form a natural grouping, falling into a tree rooted by the possession of at least one shared-derived character. If this is the case, then the group is considered monophyletic (“one tree”). However, there are other possibilities. A defined group might be rooted to a node but not include some other members that also root to that node. Such groups are called paraphyletic (“partial tree”). Also, a polyphyletic (“many trees”) group might include members that do not directly connect to a single node. Figs. 2–4 depict how each of these is diagnosed.
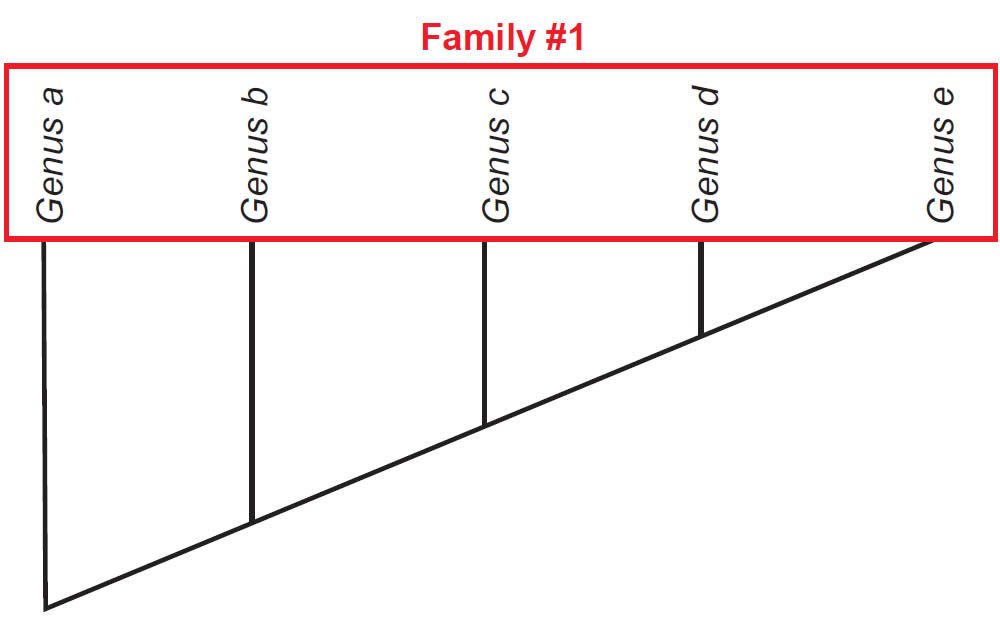
Fig. 2. A monophyletic tree for Family #1. All members of the family are united by the shared-derived characters possessed at the bottom left node of the cladogram.
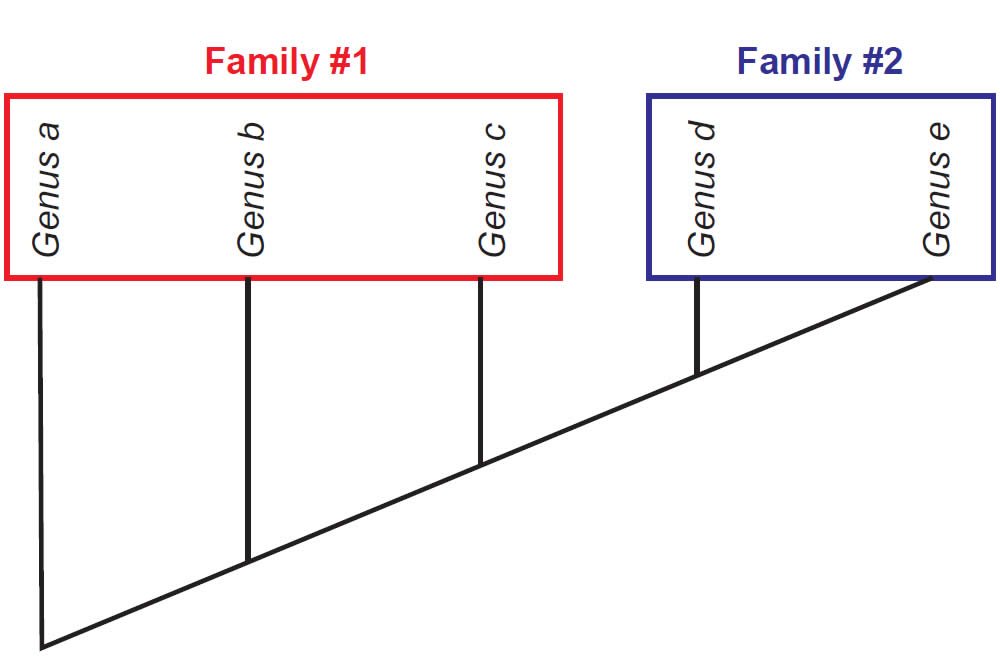
Fig. 3. A paraphyletic tree for Family #1 and a monophyletic tree for Family #2. Family #1 is paraphyletic because Genus b and Genus c share more derived characters with Genus d and Genus e than they do with Genus a. Family #2 is monophyletic because Genus d and Genus e are united to one node by a set of shared-derived characters.
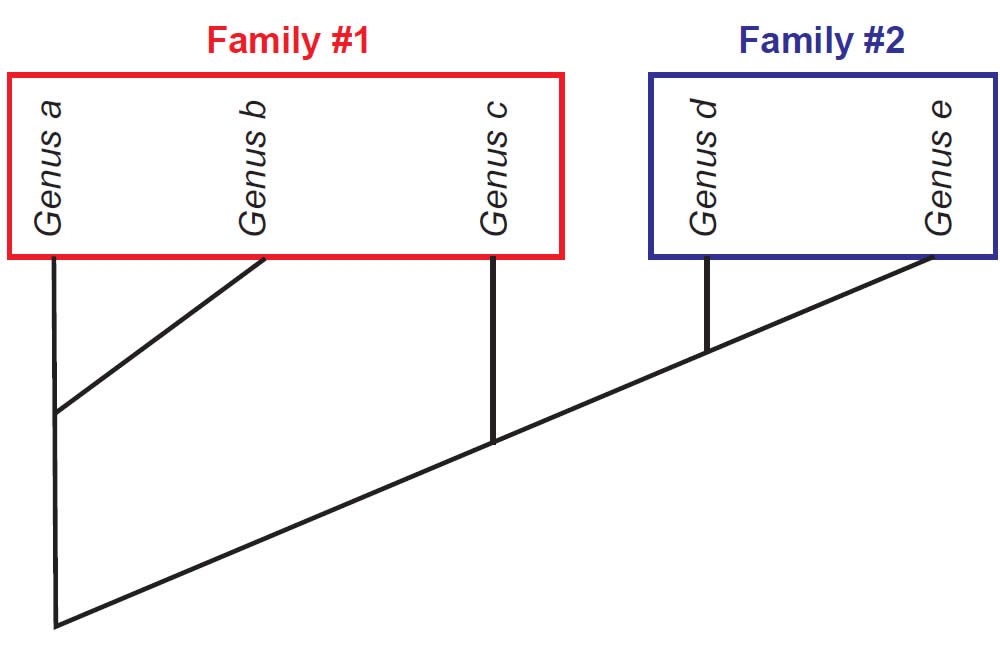
Fig. 4. Family #1 is polyphyletic, because Genus c is included in the family, though it actually shares more characters in common with Genus d and Genus e. Family #2 is monophyletic.
Taxonomic Issues with the “Amphibia”
It is necessary to illustrate these systems and issues, as all current vertebrate paleontological taxonomies make use of cladistics in categorizing all known fossil taxa. This study follows these resources, and to a great extent is bound to established taxonomies. It is far beyond the scope of this paper to investigate the baraminological relationships among the diverse menagerie of fossil “amphibians,” though this may be useful to young-earth creationism.
Relevant to this investigation, the application of cladistics has illuminated an interesting problem in diagnosing the tetrapod (limbed vertebrate) classes Amphibia and Reptilia. Consider that, according to evolutionary theory, a certain group of “amphibian” tetrapods is supposed to have evolved into a group of primitive “reptiles.” Therefore, the “Amphibia” is defined as being those tetrapods which are not amniotes, and the resulting cladogram (fig. 5) presents the “Amphibia” as a paraphyletic group. Likewise, the common term “reptile” reflects a paraphyletic or group, as this older term included amniotes that do not have the shared-derived characters of either mammals or birds, both of which are considered monophyletic by evolutionists (fig. 6).
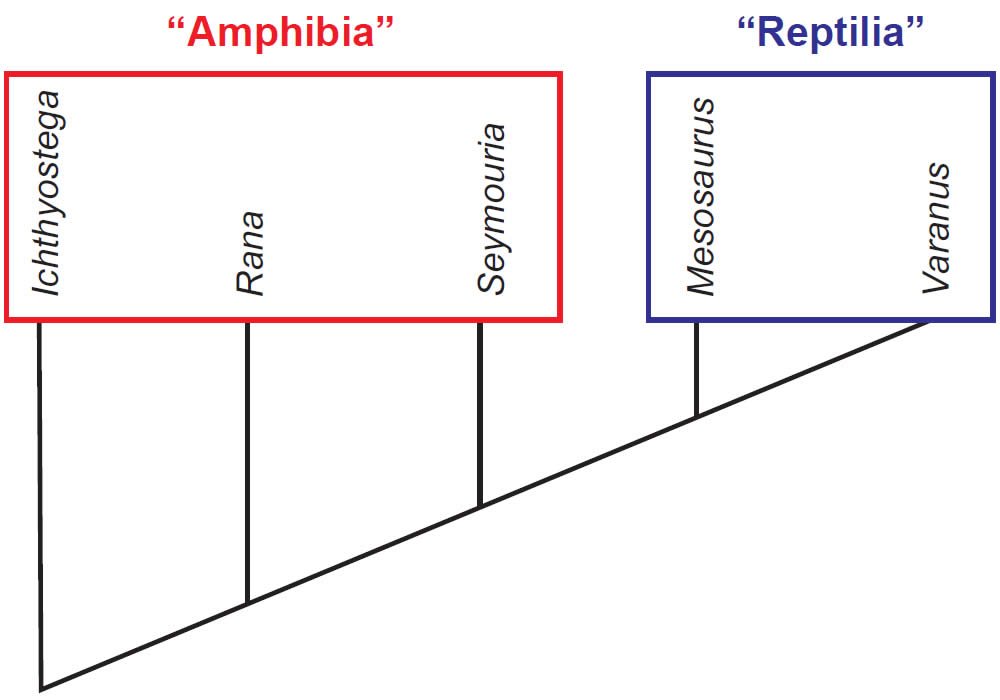
Fig. 5. A simplified cladogram of the “Amphibia,” illustrating its paraphyletic nature.
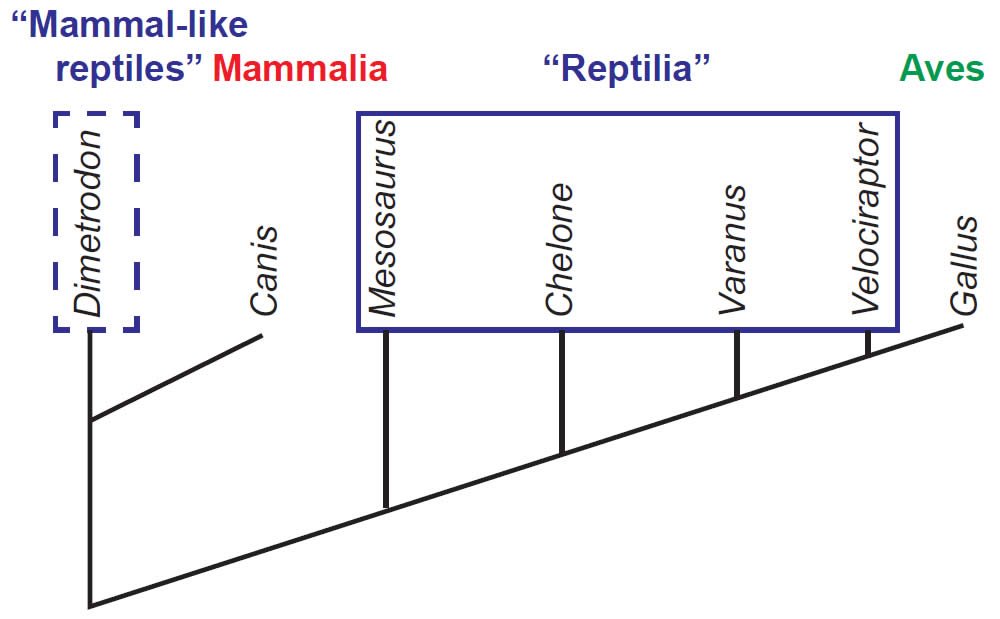
Fig. 6. A simplified cladogram of the “Reptilia,” illustrating its paraphyletic nature, which is more complicated than the case for “Amphibia.” The dashed lines around Dimetrodon indicate that the “mammal-like reptiles” were historically grouped as “reptiles,” though the term “synapsid” is used today.
As a result, in modern, cladistic-oriented taxonomies the classes Amphibia and Reptilia have no truly unifying characters to link them as a natural group. In essence, these groups are defined by what they aren’t rather than by what they are. An “amphibian” is a tetrapod that is not an amniote (fig. 5), and a “reptile” is an amniote that is neither a mammal nor a bird (fig. 6). It is unsurprising, then, that there has been a strong movement among evolutionary taxonomists to abandon these terms altogether, and use other terms that focus on monophyletic groups. So while a creationist surveying taxonomies in the most comprehensive works on the “amphibians” in vertebrate paleontology would find Class Amphibia in use by Carroll (1988), they would not find this term in Benton (2005). These texts stand astride the rise to dominance of cladistic methods.
The informal use of “amphibian” remains, but if the term Amphibia is to exist at all as a formal category, it is restricted to only the living amphibians and the extinct members of these groups (currently referred to as Lissamphibia); it will not apply to the many dissimilar fossil non-amniote tetrapods. Thus living and fossil frogs, toads, newts, salamanders, and caecilians might be properly referred to as amphibians (without quote marks), but Acanthostega, Diplocaulus, Eryops, or Seymouria cannot.
Methods
Taxonomic lists of “amphibians” were reviewed from surveys of vertebrate paleontology texts (e.g., Benton 2005; Carrol 1988; Clack 2012; Schoch 2014) and from the Paleobiology Database (www.fossilworks.org). Lightner et al. (2011) outlined a three of the most useful methods to ascertain an Ark kind: hybridization, statistical baraminology, and the cognitum. Given the quite deceased nature of their subjects, hybridization is a non-starter. Paleontologists must of course define species morphologically as skeletal clues are the primary data available from which to make inferences about which animals may be unified into a biblical “kind.” This leaves statistical baraminology and the cognitum. As mentioned above, this paper does not seek to evaluate the “amphibians” using statistical baraminology. Indeed, given the often fragmentary nature of the “amphibian” fossil record, large numbers of taxa that can be confidently recognized as distinct species and genera would be excluded from BDIST and MDS evaluations due to their low overall percentage of known character states. This would greatly reduce our confidence in recognizing Ark kinds when conducting a comprehensive evaluation of the group. Perhaps smaller, more targeted evaluations could provide better results, but this is a topic for further research both among the “amphibians,” as well as more broadly regarding the application of statistical baraminology to fossil taxa.
This leaves a cognitum-based approach. Despite the occasional vagaries of higher-level taxonomy, various species and genera typically cluster into slightly broader and well-defined families recognized by many authors, and these are here used as a proxy for the “amphibian” Ark kinds. Table 2 presents the tabulation of all families evaluated in this paper, summarizing the number of genera per family and whether the group was fully aquatic or not. Those that were aquatic are not included in the tally of Ark-borne animals.
Table 2. Taxonomy of the “amphibian” families.
| Higher-level Designators | Order (Suborder) | Family | # Genera | Aquatic (y/n) | |
|---|---|---|---|---|---|
| Superclass Tetrapoda | |||||
| Ichthyostegalia | 12 | y | |||
| Other basal tetrapods | Colosteidae | 3 | y | ||
| Whatcheeriidae | 2 | n | |||
| Baphetidae | 5 | n | |||
| Temnospondlyi | |||||
| Cochleosauridae | 5 | n | |||
| Dendrerpetontidae | 3 | n | |||
| (Dvinosauri) | |||||
| Trimerorhachidae | 5 | y | |||
| Tupilakosauridae | 3 | y | |||
| Zatracheidae | 3 | n | |||
| Saurerpetontidae | 2 | n | |||
| Dissorpohidae | 14 | n | |||
| Trematopidae | 7 | n | |||
| Branchiosauridae | 5 | y | |||
| Micromelerpetontidae | 4 | y | |||
| Amphibamidae | 11 | n | |||
| Eryopidae | 4 | n | |||
| Parioxydae | 1 | n | |||
| Archegosauridae | 6 | n | |||
| Melosauridae | 2 | n | |||
| Intasuchidae | 2 | n | |||
| (Sterospondyli) | |||||
| Rhinesuchidae | 6 | n | |||
| Lapillopsidae | 3 | n | |||
| Capitosauridae | 6 | y | |||
| Mastodonsauridae | 6 | y | |||
| Sclerothoracidae | 1 | n | |||
| Brachyopidae | 15 | n | |||
| Plagiosauridae | 11 | y | |||
| Chigutisauridae | 6 | n | |||
| Trematosauridae | 6 | y | |||
| Metoposauridae | 5 | y | |||
| Laidleriidae | 1 | n | |||
| Infraclass Lissamphibia | |||||
| Order Allocaudata | |||||
| Albanerpetontidae | 4 | n | |||
| Order Anura | |||||
| Gobiatidae | 3 | n | |||
| Triadobatrachidae | 1 | n | |||
| Infraclass Lepospondyli | |||||
| Order Lysorophia | |||||
| Lysorophidae | 6 | n | |||
| Order Microsauria | |||||
| Brachystelechidae | 3 | n | |||
| Hyloplesiontidae | 1 | n | |||
| Microbrachidae | 1 | n | |||
| Odonterpetontidae | 1 | n | |||
| Gymnarthridae | 10 | n | |||
| Hapsidopareiontidae | 2 | n | |||
| Ostodolepidae | 5 | n | |||
| Pantylidae | 1 | n | |||
| Trihecatontidae | 1 | n | |||
| Tuditanidae | 3 | n | |||
| Order Nectridea | |||||
| Diplocaulidae | 7 | y | |||
| Scincosauridae | 1 | n | |||
| Urocordylidae | 5 | n | |||
| Order Aïstopoda | |||||
| Lethiscidae | 1 | n | |||
| Oestocephalidae | 1 | n | |||
| Ophiderpetontidae | 2 | n | |||
| Phlegothontiidae | 1 | n | |||
| Pseudophlegethontiidae | 1 | n | |||
| Order Insertae sedis | |||||
| Adelogyrinidae | 4 | y | |||
| Acherontiscidae | 1 | y | |||
| Infraclass Reptilomorpha | |||||
| Order Anthracosauria | |||||
| Caerorhachidae | 1 | n | |||
| (Embolomeri) | |||||
| Eoherpetontidae | 1 | n | |||
| Proterogyrinidae | 2 | n | |||
| Eogyrinidae | 8 | n | |||
| Archeriidae | 3 | n | |||
| Anthracosauridae | 1 | n | |||
| (Seymouriamopha) | |||||
| Waggoneriidae | 1 | n | |||
| Seymouriidae | 1 | n | |||
| Karpinskiosauridae | 1 | n | |||
| Gephyrostegidae | 3 | n | |||
| Discosauriscidae | 5 | y | |||
| Kotlassidae | 4 | n | |||
| (Chroniosuchia) | |||||
| Chroniosuchidae | 6 | n | |||
| Bystrowianidae | 5 | n | |||
Some general comments are presented for the larger divisions (e.g. major clades, orders, and suborders) into which the families are assigned. Each family description follows a basic pattern of a few sentences: general anatomy and mode of life; geographic and geologic distribution; and number of genera within the family. Unless otherwise stated, the family is included in the tally of Ark-borne animals. Where available, images are provided following the description to assist the reader in visualizing members of the family. Typically, a reference is provided for each family to point the reader to additional technical materials. The best sources for a review of these groups are Clack (2012) and Schoch (2014), both experts in this field. These works are thorough, up-to-date, contain copious images, detailed discussions, and are extensively referenced. Clack’s book is narrative in style, while Schoch’s is organized more as a textbook.
Standard chronostratigraphic terminology is used within each description, such as Permian, Jurassic, and Eocene. This is done to help the reader appreciate the location of these fossils within the overall geological record. In this sense, the use of these terms is a relative dating construct (e.g., fossils found in Pennsylvanian rocks are deeper in the sedimentary record than Jurassic rocks, both deposited during the Flood). The multi-million year dates commonly assigned to these terms is not implied by their use in this work.

Fig. 7. Eliginerpeton. Retrieved from http://en.wikipedia.org/wiki/Elginerpeton.

Fig. 8. Ichthyostega. Retrieved from http://en.wikipedia.org/wiki/Ichthyostega.

Fig. 9. Acanthostega. Retrieved from http://en.wikipedia.org/wiki/Acanthostega.
Superclass Tetrapoda
Order Ichthyostegalia
This order is technically paraphyletic, but is used to unify its members based on overall body form and mode of life. Ichthyostegalians were aquatic “basal” tetrapods with a life almost exclusively relegated to shallow waters, though some could possibly (if awkwardly) venture onto land. All were carnivorous, with flat skulls, labyrinthodont dentition, paddle-like, polydactyl hands and feet, and expanded fins on their tails. Most possessed internal gills (Tulerpeton being a possible exception), and adults of the various genera ranged from about 1–2.5 m (3.2–8.2 ft) in length. Due to the fact that much or all of their time was spent in the water, combined with their overall fish-like appearance and typical possession of gills, it is unlikely that any of these organisms would have been brought aboard the Ark.
All known body fossils are thus far largely restricted to upper Devonian rocks in the northern hemisphere (North America, Ireland, Poland, and Russia), though remains are also known from Australia. Interestingly, the stratigraphically lowest evidence of the Ichthyostegalia is not from body fossils, but from a middle Devonian trackway in Poland. The tracks not only pre-date the body fossils of all members of this group by 18 million years on the conventional time scale, they also predate the most likely “ancestors” of the tetrapods (the sarcopterygian group known as elpistostegalians) by 10 million years (Niedzwiedski et al., 2010). The five ichthyostegalian families contain a total of 12 genera, including the well-known genera Icthyostega and Acanthostega. For a review of this group, see Benton (2005). For more extensive discussions, see Clack (2012) and Schoch (2014).
Unassigned Families of “Basal Tetrapods”
In addition to the ichthyostegalians discussed above, there are a number of other families of primarily- to exclusively aquatic “basal tetrapods” which are not included in the suborders described below. These families include Colosteidae, Crassigyrinidae, Whatcheeriidae, and Baphetidae.
Family Colosteidae

Fig. 10. Greererpeton. Retrieved from http://en.wikipedia.org/wiki/Greererpeton.
Colosteidae were semiaquatic, carnivorous amphibians. They were about 1.5 m (4.9 ft) long, had flattened bodies, small limbs, elongate tails, and dermal scales over its skin. The skull is flat, with the orbits oriented dorsally (likely for underwater ambush predation), and possessed a lateral line system. Colosteids were capable of walking on land much better than ichthyostegalians (Benton, 2005), but Clack (2012, p. 281) notes “it is clear that colosteids were fully aquatic,” so they would not be taken aboard the Ark. Fossils are known from Carboniferous deposits in the United Kingdom and the United States. This group includes three genera (notably Colosteus and Greererpteton). See also Godfrey (1989a,b).
Fig. 11. Pederpes. Retrieved from http://en.wikipedia.org/wiki/Pederpes.
Family Whatcheeriidae
Whatcheeriids were semiaquatic, piscivorous amphibians. Two genera, Pederpes and Whatcheeria belong to the family. They possess several fish-like structures in their skull, such as a cleithrum, lateral line, and teeth on the palatal bones. Yet they also possess true limbs, with particularly robust hind limbs compared to the front limbs, and appear capable of walking on land. The eyes of Pederpes are oriented more laterally than dorsally. Fossils of this group are about 1m (3.2 ft) long, and have been discovered in Mississippian/early Carboniferous deposits in the USA and Scotland. See Clack and Finney (2005) and Lombard and Bolt (1995).
Family Baphetidae
The five baphetid genera are known almost exclusively from skull material, with little postcranial remains to determine their overall shape and likely mode of life. The group’s flat, wide skulls can measure up to 30 cm (11.8 in) long or more. Baphetids were carnivorous/piscivorous and characterized by rather unusual, keyhole-shaped orbits. They are known almost exclusively from Upper Carboniferous deposits of the United Kingdom as well as Nova Scotia, Canada, and the United States. See Milner and Lindsay (1998) and Milner et al. (2009).
Order Temnospondyli
Temnospondyls were a very diverse assemblage of fossil “amphibians.” Carroll (1988) lists 34 families in the order, while Benton (2005) claims 40 in the body of the text (p. 88), though his classification appendix lists only 17 of the notable families. The paleobiology database lists 26 families, though some have been subsumed into other families and there are also genera listed that are not included in designated families (due to the use of cladistic methods). Moreover, the Lissamphibia (discussed below) is now considered to nest in part or in full within the Temnospondyli. Excluding the Lissamphibia (as is done here) technically renders the Temnospondyli paraphyletic.
Temnospondyls are united by a suite of similar structures in the skull (particularly an expanded opening in the palate), vertebrae, and hips (see Godfrey, Fiorillo, and Carroll 1987). The group consists primarily of semi- to and fully aquatic organisms, though some (including the stratigraphically lowest members of the group) were quite terrestrial. They ranged from small (roughly 20 cm [7.8 in]) to the largest of all known “amphibians” (9 m [29.5 ft]). Most were 1 m (3.2 ft) long or larger, with broad, flat heads that are either blunt or elongated with extensive pits and ridges. Some had canal-like grooves (sulci) in their skulls that housed a lateral line system for detecting movement in the water, but this feature is not present in the terrestrial forms, even among the earliest members. Some possessed scales, claws, and/or bony plates. Superficially, many resembled either crocodilians or large salamanders. One member of this very diverse group, Eryops, is rather familiar due to its common portrayal in children’s or other lay books on prehistoric life. Aside from Eryops, few outside of vertebrate paleontology would be familiar with any of the many variety of temnospondyls.
Temnospondyls have been found on every continent, primarily from upper Carboniferous (Pennsylvanian) through Triassic deposits, with a few known from as high as lower Cretaceous deposits. The group as a whole and a number of the families below are discussed in Carroll (1988), Clack (2012), and Schoch (2014). The following taxonomic list broadly follows Schoch’s (2013) recent cladistic evaluation of the Temnospondyli.
Family Cochleosauridae

Fig. 12. Dendrerpeton. Retrieved from http:// en.wikipedia.org/wiki/Dendrerpeton.
Cochleosaridae were medium sized temnospondyls (1–2 m [3.2–6.5 m]) from the Carboniferous and Permian of New Mexico, Canada, Europe, and Niger. It was a carnivorous semiaquatic predator and probably hunted in a manner similar to crocodiles. This family contains five genera. See Sequeria (2004) and Steyer et al. (2006).
Clade Eutemnospondyli
Family Dendrerpetontidae
Dendrerpetonts were terrestrial temnospondyls, similar in general appearance to lizards. Species size ranged between 15 cm (5.9 in) and 1.5 m (59 in). Their diet likely consisted of insects and other invertebrates. They are distributed in North America and Ireland in Late Carboniferous (roughly Pennsylvanian) deposits, and are among the stratigraphically lowest members of the temnospondyls. The group includes three genera. See Holmes, Carroll, and Reisz (1998) and Millner (1980).
Suborder Dvinosauria
Family Trimerorhachidae
Fig. 13. Trimerorhachis. Retrieved from http://commons.wikimedia.org/wiki/File:Trimerorhachis_Cope.jpg.
Trimerorhachidae were fully aquatic temnospondyls which likely ate fish and small invertebrates. The largest of the genera reached about 1 m (3.2 ft) in length. They retained external gills throughout their lives and possessed bony plates in their skin. One specimen of Trimerorhachis included juvenile specimens in the pharyngeal region, indicating either brooding behavior or cannibalism. This family contains four genera known from Permian deposits of the Central United States. See Olson (1979). Due to their fully aquatic behavior, they are not included in the tally of Ark-borne animals.
Family Tupilakosauridae
Tupilakosauridae were aquatic temnospondyls found in Greenland and South Africa. A reduced postfrontal bone, the postorbital-parietal suture, and a notch on the posterior pterygoid identify them. This family contains three genera known from the lower Permian to lower Triassic deposits in Texas and France. See Milner and Sequeria (2003) and Werneberg et al. (2007) Due to their fully aquatic behavior, they are not included in the tally of Ark-borne animals.
Family Zatracheidae
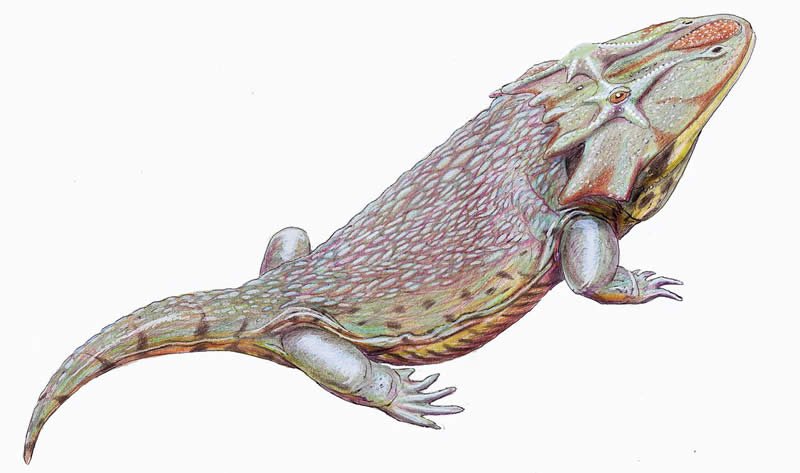
Fig. 14. Dasyceps. Retrieved from http://en.wikipedia.org/wiki/Dasyceps.
Zatracheidae were terrestrial temnospondyls with somewhat salamander-like bodies, and possessed expanded skull bones forming horn-like projections and knobs. Fossils from multiple life-stages are known and indicate that zatracheids metamorphosed from aquatic to terrestrial forms over their lifetime. This family contains three genera known from Carboniferous through early Permian deposits in the United States, England, and Germany. See Patton (1975) and Schoch (2007).
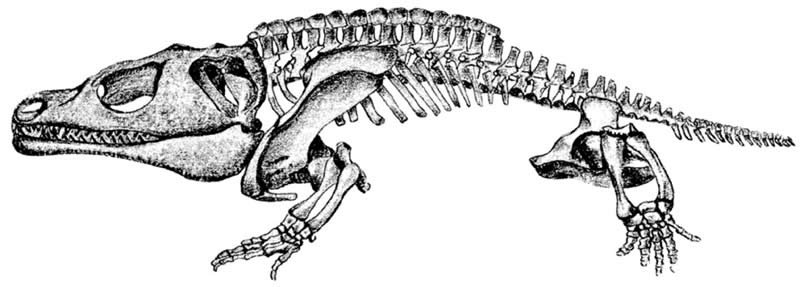
Fig. 15. Dissorophid Cacops. Retrieved from http://en.wikipedia.org/wiki/Cacops.

Fig. 16. Cacops. Retrieved from http://en.wikipedia.org/wiki/Cacops.
Family: Saurerpetontidae
Saurerpetontids were semiaquatic temnospondyls characterized by a short face, a reduced otic notch, by several unique features among the skull bones (tabulars, palatines, and parasphenoid), and rows of rectangular dermal armor along the lower jaw. This family contains two genera known from Pennsylvanian and Permian deposits in the U.S. See Sequeria (1998).
Superfamily Dissorophoidea
Family Dissorophidae
Dissorophidae were medium-sized temnospondyls, with an adult size range between 20 cm (7.8 in)and 1 m (3.2 ft) in length. Dissorophids are some of the most terrestrial of the “amphibians,” with well-developed limbs, solid vertebrae, short tails, and many possessed dermal armor covering the trunk region. They possessed very large fangs, indicating that they preyed on larger animals than other, anatomically similar groups (superfamily Dissorophoidea). They are distributed in North America and Europe in upper Pennsylvanian and lower Permian deposits. The group includes fourteen genera. See Carroll (1988), Clack (2012), and Schoch (2014).
Family Trematopidae

Fig. 17. Acheloma. Retrieved from http://en.wikipedia.org/wiki/Acheloma.
Trematopidae were terrestrial temnospondyls with skulls that are tall and broad posteriorly, but more narrow and U-shaped anteriorly, covered with pitted dermal bone. They were approximately one meter long and, like the Dissorophidae, their dentition included large fang-like teeth on the palatal bones. This family contains seven genera from Carboniferous and Permian rock layers of Germany and the south-central United States. See Polley and Reisz (2011).
Family Branchiosauridae
Branchiosauridae were rather unusual temnospondyls which retained many juvenile traits into their adult forms (including external gills, delayed development of the jaw, cheek elements, and dermal armor, and a lack of ossification among some bones). They had specialized pharyngeal denticles that served as an aquatic filter-feeding device. Branchiosauridae are distributed around Europe in Carboniferous and Permian deposits (and perhaps lower Triassic). The group includes five genera. See Clack (2012), Schoch and Milner (2008), and Schoch (2014). Due to their obligate aquatic behavior, they are not included in the tally of Ark-borne animals.

Fig. 18. Branchiosaurus salamandroides. Retrieved from http://en.wikipedia.org/wiki/Branchiosauridae.
Family Micromelerpetontidae
Micromelerpetontids are very similar to the branchiosaurs described above. They are small (10–30 cm [3.9–11.8 in]), fully aquatic, and likely neotenic temnospondyls. Micromelerpetontidae includes four genera which are known from Permian deposits of Germany. See Schoch (2014). They are likewise not included in the tally of Ark-borne animals.
Family Amphibamidae
Amphibamidae were small (5–15 cm [1.9–5.9 in]) armorless temnospondyls with a salamander-like body, long and slim limbs, shortened trunks. Some genera were apparently sexually dimorphic (e.g., Micropholis; see Schoch and Rubidge 2005). The skull is broad with large, anteriorly placed eyes. Larval forms are known from this group. Their diet probably consisted of small insects. They are distributed in North America, Europe, and South Africa in upper Carboniferous through lower Triassic deposits. The group includes eleven genera. See Clack (2012) and Schoch (2014) for a review.

Fig. 19. Amphibamus grandiceps. Retrieved from http://en.wikipedia.org/wiki/Amphibamus.
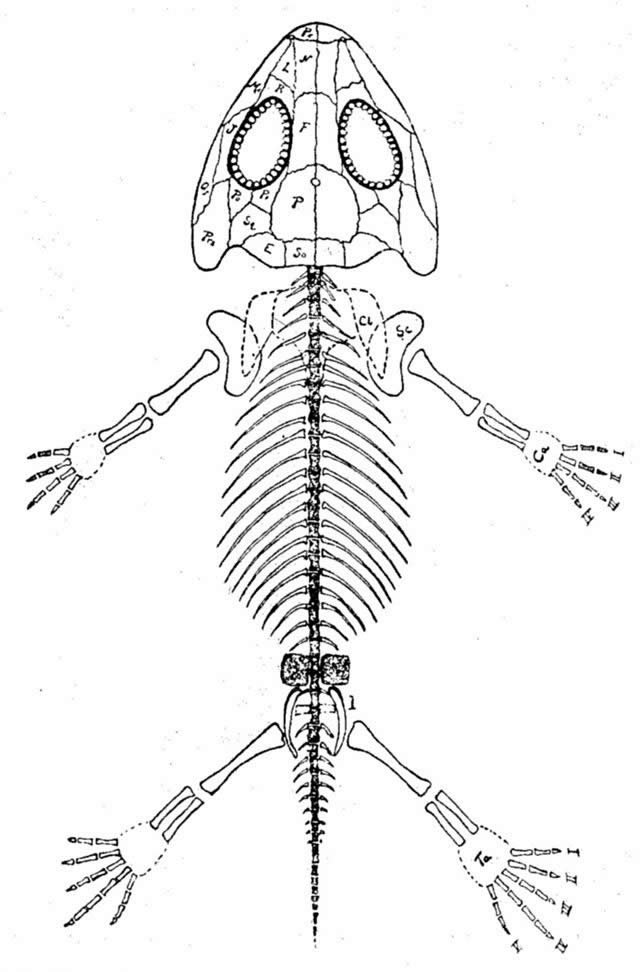
Fig. 20. Amphibamus grandiceps skeleton. Retrieved from http://en.wikipedia.org/wiki/Amphibamus.
Clade Eryopiformes

Fig. 21. Skeleton of Eryops on display at the Field Museum of Natural History. Photograph: M. Ross.
Family Eryopidae
Eryopidae were medium to large piscivorous temnospondyls up to 2 m (6.5 ft) in length. Characterized by a long, low face with large nostrils, stout ribs and strong hips, the group includes the well-known Eryops (often illustrated in books on fossils, usually with the synapsid Dimetrodon lurking nearby). Fossils of young organisms tentatively assigned to eryopids indicate that members of this group passed through an aquatic “tadpole” stage, while adults were semiaquatic. The group includes four genera known from Permian deposits in North America and Europe. See Sawin (1941).
Family Parioxydae
A monogeneric family consisting of Parioxys, known from several partial skulls and skeletons from upper Permian deposits of Texas. Vertebrae near the sacral region possess lateral expansions, similar to those in Eryops (but not seen among other dissorophid temnospondyls). Individuals were approximately 1 m (2.3 ft) long. See Carrol (1964) and Moustafa (1955).
Clade Stereospondylmorpha
Family Archegosauridae
Archegosaurs were semi-aquatic temnospondyls. They had long snouts and were the largest known “amphibian” group, ranging from 2–9 m (6.5–29.5 ft) as adults. Their lifestyle may have been similar to that of crocodilians, with a diet that consisted of fish and other small amphibians. The group includes the 9 m (29.5 ft) long Prionosuchus, the largest “amphibian” ever discovered. The group includes six genera from Permian deposits throughout Europe. See Gubin (1997).
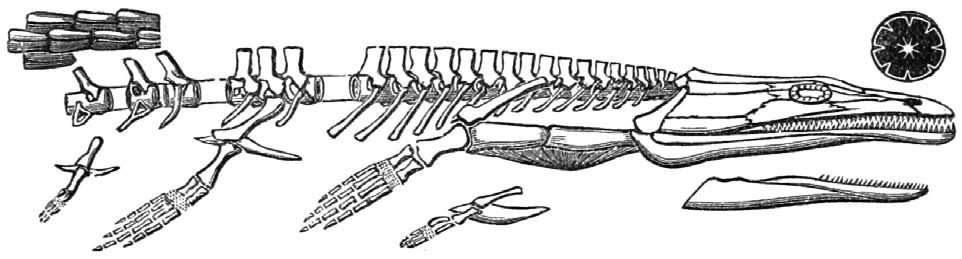
Fig. 22. Archegosaurus. Retrieved from http://commons.wikimedia.org/wiki/Category:Archegosaurus.

Fig. 23. Archegosaurus decheni. Retrieved from http://en.wikipedia.org/wiki/Archegosaurus.
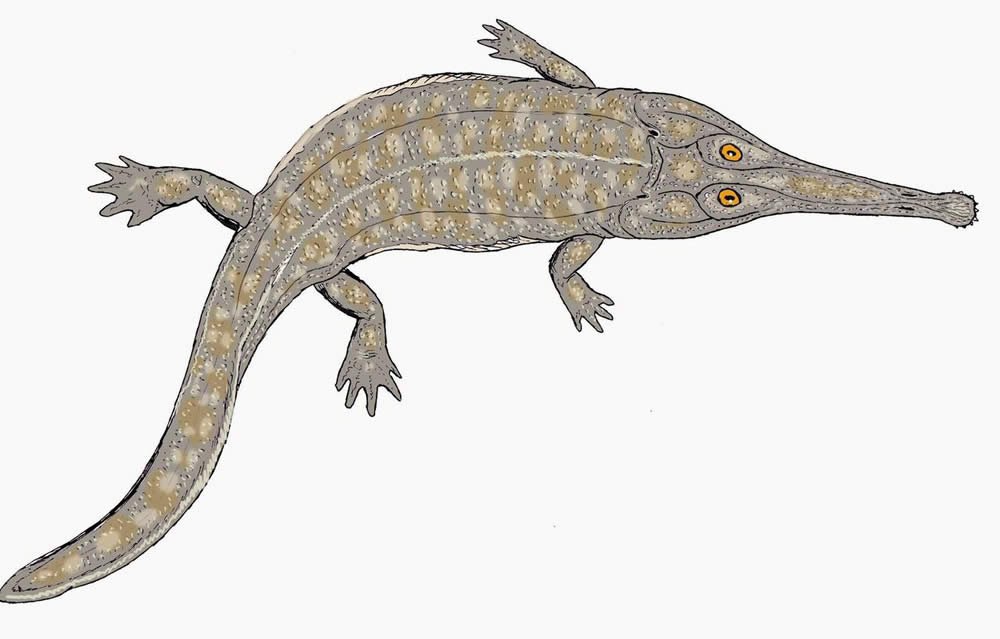
Fig. 24. Australerpeton. Retrieved from http://en.wikipedia.org/wiki/Australerpeton.

Fig. 25. Collidosuchus. Retrieved from http://en.wikipedia.org/wiki/Collidosuchus.

Fig. 26. Platyposaurus stuckenbergi. Retrieved from http://en.wikipedia.org/wiki/Platyoposaurus.
Family Melosauridae
Melosauridae were large (2–3 m [6.5–9.8 ft]) aquatic temnospondyls with elongate, alligator-like skulls. This family contains two genera known from Permian deposits of Russia. See Shiskin, Novikov, and Gubin (2000).

Fig. 27. Melosaurus. Retrieved from http://en.wikipedia. org/wiki/Melosaurus.
Family Intasuchidae
Intasuchidae were carnivorous temnospondyls which possessed a triangular skull that narrowed anteriorly and is characterized by a pair of well-developed preorbital ridges. They possessed numerous teeth, including small fangs on their palatines and vomers (bones that form the roof of the mouth). This family contains two genera from upper Permian deposits of Russia. See Shiskin, Novikov, and Gubin (2000).
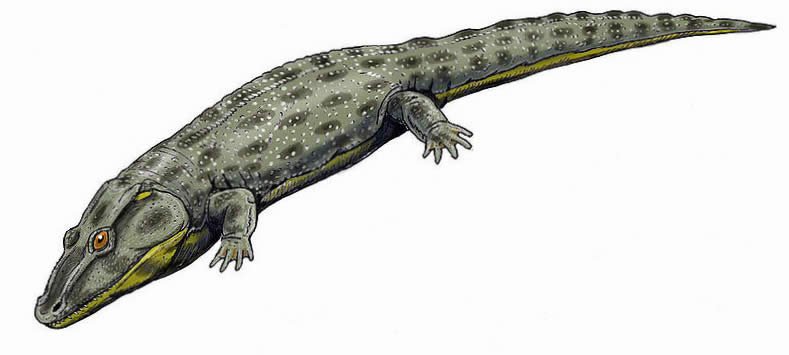
Fig. 28. Intasuchus. Retrieved from http://en.wikipedia. org/wiki/Intasuchus.
Suborder Stereospondyli

Fig. 29. Lapillopsis nana. Retrieved from http:// en.wikipedia.org/wiki/Lapillopsidae.
Family Rhinesuchidae
Rhinesuchids were semi- to completely aquatic temnospondyls. They were large in size (2–4 m [6.5–13.1 ft]), had large flat heads, with small upward-facing eyes toward the rear of the skull. The mouth had numerous tiny teeth located on the palatines. The group includes six genera found in upper Permian and lower Triassic deposits from South Africa. See Damaini and Rubidge (2003).
Family Lapillopsidae
Lapillopsidae consists of small (<10 cm [3.9 in]) semiterrestrial temnospondyls. Three genera are known from lower Triassic deposits of Australia and India. See Yates (1999, 2002).

Fig. 30. Capitosaurus. Retrieved from http://en.wikipedia. org/wiki/Capitosaurus.

Fig. 31. Capitosaurus. Retrieved from http://en.wikipedia. org/wiki/Capitosaurus.
Superfamily Capitosauroidea
Family Capitosauridae
Semi- to fully aquatic temnospondyls with massive, flat heads, long snouts and posteriorly placed eyes. Their overall length could range from 3–5 m (9.8–16.4 ft). Capitosaurs are distributed globally (Europe, Portugal, Australia, South Africa, Greenland, and North America) in Triassic deposits. The group contains six genera. See Fortuny, Galobart, and De Santisteban (2011). Due to some members’ obligate aquatic behavior, they are not included in the tally of Ark-borne animals.
Family Mastodonsauridae
Mastodonsaurs were large temnospondyls with broad, flat heads and somewhat shortened legs. Adults reached up to 6 m (19.6 ft) long and could weigh 200–500 kg (440.9–1102.3 lb). Notably, they possessed enlarged teeth on the lower jaw that actually protruded through holes in the bones of the upper snout. They are distributed across the globe (North America, Greenland, Europe, Asia, and Australia) in upper Triassic deposits. The group is a combination of three former families (Cyclotosauridae, Paracyclotosauridae, and Wetlugasauridae) and contains six genera from North America, Europe, Asia, and Australia. See Damaini (2001). Due to some members’ obligate aquatic behavior, they are not included in the tally of Ark-borne animals.

Fig. 32. Mastodonsaurus. Retrieved from http://de.wikipedia.org/wiki/Mastodonsaurus.
Family Sclerothoracidae
Sclerothoracidae is a monospecific family comprised of Sclerothorax hypselonotus, Schlerothorax is characterized by body proportions typical of a primarily terrestrial animal, though it possessed a lateral-line system for sensing movement in the water. Sclerothorax is distinguished by its flat and broadly U-shaped head. It was about 1m (3.2 ft) long and possessed elongate neural spines and a series of subdermal plates that armored its back. Specimens of Sclerothorax are known from Triassic deposits of Germany. See Schoch et al. (2007)
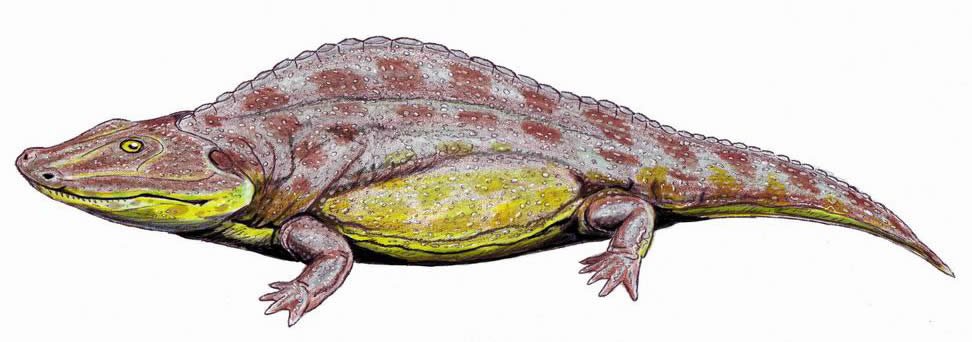
Fig. 33. Mastodonsaurus. Retrieved from http://en.wikipedia.org/wiki/Sclerothorax.
Clade Trematosauroidea
Family Brachyopidae
Brachyopidae were large temnospondyls with broad, flat skulls and somewhat reduced limbs. These were highly aquatic carnivores which included one of the largest “amphibians” known (approximately 7 m [22.9 ft] long). This family contains 15 genera known from Permian through Jurassic deposits throughout much of the world, including Asia, Africa, Eastern Europe, and Australia. See Warren and Marsicano (2000).
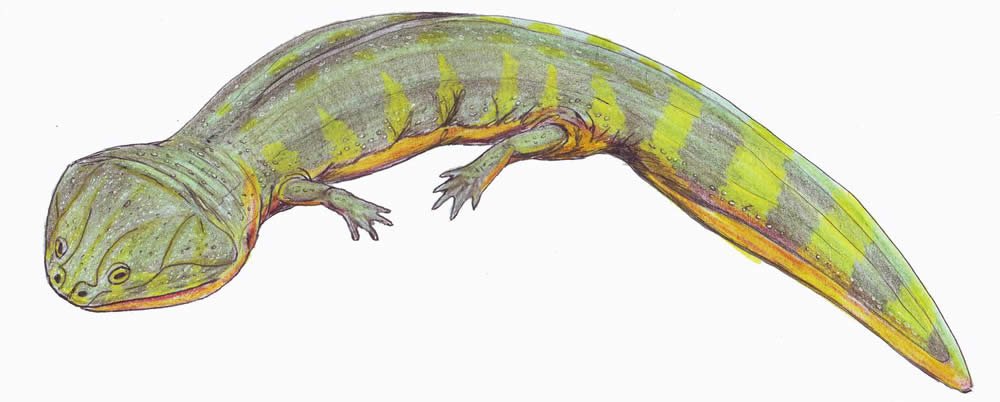
Fig. 34. Batrachosuchus. Retrieved from http://en.wikipedia.org/wiki/Batrachosuchus.
Family Plagiosauridae
Plagiosauridae were fully aquatic temnospondyls which retained their gills as adults, and were quite similar to Laidleria (above). Their heads were very wide and flat, and adult sizes reached approximately 1 m (3.2 ft) long. The family contains 11 genera from Triassic deposits of Europe, Australia, and Brazil. See Milner (1994). Due to their fully aquatic behavior, they are not included in the tally of Ark-borne animals.

Fig. 35. Gerrothorax pustuloglomeratus. Retrieved from http://en.wikipedia.org/wiki/Gerrothorax.
Family Chigutisauridae
Chigutisauridae are a very similar group to the brachyopoids (above). They were broad-headed, salamander-like predators that consumed prey by suction-gulping. One notable genus is the stratigraphically highest of all temnospondyls, Koolasuchus, a massive (5 m [16.4 ft]) salamander-like chigutisaur from Australia. Fossils are known from lower Triassic through lower Cretaceous deposits of Australia, Argentina, India, and South Africa. This family contains six genera. Warren, Damiani, and Yates (2001).
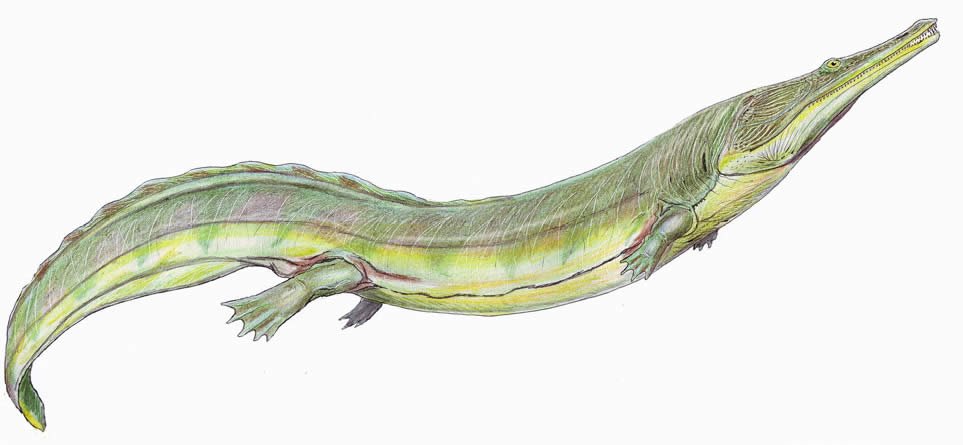
Fig. 36. Trematosaurus. Retrieved from http:// en.wikipedia.org/wiki/Trematosaurus.

Fig. 37. Trematosaurus brauni. Retrieved from http:// en.wikipedia.org/wiki/Trematosaurus.
Superfamily Trematosauroidea
Family Trematosauridae
Some of these fully marine amphibians had extremely long, slender snouts with the nostrils located midway on the snout and posteriorly placed eyes. Their bodies were crocodile-like and adults measured 3–4 m (9.8–13.1 ft) in length. Trematosaurs include six genera from Triassic deposits in Algeria, Germany, Russia, and the United States. See Steyer (2002) and Schoch (2014). Due to some members’ obligate aquatic behavior, they are not included in the tally of Ark-borne animals.
Family Metoposauridae
Metoposaurs are a group of relatively large (up to 3 m [9.8 ft]) and primarily aquatic predators. The skull is long, flat, and U-shaped, the orbits oriented anterodorsally, and the group likely fed by suction gulping. Limbs are reduced in size. The group includes five genera known from Triassic and Jurassic deposits of Europe, Asia, Africa, and USA. See Clack (2012). Due to some members’ aquatic behavior, they are not included in the tally of Ark-borne animals.
Family Laidleriidae
A monspecific family consisting of Laidleria gracilis. They were small (< 1 m [3.2 ft]) and were characterized by a flattened, nearly equilateral, triangular skull with laterally projecting posterior corners, pitted dermal sculpturing, very small teeth, and orbits positioned close to the skull margin. They are known from a Permian deposit in Uruguay. See Warren (1998).
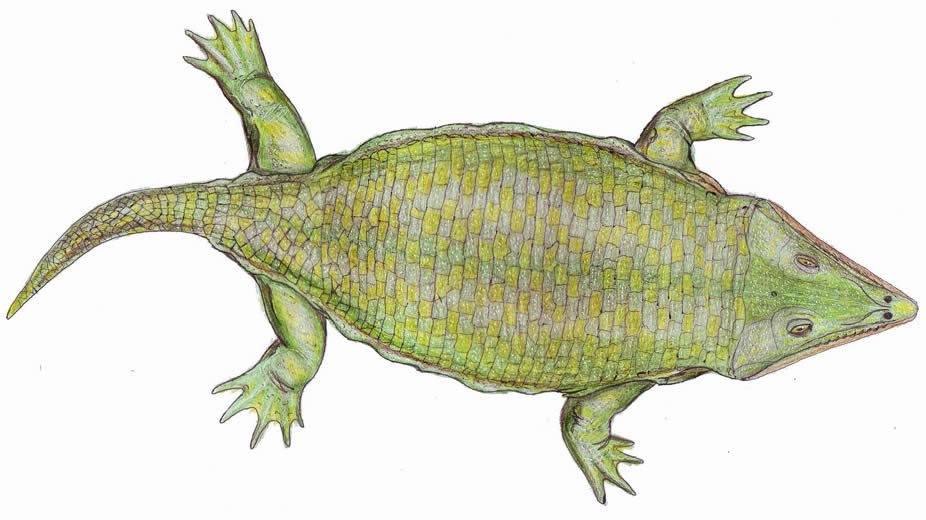
Fig. 38. Laidleria gracilis. Retrieved from http:// en.wikipedia.org/wiki/Laidleria.
Infraclass Lissamphibia
The Lissamphibia includes the three living orders of amphibians: Anura (frogs/toads), Caudata (newts/salamanders), and Gymnophiona (caecilians). For descriptions of their characters and biology, see Hennigan (2013 a, b). The fossil record of the Lissamphibia also includes a fourth order, Allocaudata, which is represented by one family of scaled salamanders, the albanerpetontids. There are also two families of extinct anurans. While there are many additional fossils of lissamphibians, most of them either fall into extant families or cannot be determined to genus or family rank.
Order Allocaudata
The Allocaudata (“other salamanders”) is an order-level designation consisting of one family, the Albanerpetontidae.

Fig. 39. Albanerpeton. Retrieved from http://en.wikipedia. org/wiki/Albanerpeton.
Family Albanerpetontidae
Albanerpetontids were very small (<10 cm [3.9 in]), salamander-like, carnivorous lissamphibians thought to live in forest or grassland habitats. Once thought to be true salamanders, they are characterized by several distinct skeletal features, particularly their chisel-shaped teeth, modified cervical vertebrae, and the possession of numerous scale-like denticles on their skin. They are distributed primarily in Laurasian continents (N. America, Europe, and Asia, but also Morocco), in Middle Jurassic to Pliocene deposits, with the bulk known from Cretaceous deposits. Albanerpetontidae includes four genera. See Gardner and Bohme (2008) and Schoch (2014).
Order Anura
Anurans are frogs, toads, and other frog-like amphibians with short bodies and mostly tailless adult forms. Fossil anurans are first seen in Triassic deposits, though some evolutionary paleontologists believe that the record could extend to the Permian. They have a stout body, protruding eyes, glandular skin, and legs tucked underneath the body. They are generally semi-aquatic, and most spend part of their life cycle in water.
Family Gobiatidae
Gobiatids are an extinct family of frogs. They are distinguished by characteristics of their vertebrae and maxillae. Fossils have been located in Texas, U.S., Uzbekistan, and Mongolia, in upper Cretaceous strata. The name was derived from the Gobi Desert of Mongolia, in which fossils were first found. There are three genera. See Roček (2008).

Fig. 40. Triadobatrachus. Retrieved from http://en.wikipedia.org/wiki/Triadobatrachus.
Family Triadobatrachidae
This monospecific family consists of Triadobatrachus massinoti. The species was about 10 cm (3.9 in) long, carnivorous, and likely semiaquatic. It is the stratigraphically lowest frog in fossil record, and has been found only in northern Madagascar in lower Triassic marine deposits. It is proposed as a transitional form because of its short tail and possession of comparably more vertebrae than modern frogs. See Rage and Roček (1989).
Infraclass Lepospondyli
The lepospondyls are a large and diverse group comprised of four orders: Adelospondyli, Lysorophia, Microsauria, Aïstopoda, and Nectridia. These groups share several anatomic features, including a reduced number of skull bones, more numerous vertebrae compared to other “amphibian” groups, vertebrae composed of a single central element (the centrum), which is fused to the neural arch, and more rounded ribs capable of flexing, which likely permitted costal ventilation. Juvenile fossils from various species display direct development rather than undergoing a metamorphosis from a morphologically distinct larval form. See Carroll (1988), Clack (2012), and Schoch (2014) for a review of the group and its major divisions.
Order Lysorophia
Family Lysorophidae
Lysorophidae is a family group of burrowing amphibians resembling small snakes. They have a lightly-built skull with a fused braincase, very large orbits, an exceptionally elongate body with up to 100 presacral vertebrae, reduced or absent limbs, and were capable of estivating (a state of dormancy typically used to protect from desiccation). Lysorophids are known from Pennsylvanian and Permian deposits of North America and England. This group contains six genera. See Clack (2012) and Olson (1971).
Order Microsauria

Fig. 41. Brachydectes. Retrieved from http://en.wikipedia.org/wiki/Brachydectes.
The most diverse group of lepospondyls is the microsaurs. They include 12 families divided into two suborders, the microbrachomorphs and the tuditanomorphs, based on differences in the construction of the skull roof. All microsaurs share a unique occipital condyle-atlas-axis complex, retained their limbs (reduced in some forms), and most were lizard-like or salamander-like in general appearance. The group is considered paraphyletic by some authors, as recent cladistic analyses include some of the lepospondyl families within the traditional Microsauria. Some appear to have been burrowers. Microsaurs are known from upper Mississippian through upper Permian deposits in Scotland, Germany, Russia, the United States, and Nova Scotia, Canada. See Carroll (1988) and Clack (2012) for an overview of Microsauria. For detailed descriptions, see the exceptional descriptive paper by Carroll and Gaskill (1978).
Family Brachystelechidae
The brachystelechids are a family of small (<20 cm [7.8 in]) burrowing microsaurs. The body is overall lizard-like. The three genera of the family are known from Permian deposits of Germany and Texas. See Carroll and Gaskill (1978).

Fig. 42. Hyloplesion. Retrieved from http://en.wikipedia. org/wiki/Hyloplesion.
Family Hyloplesiontidae
Hyloplesion is the only genus in the Hyloplesiontidae. It is an extinct salamander-like animal whose overall length was less than 15 cm (5.9 in). Numerous well-preserved specimens provide much anatomic data, though it is unclear if Hyloplesion was aquatic or terrestrial; it may have been comfortable in either habitat, as in modern salamanders. Specimens are known from middle Pennsylvanian deposits of the Czech Republic. See Carroll and Gaskill (1978).
Family Microbrachidae
Microbrachidae is an extinct monospecific family from the Carboniferous period. Microbrachis resembled a salamander about 15 cm (5.9 in) long. It had small limbs and used lateral body movement to move. It preserved its larval gills through adulthood and likely ate freshwater plankton. Microbrachis is known from middle Pennsylvanian deposits of the Czech Republic. See Carroll and Gaskill (1978) and Milner (1980b).
Family Odonterpetontidae
This is a monospecific family consisting of the very small (perhaps 5 cm [1.9 in]; the specimen is incomplete) Odonterpeton triangularis, known from a well-preserved torso and skull. This species of microsaur had anteriorly placed eyes, probably external gills as an adult, and very small front limbs. Carroll and Gaskill (1978) consider the animal obligately aquatic, and it is therefore excluded from the tally of Ark-borne organisms.

Fig. 43. Microbrachis. Retrieved from http://en.wikipedia. org/wiki/Microbrachis.
Suborder Tuditanomorpha
Family Gymnarthridae
Gymnarthridae was a family of small terrestrial and semiaquatic amphibians with four short limbs and elongated bodies. The jaw of their small and simple skull is lined with a single row of proportionally large conical teeth. Fossils have been discovered in the Czech Republic, Nova Scotia, and in the United States from Pennsylvanian to lower Permian deposits. This group includes ten genera. See Carroll and Gaskill (1978).

Fig. 44. Cardiocephalus. Retrieved from http:// en.wikipedia.org/wiki/Cardiocephalus.
Family Hapsidopareiontidae
Haspidopareiontidae is a family of small (<10 cm [3.9 in]) terrestrial microsaurs characterized by a lizard-like body, pitting of the skull roof bones, and the presence of a large temporal embayment in the cheek region, the function of which remains unclear. They are distributed in the United States in lower Permian deposits. This family includes two genera. See Bolt and Rieppel (2009) and Carroll and Gaskill (1978).

Fig. 45. Llistrofus. Retrieved from http://en.wikipedia. org/wiki/Llistrofus.
Family Ostodolepidae
Members of Ostodolepidae were terrestrial burrowing microsaurs less than 1 m (3.2 ft) in length. They possessed an elongate body, short and stubby limbs, a sharp snout, and a short tail. They are found in Early Permian deposits primarily from the central United States, with one specimen found in Germany. The group contains five genera. See Anderson, Scott, and Reisz (2009), Carroll and Gaskill (1978), and Henrici et al. (2011)
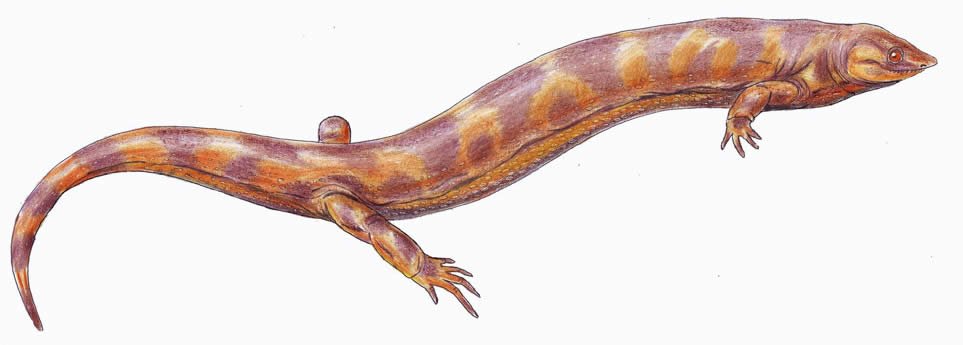
Fig. 46. Pelodosotis. Retrieved from http://en.wikipedia.org/wiki/Pelodosotis.
Family Pantylidae
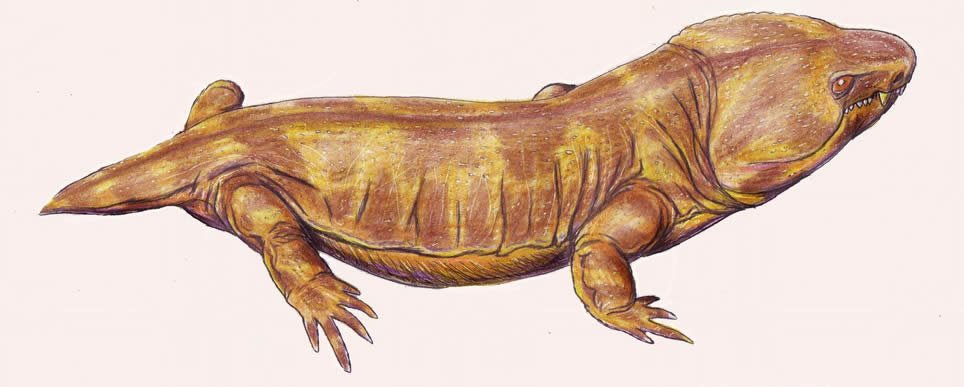
Fig. 47. Pantylus. Retrieved from http://en.wikipedia.org/wiki/Pantylus.

Fig. 48. Pantylus cordatus skull. Retrieved from http://en.wikipedia.org/wiki/Pantylus.
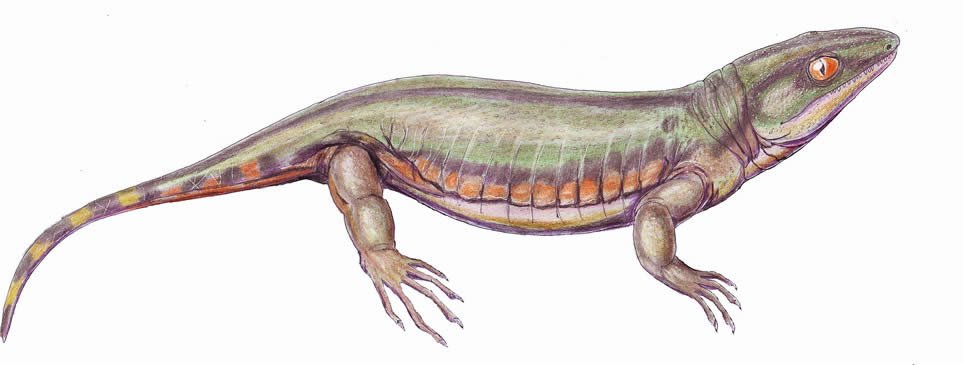
Fig. 49. Tuditanus. Retrieved from http://en.wikipedia.org/wiki/Tuditanus.
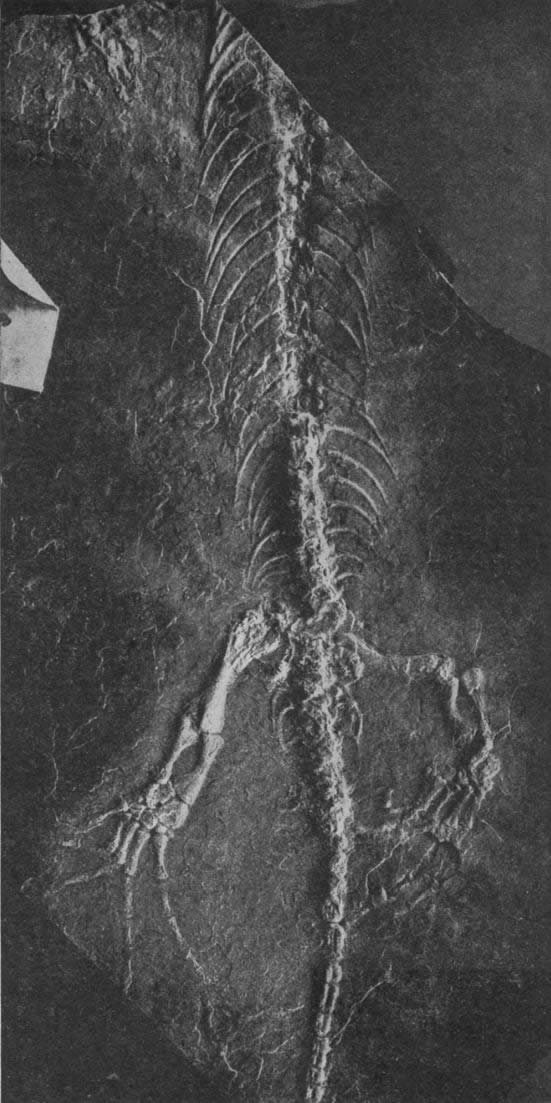
Fig. 50. Tuditanus punctulatus skeleton. Retrieved from http://commons.wikimedia.org/wiki.
A monospecific family consisting of Pantylus cordatus. Though comprised on only one species, P. cordatus it is the best known of all microsaurs, as it is represented by numerous partial and complete skeletons. P. cordatus was about 25 cm (9.8 in) in length and bore short, stocky limbs and a very small tail. The head was quite large, flat, and broadly triangular in shape with many blunt teeth in the marginal dentition as well as the bones of the upper palate, indicating a diet requiring the crushing of shelled invertebrates. Its shortened trunk has the fewest number of presacral vertebrae (24) among all microsaurs. P. cordatus is known from Permian deposits in Texas. See Carroll and Gaskill (1978).
Family Trihecatontidae
This is a monospecific family consisting of Trihecatontidae howardinus. The only specimen (a partial skeleton and lower jaws) was found in the upper Pennsylvanian of Colorado. The animal was less than 50 cm (19.6 in) in total length, had an elongate body and somewhat reduced limbs. See Carroll and Gaskill (1978).
Family Tuditanidae
Less elongate than other microsaurs, the Tuditanidae were medium-sized for the order. They were terrestrial and bore a superficial resemblance to lizards. All four limbs were well-developed. They fed on various invertebrates and arthropods. Specimens from lower to upper Pennsylvanian deposits have been found in Nova Scotia, Ohio, the Czech Republic. The family contains three genera. See Carroll and Gaskill (1978).
Order Nectridea
The nectrideans were dominated by fully aquatic forms which lived in lakes and streams. The three families are united by the possession of a number of common skeletal structures, including a relatively compact truck and an elongated tail with symmetrically-sized neural and haemal arches, both of which are wedge- or hatchet-shaped. They include perhaps the most unusual of all “amphibians”, the boomerang-headed diplocaulids. See Carroll (1988) and Clack (2012) for an overview of the Nectridea.
Family Diplocaulidae (=Keraterpetontidae)

Fig. 51. The skull of Diplocaulus magnicornis, on display at the Field Museum of Natural History. Photograph: M. Ross.
Diplocaulidae consists of fully aquatic amphibians 30 cm–1.5 m (11.8–4.9 ft) in length and exhibit a relatively large amount of variation in tail length and limb strength. Some genera, particularly Diplocaulus and Diploceraspis had a unique, virtually immobile arrowhead- or boomerang-shaped skull and possessed external gills. Resting impressions indicate that the wings of the skull were attached to the body by skin flaps that may have served as a hydrofoil to aid in swimming. The diplocaulids are known from Pennsylvanian and Permian deposits in North America and Africa. The family contains seven genera. See Milner (1980a). Due to the fully aquatic behavior of members of this family, they are excluded from the tally of Ark-borne animals.

Fig. 52. Scincosaurus. Retrieved from http:// en.wikipedia.org/wiki/Scincosaurus.
Family Scincosauridae
This is a monospecific family consisting of Scincosaurus crassus, which had highly developed limbs and, unlike other nectrideans, is believed to be primarily terrestial S. crassus is known from upper Pennsylvanian deposits of France and the Czech Republic. See Ruta, Coates, and Quicke (2003) and Clack (2012).
Family Urocordylidae
Urocordylidae includes both aquatic and terrestrial forms. The tail was very long (about twice the snout-vent length) and powerful for swimming, but the well-developed limbs allowed them to move about more freely on the ground. Skulls tend to be elongate and show evidence of kinesis. They are known from the Carboniferous to the early Permian of Oklahoma, Texas, Arizona, and Alabama. The family contains five genera. See Carlson (1999).

Fig. 53. Sauropleura. Retrieved from http://en.wikipedia. org/wiki/Sauropleura.
Order Aïstopoda
The Aïstopoda consists of a group of completely limbless “amphibians.” While a number of excellent specimens exist, it has not been determined if their snake-like forms were indicative of terrestrial or aquatic habitats. The heads are characterized by rather lightweight construction, strong jaws, and orbits positioned in the front half of the skull. They typically possess nearly 100 vertebrae, with some forms having nearly 250. There are no pectoral or pelvic girdle elements. The stratigraphically lowest member (Lethiscus, middle Mississippian) already displays all of the defining physical characters of the group, and is among the earliest of the post-Devonian tetrapods. Indeed, the term “tetrapod” here is admittedly a strange designation, since aïstopods possess no limbs. Other members appear in the stratigraphically higher rocks of the Pennsylvanian (upper Carboniferous). Fossils of the group are known in North America and Europe. See Clack (2012) and Schoch (2014) for a review of this group.
Family Lethiscidae

Fig. 54. Oestocephalus. Retrieved from http://en.wikipedia.org/wiki/Oestocephalus.
Lethiscidae consists of a single genus (Lethiscus), an elongate and limbless aïstopod with a distinctly snake-like appearance. This is the earliest known record of the Aïstopoda, and such a highly specialized organism is surprising under evolutionary expectations of early tetrapods. Lethiscus has a light skull, with the anteriorly place eyes and about thirty closely spaced teeth on each maxilla and dentary. Lethiscus ranged in size from 50 cm to 1 m (19.6–39 in) in length, and it is unclear if the animal was aquatic or terrestrial. Fossils are known from middle Mississippian deposits of Scotland. See Anderson, Carroll, and Rowe (2003) and Carroll (1988).
Family Oestocephalidae
Oestocephalidae consists of a single genus, Oestocephalus, which had a robust skull and a narrow, round snout. The cheeks were covered by a series of small plates, which provided room for very large jaw muscles. It had ossified dermal bones and dorsal scales. Oestocephalus possessed up to 100 vertebra and lacked limbs and girdles. It may have been semiaquatic. Fossils are known from a wide range of localities, including upper Carboniferous (middle and upper Pennsylvanian) deposits in Ohio, Illinois, Colorado, the Czech Republic, and England. See Anderson (2003) and Carroll et al. (1998).
Family Ophiderpetontidae

Fig. 55. Ophiderpeton. Retrieved from http://en.wikipedia.org/wiki/Ophiderpeton.
Ophiderpetontids have a snake-like form and their vertebral column contains up to 100 vertebrae. They displayed blunt snouts and eyes that positioned anteriorly on the skull. Ophiderpetontids were less than 1 m (3.2 ft) long, limbless and may have been either terrestrial or aquatic. The family contains two genera found in Pennsylvanian deposits from Arizona, Colorado, and the United Kingdom. See Anderson, Carroll, and Rowe (2003) and Milner (1994).
Family Phlegethontiidae
This family contains one genus, Phlegethontia, an elongate, limbless aïstopod with up to 250 vertebrae. It is known from lower Pennsylvanian through Permian deposits in the Illinois, Ohio, and Texas, and the Czech Republic. See Anderson (2002a, 2007).
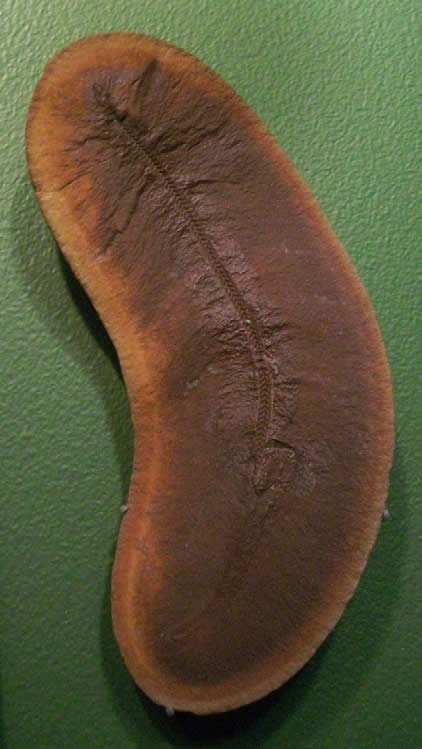
Fig. 56. Pseudophlegethontia turnbullorum. Retrieved from http://en.wikipedia.org/wiki/Pseudophlegethontia.
Family Pseudophlegethontiidae
This family contains one genus, Pseudophlegethontia, which was an elongate, limbless aïstopod. Pseudophlegethontia possessed unusual, K-shaped ribs, which may have aided in “rib crawling,” a behavior seen in some snakes. Pseudophlegethontia was less than 1 m (3.2 ft) long, and is known from the middle Pennsylvanian Mazon Creek deposits in Illinois. See Anderson (2002b).
Order Incertae sedis
Two “amphibian” families of uncertain relationships are the Adelogyrinidae and the Archerontiscidae. They are here placed within Lepospondyla following previous conventions (e.g., Carroll 1988), though their placement within the Lepospondyla and/or with respect to Lepospondyla (particularly the Archerontiscidae) may be questioned.
Family Adelogyrinidae
The Adelogyrinidae is a family of aquatic “amphibians.” The orbits are located anteriorly on the skull, which was roofed by dermal armor and likely capable of a wide gape for suction feeding. The shoulder girdle is large, but fore limbs have been preserved (they may have been cartilaginous). Hind limbs are apparently absent in the group. Overall size is likely about 0.5 m (1.6 ft), though none of the specimens are complete. Four genera are known from lower Carboniferous (middle Mississippian) deposits in Scotland, though the genera may reflect different components and/or orientations of only one genus. Due to their aquatic nature, they are not included in the tally of Ark-borne animals. See Clack (2012) for an overview of the group.
Family Acherontiscidae
A monospecific family composed of Acheroniscus caledoniae. The skull of Acheroniscus has dermal armor and displays lateral line grooves, indicating an aquatic habitat. Like the adelogyrnids above, shoulder girdle elements are present, but no forelimbs. Hind limbs are also absent, but this may be due to poor preservation. The vertebrae were embolomerous (both the pleurocentra and intercentra are present and display a hole through which the notochord passed). Total length is approximately 15 cm (5.9 in) or less. The single specimen was recovered from lower Carboniferous (middle Mississippian) deposits in Scotland. Due to its aquatic nature, A. caledoniae is not included in the tally of Ark-borne animals. See Carroll (1969) for the original description.
Infraclass Reptilomorpha
Order Anthracosauria
The anthracosaurs are a group of small to medium-sized, roughly crocodile-proportioned “amphibians.” Two particular traits unify the group: a sutured connection of the parietal and tabular bones of the skull and elongated ribs. Typically, the tabular bone extends posteriorly from the back of the skull, forming a “tabular horn.” Though primarily aquatic (most possessed the lateral line system), they were certainly capable of forays onto the land, as evidenced by their typically large and well-ossified pelvic girdle and hind limbs. Anthracosaur respiration was dominated by lungs, rather than gills or skin, since the skin was covered with bony denticles and skull bones associated with the possession of gills are unknown in the group. Anthracosaurs are considered by evolutionary paleontologists as the “amphibian” lineage leading to amniotes, and amniotes are therefore cladistically included under the clade Anthracosauria, with seymouriids (discussed below) straddling the line between the pseudoanamniotic and fully amniotic condition. Here I restrict the use of Anthracosauria to the anamniote members. More classically, the embolomeres (described below) make up the bulk of taxa commonly referred to as anthracosaurs. Fossils of this group are known from upper Mississippian through Triassic deposits in North America, Europe, and Asia. See Clack (2012) and Schoch (2014) for a review of this group.
Family Caerorhachidae
Contains one genus, Caerorhachis, a terrestrial “amphibian” with an overall length less than 50 cm (19.6 in) long. Its taxonomic position is somewhat unsettled, as Carroll (1988) places it within the temnospondyls, whereas Ruta, Coates, and Quicke (2003; see fig. 10) place it as a basal member just outside of the Anthracosauria proper. See Ruta, Milner, and Coates (2001) for the most recent redescription of the sole specimen from lower Carboniferous (upper Mississippian) deposits of Scotland.
Suborder Embolomeri
This group includes medium-sized semiaquatic predatory amphibians that make up the bulk of the anthracosaurs as typically discussed. They possessed massive skulls (45cm long, 25cm wide), a flared cheek region, large fluted teeth, and enlarged palatal tusks. Their bodies were elongate (up to 4 m [13.1 ft] long), with a long tail for propulsion. They have been found in the eastern United States, the United Kingdom, eastern Europe, Russia, and Tajikistan in Carboniferous and Permian deposits. The Embolomeri includes 12 families. See Schoch (2014) for a review.
Family Eoherpetontidae
This is a monospecific family consisting of Eoherpeton watsoni, a terrestrial embolomere that lacks traces of the lateral line system along its skull and possesses rather long limbs. E. watsoni’s length was approximately 1 m (3.2 ft). This is one of the stratigraphically lowest occurrences of embolomeres, known only from Mississippian deposits of England. See Panchen (1975) and Smithson (1985).
Family Proterogyrinidae

Fig. 57. Proterogyrinus. Retrieved from http://en.wikipedia.org/wiki/Proterogyrinus.
The Proterogyrinidae is consists of the genus Proterogyrinus and possibly Papposaurus. Proterogyrinus is known from multiple specimens, and was a semiterrestrial embolomere about 1 m (3.2 ft) long. Among the stratigraphically lowest occurences of the embolomeres, proterogyrinids are known from the upper Mississippian of West Virginia (USA) and Scotland. See Holmes (1984).
Family Eogyrinidae
The Eogyrinidae is relatively diverse family characterized by strongly aquatic features (very elongate bodies, lateral line system, and very reduced limbs) were some of the largest of the embolomeres, with some reaching 4 m (13.1 ft) in length. Skulls were powerfully constructed with large palatal fangs. One small skull of the genus Calligenethlon may represent a juvenile. If so, then it appears that it (and likely most embolomeres) did not undergo substantial morphological changes from young to adult, unlike many temnospondyls and modern lissamphibians. Eight genera are known from Pennsylvanian deposits of Nova Scotia. See Panchen (1972) for a description of Pholiderpeton (=Eogyrinus), the best-known genus of this family.
Family Archeriidae
The Archeriidae is family of relatively large (up to 2 m [6.5 ft]), primarily aquatic embolomeres. Their skulls were elongate and somewhat crocodile-like, with numerous teeth indicating piscivory, and their bodies and tail were long compared to limb lengths. The best-known genus, Archeria, had expanded neural spines and haemal arches on the tail vertebrae to support an expanded fin. Three genera are known from Pennsylvanian deposits in Illinois and Permian deposits in Oklahoma and Texas. See Romer (1957) and especially Holmes (1989).

Fig. 58. Cricotus. Retrieved from http://en.wikipedia.org/wiki/Cricotus.
Family Anthracosauridae

Fig. 59. Anthracosaurus russeli. Retrieved from http://en.wikipedia.org/wiki/Anthracosaurus.
This is a monogeneric family consisting of Anthracosaurus, a large (likely 3 m [9.8 ft]) embolomere known primarily from skull material. The skull is large and solidly constructed, and Anthracosaurus possessed a reduced number of greatly enlarged teeth. Fossils are known from upper Carboniferous (lower Pennsylvanian) deposits of England. See Clack (1987) for a description.
Suborder Seymouriamorpha
Seymouriamorpha includes six families: Discosauriscidae Gephyrostegidae, Karpinskiosauridae, Kotlassiidae, Seymouriidae, and Waggoneriidae. The skulls of seymouriamorphs tend to be deeper and wider than anthracosaurs. Seymouriamorphs fall into either largely terrestrial (Seymouriidae) or largely aquatic adult forms (all other families, but dominated by the Discosauriscidae), though similar gill-bearing larval stages are known from multiple genera across the Seymouriamorpha, so all members included at least a temporary aquatic mode of life. However, these larval forms were effectively small versions of the adult form; they do not appear to have undergone a dramatic metamorphosis from a “tadpole” stage. See Clack (2012) and Schoch (2014) for a review.
Family Waggoneriidae
Waggoneriidae is a monospecific family comprised of Waggoneria knoxensis, a semiaquatic predatory “amphibian” with deep jaws that held at least four rows of rather rounded, bulbous teeth which suggests they used them to crush hard-shelled prey. Remains are known from lower Permian deposits in Texas. See Olson (1951).
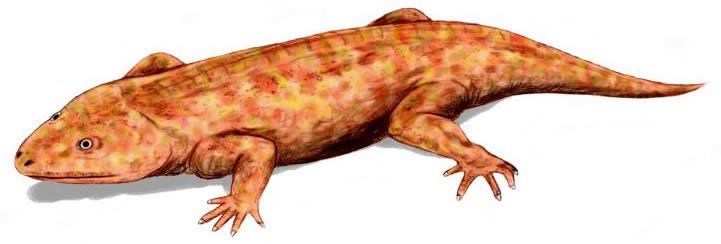
Fig. 60. Seymouria. Retrieved from http://fr.wikipedia. org/wiki/Seymouria.

Fig. 61. Seymouria. Retrieved from http://en.wikipedia. org/wiki/Seymouria.
Family Seymouriidae
A monogeneric family consisting of Seymouria, Seymouriidae is the most terrestrial family of anthracosaurs as determined to limb proportions and vertebral architecture (which display close similarities to basal amniotes). Larval forms displaying external gills are known from this group, so their life cycle included an aquatic early phase followed by a more fully terrestrial adult period, with eggs laid presumably in or very close to water. Adults were able to hold their body off of the ground at least temporarily, and Seymouria reached lengths of 0.5–1 m (1.6–3.2 ft). Seymouria is known Permian deposits of North America and Europe. See Clack (2012), Laurin (1996), and Schoch (2014).
Family Karpinskiosauridae
This is a monogeneric family consisting of Karpinskiosaurus. Several partial skulls and skeletons of this fairly small genus (<50 cm [19.6 in] long) are known, which displays orbits oriented anteriorly on the snout and a lack of lateral line grooves (indicating a more terrestrial mode of life). Fossils are known from upper Permian deposits in Russia. See Klembara (2011).
Family Gephyrostegidae
The Gephyrostegidae is a family of rather small (<20 cm [7.8 in]), primarily terrestial embolomeres. They have relatively long limbs compared to their overall trunk length, with proportions similar to lizards. They have very large orbits and numerous small teeth, indicating an insectivorous diet. Three genera are known from Pennsylvanian deposits in Ohio and the Czech Republic. See Boy and Bandel (1973) and Carroll (1970).
Family Discosauriscidae
A diverse family of small (10–20 cm [3.9–7.8 in]), rather salamander-like seymouriamorphs, discosauriscids possessed external gills and long tails. This family is best represented by numerous specimens in various growth stages of Discosauriscus. It had short limbs with a fairly long tail, wide jaws with small sharp teeth, and a lateral line system with rounded scales covering its skin. It is believed to retain gills through adult form and have matured fully in the water, though large adults may have terrestrial affinities (loss of lateral line and more robust limbs). This family contains five genera known from early Permian deposits in central and eastern Europe and Tajikistan. See Klembara (2009). Due to their apparently obligate aquatic behavior, this family is excluded from the tally of Ark-borne animals.
Family Kotlassidae
The kotlassids had rather stout, expanded ribs, and adult forms were approximately 1 m (3.2 ft) long and terrestrial. Four genera are known from Permian deposits of Russia. See Bystrow (1944).
Suborder Chroniosuchia
A distinctive but poorly understood group, the chroniosuchians are somewhat crocodile-shaped, with elongate snouts and long bodies between 0.5 and 1.5 m (1.6–4.9 ft) long. Their vertebrae display an unusual and characteristic ball-and-socket joint between the pleurocentrum and intercentrum, giving the trunk region a high degree of mobility. Yet that mobility was tempered by large osteoderms along the dorsal surface of their body. Their mode of life was at least partially (if not predominantly) terrestrial, given the absence of lateral line grooves, but their overall small limbs (of what little is known) makes this determination somewhat uncertain.
The position of the chroniosuchians within the anthracosaurs not fully agreed upon. They may be embolomeres, or perhaps are placed outside the embolomeres + seymouriamorphs. Two families are recognized, primarily on the basis of osteoderm morphology: the Chroniosuchidae (six genera) and the Bystrowianidae (five genera), known from Permian and Triassic deposits of Germany, Russia, and China. See Clack (2012) and Schoch (2014).
Conclusion
This review of fossil “amphibians” has described 69 families of extinct “amphibians” from eight orders. In total, this represents 268 genera. Of this number, 15 families are obligately aquatic, bringing the number of Ark-borne families to 54. While there are additional fossil “amphibian” genera that are not assigned to these or other families (primarily due to their placement in cladistic analyses), the groups reported here include all of the well-known families, as well as many poorly-understood and obscure groups, even among vertebrate paleontologists (e.g, 20 of the families discussed above are monogeneric and/or monospecific). As such, an estimate of 54 pairs of fossil “amphibians” represents a realistic number of animals brought on board the Ark if the taxonomic rank of family closely approximates the “kinds” brought to the Ark by God. These are added to the 140 extant anuran kinds, 53 caudate kinds, and one gymnophionan kind reported by Hennigan (2013a, b), to bring the total number of “amphibian” kinds to 248.
Acknowledgments
I am deeply indebted to the many colleagues and students who aided in this project. My thanks go first to the students of my 2012 Vertebrate Paleontology course (BIOL 371), whose writing projects on fossil vertebrate families form the backbone (so to speak) of this paper. In particular, I thank S. Beech for his initial work in collating the families, and D. Ehrich and B. Ray for aid in preparing this manuscript. I am grateful to Georgia Purdom, who invited me to join the Ark kinds team. The many helpful discussions with her, Jean Lightner, and Tom Hennigan helped bring focus to this first component of the vertebrate paleontology portion of the Ark kinds project. As editor of Answers Research Journal, Andrew Snelling has been both kindly encouraging and mercifully patient regarding the preparation, submission, and editing of this manuscript and in his role in overseeing the Ark kinds team.
Reviewers are often thanked with a passing mention, but the three individuals who reviewed this work provided thorough and challenging evaluations. Their expertise exposed areas of weakness and error in my writings, and did so with obvious desires to help improve this work. For them, I am immensely grateful.
Soli Deo Gloria.
References
Anderson, J. S. 2002a. Revision of the Aïstopod genus Phlegethontia (Tetrapoda: Lepospondyli). Journal of Paleontology 76, no. 6:1029–1046.
Anderson, J. S. 2002b. A new aïstopod (Tetrapoda:Lepospondyli) from Mazon Creek, Illinois. Journal of Vertebrate Paleontology 23, no. 1:79–88.
Anderson, J. S. 2003. Cranial anatomy of Coloraderpeton brilli, postcranial anatomy of Oestocephalus amphiuminus, and reconsideration of Ophiderpetontidae (Tetrapoda: Lepospondyli: Aistopoda). Journal of Vertebrate Paleontology 23, no. 3:532–543.
Anderson, J. S. 2007. Incorporating ontogeny in the matrix: a phylogenetic evaluation of developmental evidence for the origin of modern amphibians. In Major transitions in vertebrate evolution, ed. J. S. Anderson and H.-D. Seus, pp. 182–227. Bloomington, Indiana: Indiana University Press.
Anderson, J. S., R. L. Carroll, and T. B. Rowe. 2003. New information on Lethiscus stocki (Tetrapoda: Lepospondyli: Aistopoda) from high-resolution computed tomography and a phylogenetic analysis of Aistopoda. Canadian Journal of Earth Sciences 40, no. 8:1071–1083.
Anderson, J. S., D. Scott, and R. R. Reisz. 2009. Nannaroter mckinziei, a new ostodolepid ‘microsaur’ (Tetrapoda, Lepospondyli, Recumbirostra) from the Early Permian of Richards Spur (Ft. Sill), Oklahoma. Journal of Vertebrate Paleontology 29, no. 2:379–388.
Beech, S., and M. R. Ross. 2012. A preliminary (re)estimation of the Ark-borne animals. Journal of Creation Theology and Science, Series B. 2:1.
Benton, M. J. 2005. Vertebrate palaeontology, 3rd ed. Malden, Massachusetts: Blackwell Publishing.
Bolt, J. R., and O. Rieppel. 2009. The holotype skull of Llistrofus pricei Carroll and Gaskill, 1978 (Microsauria: Hapsidopareiontidae). Journal of Paleontology 83, no. 3:471–483.
Boy, A. J., and K. Bandel. 1973. Bruktererpeton fiebigi n. gen. n. sp. (Amphibia: Gephyrostegida) der erste Tetrapode aus dem rheinisch-westfaelischen Karbon (Namur B; W. Deutschland). Palaeontographica, A 145:39–77.
Buteo, J. 1554. De arca Noe, cuius formae capacitatisque fuerit libellus, in Opera geometrica. Lyons.
Bystrow, A. P. 1944. Kotlassia prima Amalitzky. Geological Society of America Bulletin 55, no. 4:379–416.
Carlson, K. J. 1999. Crossotelos, an Early Permian nectridian amphibian. Journal of Vertebrate Paleontology 19, no. 4: 623–631.
Carroll. R. L. 1964. The relationships of the rhachitomous amphibian Parioxys. American Museum Novitates 2167:1–11.
Carroll, R. L. 1969. A new family of Carboniferous amphibians. Palaeontology 12, no. 4:537–548.
Carroll, R. L. 1970. The ancestry of reptiles. Philosophical Transactions of the Royal Society B 257, no. 814:267–308.
Carroll R. L. 1988. Vertebrate paleontology and evolution. New York, New York: W. H. Freeman and Company.
Carroll, R. L., and P. Gaskill. 1978. The order Microsauria. Memoirs of the American Philosophical Society 126:1–211.
Carroll, R. L., K. A. Bossy, A. V. Milner, S. M. Andrews, and C. F. Wellstead. 1998. Lepospondyli. Encyclopedia of paleoherpetology, vol. 1. München, Germany: Verlag Dr. Friedrich Pfeil.
Clack, J. A. 1987. Two new specimens of Anthracosaurus (Amphibia: Anthracosauria) from the Northumberland coal measures. Palaeontology 30, no. 1:15–26.
Clack, J. A. 2012. Gaining ground: The origin and evolution of the tetrapods, 2nd ed. Bloomington, Indiana: Indiana University Press.
Clack, J. A. and S. M. Finney. 2005. Pederpes finneyae, an articulated tetrapod from the Tournaisian of western Scotland. Journal of Systematic Palaeontology 2, no. 4:311–346.
Damiani, R. J. 2001. A systematic revision and phylogenetic analysis of Triassic mastodonsauroids (Temnospondyli: Stereospondyli). Zoological Journal of the Linnean Society 133, no. 4:379–482.
Damiani, R. J. and B. S. Rubidge. 2003. A review of the South African temnospondyl amphibian record. Palaeontologia Africana 39:21–36.
Fortuny, J., A. Galobart, and C. De Santisteban. 2011. A new capitosaur from the Middle Triassic of Spain and the relationships within the Capitosauria. Acta Palaeontologica Polonica 56, no. 3:553–566.
Gardner, J. D., and M. Bohme. 2008. Review of the Albanerpetontidae (lissamphibia), with comments on the paleoecological preferences of European Tertiary albanerpetontids. In Vertebrate microfossil assemblages: their role in paleoecology and paleobiogeography, ed. J. Sankey and S. Baszio, pp. 178–218. Bloomington, Indiana: Indiana University Press.
Godfrey, S. J. 1989a. The postcranial skeleton of the Carboniferous tetrapod Greererpeton burkemorani Romer, 1969. Philosophical Transactions of the Royal Society of London B 323:75–153.
Godfrey, S. J. 1989b. Ontogenetic changes in the skull of the Carboniferous tetrapod Greererpeton burkemorani Romer, 1969. Philosophical Transactions of the Royal Society of London B 323:135–153.
Godfrey, S. J., A. R. Fiorillo, and R. L. Carroll. 1987. A newly discovered skull of the temnospondyl amphibian Dendrerpeton acadianum Owen. Canadian Journal of Earth Sciences 24, no. 4:796–805.
Griffith, T., and N. Miller. 2008. Johannes Buteo’s The shape and capacity of Noah’s Ark: A translation of Johannes Bureo’s 1554 edition. Center for Origins Research Issues in Creation, no. 2. Eugene, Oregon: Wipf and Stock.
Gubin, Y. M. 1997. Skull morphology of Archegosaurus decheni Goldfuss (Amphibia, Temnospondyli) from the Early Permian of Germany. Alcheringa 21, no. 2:103–121.
Hennigan, T. 2013a. An initial estimate toward identifying and numbering amphibian kinds within the orders Caudata and Gymnophiona. Answers Research Journal 6:17–34.
Hennigan, T. 2013b. An initial estimate toward identifying and numbering the frog kinds on the Ark: Order Anura. Answers Research Journal 6:335–365.
Henrici, A. C., T. Martens, D. S. Berman, and S. S. Sumida. 2011. An ostodolepid ‘microsaur’ (Lepospondyli) from the Lower Permian Tambach Formation of central Germany. Journal of Vertebrate Paleontology 31, no. 5:997–1004.
Holmes, R. 1984. The Carboniferous amphibian Proterogyrinus scheeli Romer, and the early evolution of tetrapods. Philosphical Transactions of the Royal Society of London B 306:431–524.
Holmes, R. B. 1989. The skull and axial skeleton of the Lower Permian anthracosauroid amphibian Archeria crassidisca Cope. Palaeontographica 207:161–206.
Holmes, R. B., R. L. Carroll, and R. R. Reisz. 1998. The first articulated skeleton of Dendrerpeton acadianum (Temnospondyli: Dendrerpetontidae) from the Lower Pennsylvanian locality of Joggins, Nova Scotia and a review of its relationships. Journal of Vertebrate Paleontology 18, no. 1:64–79.
Jones, A. J., 1973. How many animals on the Ark? Creation Research Society Quarterly 10, no. 2:102–108.
Klembara, J. 2009. New cranial and dental features of Discosauriscus austriacus (Seymouriamorpha, Discosauriscidae) and the ontogenetic conditions of Discosauriscus. Special Papers in Paleontology 81:61–69.
Klembara, J. 2011. The cranial anatomy, ontogeny, and relationships of Karpinskiosaurus secundus (Amalitzky) (Seymouriamorpha, Karpinskiosauridae) from the Upper Permian of European Russia. Zoological Journal of the Linnean Society 161, no. 1:184–212.
Laurin, M. 1996. A redescription of the cranial anatomy of Seymouria baylorensis, the best known Seymouriamorph (Vertebrata: Seymouriamorpha). PaleoBios 17, no. 1:1–16.
Lightner, J., B. Hodge, G. Purdom, and T. Hennigan. 2011. Determining the Ark kinds. Answers Research Journal 4:195–201.
Lombard R. E., and J. R. Bolt. 1995. A new primitive tetrapod, Whatcheeria deltae, from the Lower Carboniferous of Iowa. Palaeontology 38, no. 3:471–494.
Milner, A. C. 1980. A review of the Nectridea (Amphibia). In The terrestrial environment and the origin of land vertebrates, ed. A. L. Panchen, pp. 377–405. London, England: Academic Press.
Milner. A. C. 1994. The aïstopod amphibian from the Viséan of East Kirkton, West Lothian, Scotland. Transactions of the Royal Society of Edinburgh: Earth Sciences 84, no. 3–4:363–368.
Milner, A. C., and W. Lindsay. 1998. Postcranial remains of Baphetes and their bearing on the relationships of the Baphetidae (=Loxommatidae). Zoological Journal of the Linnean Society 122, no. 1–2:211–235.
Milner, A. C., A. R. Milner, and S. A Walsh. 2009. A new specimen of Baphetes from Nýřany, Czech Republic and the intrinsic relationships of the Baphetidae. Acta Zoologica 90 (Supplement 1):318–334.
Milner, A. R. 1980a. The temnospondyl amphibian Dendrerpeton from the upper Carboniferous of Ireland. Paleontology 23, no. 1:125–141.
Milner, A. R. 1980b. The tetrapod assemblage from Nýrany, Czechoslovakia. In The terrestrial environment and the origin of land vertebrates, ed. A. L. Panchen, pp. 439–496. London, England: Academic Press.
Milner, A. R. 1994. Late Triassic and Jurassic amphibians: fossil record and phylogeny. In In the shadow of the dinosaurs: Early Mesozoic tetrapods, ed. N. C. Fraser, and H.-D. Sues, pp. 5–22. Cambridge, United Kingdom: Cambridge University Press.
Milner A. R., and S. E. K. Sequeria. 2003. Slaugenhopia texensis from the Permian of Texas is a primitive tupilakosaurid. Journal of Vertebrate Paleontology 24, no. 2:320–325.
Moustafa, Y. S. 1955. The skeletal structure of Parioxys ferricolus, Cope. Bulletin Institut d’Egypte 36:41–76.
Niedzwiedski, G., P. Szrek, K. Narkiewicz, M. Narkiewicz and P. E. Ahlberg. 2010. Tetrapod trackways from the early Middle Devonian period of Poland. Nature 463:43–48.
Olson, E. 1951. Fauna of upper vale and choza: 1-5. Fieldiana Geology 10, no. 11:89–128.
Olson, E. 1971. A skeleton of Lysorophus tricarinatus (Amphibia: Lepospondyli) from the Hennessey Formation (Permian) of Oklahoma. Journal of Paleontology 45, no. 3:443–449.
Olson, E. C. 1979. Aspects of the biology of Trimerorhachis (Amphibia: Temnospondyli). Journal of Paleontology 53, no. 1:1–17.
Paleobiology database. Retrived from www.fossilworks.org. on July 22, 2014.
Panchen, A. L. 1972. The skull and skeleton of Eogyrinus attheyi Watson (Amphibia: Labyrinthodontia). Philosophical Transactions of the Royal Society of London B 263, no. 851:279–326.
Panchen, A. L.1975. A new genus and species of anthracosaur amphibian from the Lower Carboniferous of Scotland and the status of Pholidogaster pisciformis Huxley. Philosophical Transactions of the Royal Society of London B 269, no. 900:581–637.
Patton, R. L. 1975. A lower Permian temnospondylous amphibian from the English Midlands. Palaeontology 18, no. 4:831–845.
Polley, B. P. and R. R. Reisz. 2011. A new Lower Permian trematopid (Temnospondyli: Dissorophoidea) from Richards Spur, Oklahoma. Zoological Journal of the Linnean Society 161, no. 4:789–815.
Rage, J.-C. and Z. Roček. 1989. Redescription of Triadobatrachus massinoti (Piveteau, 1936) an anuran amphibian from the early Triassic. Palaeontographica Abteilung A 206, no. 1–3:1–16.
Roček, Z. 2008. The Late Cretaceous frog Gobiates from Central Asia: its evolutionary status and possible phylogenetic relationships. Cretaceous Research 29, no. 4:577–591.
Romer, A. S. 1957. The appendicular skeleton of the Permian embolomerous amphibian Archeria. Contributions from the Museum of Paleontology, University of Michigan 13, no. 5:103–159.
Ross, M. R. 2013. No kind left behind. Answers 8, no. 1:26–29.
Ruta, M., A. R. Milner, and M. I. Coates. 2001. The tetrapod Caerorhachis bairdi Holmes and Carroll from the Lower Carboniferous of Scotland. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 92 no. 3:229–261.
Ruta, M., M. I Coates, and D. L. J. Quicke. 2003. Early tetrapod relationships revisited. Biological Reviews 78, no. 2:251–345.
Sawin, H. J. 1941. The cranial anatomy of Eryops megacephalus. Bulletin of the Museum of Comparative Zoology, Harvard 88:407–463.
Schoch, R. R. 2007. The evolution of metamorphosis in temnospondyls. Lethaia 35, no. 4:309–327.
Schoch, R. R. 2013. The evolution of major temnospondyl clades: an inclusive phylogenetic analysis. Journal of Systematic Palaeontology 11, no. 6: 673–705.
Schoch, R. R. 2014. Amphibian evolution: The life of early land vertebrates. West Sussex, United Kingdom: Wiley Blackwell.
Schoch, R. R. and B. S. Rubidge. 2005. The amphibamid Micropholis from the Lystrosaurus assemblage zone of South Africa. Journal of Vertebrate Paleontology 25, no. 3:502–522.
Schoch, R.M., M. Fastnacht, J. Fichter, T. Keller. 2007. Anatomy and relationships of the Triassic temnospondyls Schlerothorax. Acta Palaeontologica Polanica 52 no. 1:117–136.
Schoch, R. R. and A. R. Milner. 2008. The intrarelationships and evolutionary history of the Temnospondyl family branchiosauridae. Journal of Systematic Paleontology 6, no. 4:409–431.
Sequeira, S. E. K. 1998. The cranial morphology and taxonomy of the saurerpetontid Isodectes obtusus comb. nov. (Amphibia: Temnospondyli) from the Lower Permian of Texas. Zoological Journal of the Linnean Society 122, no. 1–2:237–259.
Sequeira, S. E. K. 2004. The skull of Cochleosaurus bohemicus Fric, a temnospondyl from the Czech Republic (Upper Carboniferous) and cochleosaurid interrelationships. Transactions of the Royal Society of Edinburgh: Earth Sciences 94:21–43.
Shishkin, M. A., I. V. Novikov, and Y. M. Gubin. 2000. Permian and Triassic temnospondyls from Russia. In The age of dinosaurs in Russia and Mongolia, ed. M. J. Benton, M. A. Shishkin, D. M. Unwin, and E. N. Kurochkin. Cambridge, United Kingdom: Cambridge University Press.
Smithson, T. R. 1985. The morphology and relationships of the Carboniferous amphibian Eoherpeton watsoni Panchen. Zoological Journal of the Linnean Society, 85, no. 4:317–410.
Steyer, J. S. 2002. The first articulated trematosaur ‘amphibian’ from the lower Triassic of Madagascar: Implications for the phylogeny of the group. Paleontology 45, no. 4:771–793.
Steyer, J. S. R. Damiani, C. A. Sidor, F. R. O’Keefe, H. C. E. Larsson, A. Maga, and O. Ide. 2006 The vertebrate fauna of the Upper Permian of Niger. IV. Nigerpeton ricqlesi (Temnospondyli: Cochleosairidae), and the Edopoid colonization of Gondwana. Journal of Vertebrate Paleontology 26, no. 1:18–28.
Warren, A. 1998. Laidleria uncovered: a redescription of Laidleria gracilis Kitching (1957), a temnospondyl from the Cynognathus Zone of South Africa. Zoological Journal of the Linnean Society 122, no. 1–2:167–185.
Warren, A., and C. Marsicano. 2000. A phylogeny of the Brachyopoidea (Temnospondyli, Stereospondyli). Journal of Vertebrate Paleontology 20, no. 3:462–483.
Warren, A. A., R. J. Damiani, and A. M. Yates. 2001. Palaeobiogeography of Australian fossil amphibians. Historical Biology 15, no. 1–2:171–179.
Werneberg, R., J. S. Steyer, G. Sommer, G. Gand, J. W. Schneider, and M. Vianey-Liaud. 2007. The earliest tupilakosaurid amphibian with diplospondylous vertebrae from the Late Permian of southern France. Journal of Vertebrate Paleontology 27, no. 1:26–30.
Whitcomb, J., and H. Morris. 1961. The Genesis Flood. Phillipsburg, New Jersey: P&R Press.
Wood, T. C., and M. J. Murray. 2003. Understanding the pattern of life. Nashville, Tennessee: B&H Publishing Group.
Woodmorappe, J., 1996. Noah’s Ark: A feasibility study. Santee, California: Institute for Creation Research.
Yates, A. M. 1999. The Lapillopsidae: a new family of small temnospondyls from the Early Triassic of Australia. Journal of Vertebrate Paleontology 19, no. 2:302–320.
Yates, A. M. 2002. A lapillopsid temnospondyl from the Early Triassic of India. Alcheringa 26, no. 2:201–208.















